Share This Page
Drug Price Trends for BIMATOPROST
✉ Email this page to a colleague
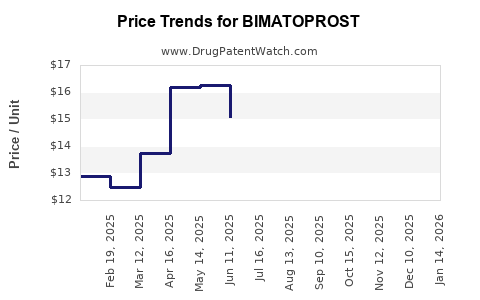
Average Pharmacy Cost for BIMATOPROST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BIMATOPROST 0.03% EYE DROPS | 62332-0507-25 | 7.79200 | ML | 2026-04-22 |
| BIMATOPROST 0.03% EYE DROPS | 60505-0583-01 | 7.82735 | ML | 2026-04-22 |
| BIMATOPROST 0.03% EYE DROPS | 62332-0507-05 | 7.82735 | ML | 2026-04-22 |
| BIMATOPROST 0.03% EYE DROPS | 42571-0128-21 | 7.82735 | ML | 2026-04-22 |
| BIMATOPROST 0.03% EYE DROPS | 60505-0583-04 | 7.79200 | ML | 2026-04-22 |
| BIMATOPROST 0.03% EYE DROPS | 42571-0128-28 | 12.26166 | ML | 2026-04-22 |
| BIMATOPROST 0.03% EYE DROPS | 42571-0128-35 | 7.79200 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BIMATOPROST
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BIMATOPROST 0.03% SOLN,OPH | Golden State Medical Supply, Inc. | 60505-0583-04 | 2.5ML | 103.97 | 41.58800 | ML | 2023-06-15 - 2028-06-14 | FSS |
| BIMATOPROST 0.03% SOLN,OPH | Golden State Medical Supply, Inc. | 60505-0583-04 | 2.5ML | 106.62 | 42.64800 | ML | 2023-06-23 - 2028-06-14 | FSS |
| BIMATOPROST 0.03% SOLN,OPH | Golden State Medical Supply, Inc. | 60505-0583-01 | 5ML | 208.35 | 41.67000 | ML | 2023-06-15 - 2028-06-14 | FSS |
| BIMATOPROST 0.03% SOLN,OPH | Golden State Medical Supply, Inc. | 60505-0583-01 | 5ML | 213.16 | 42.63200 | ML | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Bimatoprost Market Analysis and Price Projections
Executive Summary
Bimatoprost, a prostaglandin analog, is primarily utilized for treating glaucoma and ocular hypertension by reducing intraocular pressure. The global bimatoprost market is projected to reach \$1.5 billion by 2028, exhibiting a compound annual growth rate (CAGR) of 7.2%. Key growth drivers include the increasing prevalence of eye disorders, aging global populations, and advancements in ophthalmic drug delivery systems. Patent expirations have led to a competitive generic market, influencing pricing strategies. Future price trends will likely be shaped by ongoing research into new indications, novel formulations, and the entry of biosimil competitors.
What is Bimatoprost and What are its Primary Applications?
Bimatoprost is a synthetic prostamide that functions as a prostaglandin F2α analog. Its mechanism of action involves enhancing the uveoscleral outflow of aqueous humor, thereby lowering intraocular pressure (IOP). This reduction in IOP is crucial for managing conditions where elevated pressure poses a risk to the optic nerve and vision.
The primary approved indications for bimatoprost are:
- Glaucoma: Specifically, open-angle glaucoma and ocular hypertension. These conditions are characterized by damage to the optic nerve, often associated with elevated IOP. Bimatoprost is prescribed as a once-daily topical ophthalmic solution to reduce IOP in patients with these conditions.
- Hypotrichosis of Eyelashes: Bimatoprost is also approved in a lower concentration as a topical treatment to enhance the growth of eyelashes, making them longer, thicker, and darker. This application is marketed under different brand names.
Off-label uses and ongoing research explore bimatoprost's potential in other ocular conditions and its systemic effects, though these are not its primary market drivers currently.
What is the Current Market Size and Projected Growth for Bimatoprost?
The global bimatoprost market is substantial and anticipated to expand significantly.
- Current Market Size (2023 Estimate): Approximately \$980 million.
- Projected Market Size (2028 Estimate): \$1.5 billion.
- Compound Annual Growth Rate (CAGR): 7.2% (2023-2028).
This growth is underpinned by several factors:
- Increasing Ocular Disease Prevalence: The global incidence of glaucoma and ocular hypertension is rising, driven by an aging population and increased awareness of eye health. The World Health Organization estimates that over 70 million people worldwide have glaucoma, with the number expected to increase [1].
- Demographic Shifts: The elderly population, a demographic with a higher propensity for developing glaucoma, is growing. This demographic trend directly fuels demand for IOP-lowering medications like bimatoprost.
- Therapeutic Efficacy: Bimatoprost has established a strong track record as an effective monotherapy and adjunct treatment for reducing IOP, making it a preferred choice for many ophthalmologists and patients.
- Advancements in Formulations and Delivery: Ongoing research into improved drug delivery systems, such as sustained-release formulations, aims to enhance patient compliance and therapeutic outcomes, potentially expanding market share.
What is the Competitive Landscape for Bimatoprost?
The bimatoprost market is characterized by a mix of branded and generic products, with significant competition, particularly in the glaucoma segment.
Branded Products
- Lumigan® (Allergan/AbbVie): This is the original branded bimatoprost ophthalmic solution. Its patent protection has expired in major markets, leading to generic competition.
- Latisse® (Allergan/AbbVie): This is the branded product for eyelash enhancement, which also faces generic competition in some regions.
Generic Competition
The expiration of key patents for Lumigan® has opened the market to numerous generic manufacturers. This has intensified competition and significantly impacted pricing. Major generic players include:
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Apotex
- Sun Pharmaceutical Industries
- Mylan (Viatris)
- Bausch Health
The presence of multiple generic versions has led to price erosion, making bimatoprost more accessible but also necessitating competitive pricing strategies for all market participants.
Market Share Dynamics
While branded Lumigan® still holds a significant share due to established brand loyalty and physician familiarity, generic bimatoprost formulations have captured a substantial portion of the market. The eyelash enhancement segment (Latisse®) maintains a distinct market position due to its specific indication and formulation.
What are the Key Patent Expirations and Their Impact?
Patent expirations are a critical factor shaping the bimatoprost market. The primary patent for Lumigan® (US Patent No. 5,760,055), covering the composition of matter, expired in key markets, allowing for the introduction of generic versions.
- US Patent Expiration: The core composition of matter patent for bimatoprost expired in 2017 [2]. This was a pivotal event that triggered widespread generic entry.
- Formulation Patents: While the base compound patent has expired, companies may hold secondary patents on specific formulations, delivery devices, or manufacturing processes that could provide some limited protection. However, these are generally less robust than composition of matter patents.
Impact of Patent Expirations:
- Price Erosion: The most direct impact is a significant reduction in the average selling price (ASP) of bimatoprost ophthalmic solutions. Generic competition drives prices down as manufacturers compete on cost.
- Increased Market Access: Lower prices improve patient access to treatment, particularly in regions with limited healthcare budgets.
- Shift in Market Share: Branded manufacturers lose market share to generic producers. Their strategy often shifts to focusing on differentiation through branded generics, combination therapies, or exploring new indications.
- Increased R&D for Differentiation: Manufacturers may invest in developing novel bimatoprost formulations (e.g., preservative-free, sustained-release) or combination products to create new intellectual property and recapture market value.
What are the Current Pricing Trends for Bimatoprost?
Pricing for bimatoprost is highly variable, influenced by brand, generic status, volume, and geographic region.
Branded Bimatoprost (Lumigan®/Latisse®)
- Typical Price Range (per bottle, 3ml): \$80 - \$150 (US Dollars). This price reflects the costs associated with R&D, marketing, and brand premium.
- Geographic Variations: Prices can differ significantly between the US, Europe, and emerging markets due to regulatory environments, insurance coverage, and local market dynamics.
Generic Bimatoprost Ophthalmic Solutions
- Typical Price Range (per bottle, 3ml): \$20 - \$50 (US Dollars).
- Volume Discounts: Larger purchasers (e.g., hospital systems, large pharmacy chains) may negotiate lower prices through bulk purchasing agreements.
- Competition Intensity: In markets with a high number of generic manufacturers, prices can fall towards the lower end of this range.
Factors Influencing Pricing:
- Prescription Volume: Higher prescription volumes can lead to lower per-unit costs for providers.
- Insurance Reimbursement: Payer policies and formulary placement significantly impact out-of-pocket costs for patients and wholesale pricing.
- Regulatory Landscape: Approval processes and pricing regulations in different countries affect market entry and pricing strategies.
- Manufacturing Costs: Efficiency in production and supply chain management by generic manufacturers directly impacts their pricing competitiveness.
What are the Future Price Projections for Bimatoprost?
Future price projections for bimatoprost will be shaped by several evolving market forces.
- Continued Generic Dominance: The generic market for bimatoprost in its established indications is mature. Price competition is expected to remain intense, likely leading to a slight downward trend or stabilization in ASP for generic versions.
- Entry of Biosimilar/Advanced Generic Formulations:
- Preservative-Free Formulations: These are gaining traction due to concerns about preservative-related ocular surface toxicity. Companies offering preservative-free bimatoprost may command a modest premium over standard generic formulations, though this premium is likely to decrease over time as competition emerges.
- Sustained-Release Technologies: Innovations in drug delivery that reduce dosing frequency (e.g., from once daily to once weekly or monthly) could command a significant initial premium. However, the ultimate pricing will depend on manufacturing costs, clinical benefits, and payer acceptance. If successful, these could create new market segments with higher ASPs.
- New Indications and Combinations:
- If bimatoprost is successfully repurposed for new ocular diseases or developed in novel fixed-dose combinations with other IOP-lowering agents, these products could achieve higher price points initially, reflecting their innovation and expanded therapeutic value.
- For example, combination therapies with other classes of glaucoma drugs are already established and priced at a premium compared to single agents.
- Emerging Markets: As healthcare infrastructure and access improve in emerging markets, bimatoprost (both branded and generic) will see increased volume, potentially leading to price adjustments to suit local purchasing power. This could involve tiered pricing strategies.
- Manufacturing Efficiencies: Continued advancements in chemical synthesis and pharmaceutical manufacturing could lead to marginal cost reductions for generic manufacturers, potentially driving prices lower.
- Regulatory Hurdles and Market Access: Approval timelines and reimbursement decisions by regulatory bodies and payers will influence the speed and extent of market adoption for new formulations or indications, thereby impacting pricing.
Overall Projection:
- Standard Generic Ophthalmic Solutions: Expect continued low-to-mid single-digit percentage decline per annum in ASP due to intense competition, or stabilization as market share reaches equilibrium.
- Novel Formulations (e.g., Preservative-Free, Sustained-Release): These are projected to launch at a 15-30% premium over standard generics. This premium will likely erode by 5-10% annually as competitors enter.
- Combination Products/New Indications: Potential for 20-50% higher ASPs compared to single-agent generics, contingent on demonstrated clinical superiority and value.
The total market value growth will depend on the successful introduction and adoption of these newer, potentially higher-priced, formulations and indications, balancing against the ongoing price pressure in the established generic market.
What are the Key Future Market Drivers and Challenges?
Market Drivers
- Aging Global Population: The increasing number of individuals aged 60 and above is a primary driver for glaucoma and ocular hypertension treatments.
- Increasing Awareness and Diagnosis: Greater public awareness of eye health and improved diagnostic capabilities are leading to earlier detection and treatment of ocular diseases.
- Rising Incidence of Chronic Diseases: Conditions such as diabetes and hypertension are linked to an increased risk of glaucoma, further expanding the patient pool.
- Innovation in Drug Delivery Systems: Development of preservative-free formulations, sustained-release technologies, and novel administration devices can enhance patient adherence and therapeutic outcomes, driving market growth.
- Emerging Markets Growth: Increasing healthcare expenditure and access to advanced medical treatments in developing economies present significant growth opportunities.
- Research into New Indications: Exploration of bimatoprost for other ocular conditions or even non-ocular applications could unlock new market segments.
Market Challenges
- Intense Generic Competition: The mature generic market for bimatoprost leads to significant price erosion, limiting revenue growth for established products.
- Reimbursement Pressures: Payers and healthcare systems are increasingly scrutinizing drug costs, leading to tighter formulary controls and demanding evidence of cost-effectiveness.
- Development of Alternative Therapies: Ongoing research into entirely new classes of glaucoma medications or non-pharmacological treatments (e.g., surgical interventions, laser therapy) could present future competition.
- Ocular Surface Toxicity Concerns: Preservatives in traditional ophthalmic solutions can cause ocular surface disease. While preservative-free options are emerging, managing patient side effects remains a consideration.
- Regulatory Approval Pathways: Obtaining approval for new formulations or indications can be lengthy and costly, posing a challenge for market entry.
- Supply Chain Volatility: Global supply chain disruptions can impact the availability and cost of raw materials and finished products.
Key Takeaways
- The bimatoprost market is projected to grow to \$1.5 billion by 2028, driven by an aging population and rising prevalence of eye disorders.
- Patent expirations have resulted in a highly competitive generic market, leading to significant price erosion for standard ophthalmic solutions.
- Future growth will depend on the adoption of novel formulations (e.g., preservative-free, sustained-release) and potential new indications, which may command premium pricing.
- Intense generic competition and reimbursement pressures will continue to challenge pricing strategies.
- Emerging markets represent a significant opportunity for volume expansion.
Frequently Asked Questions
-
What is the primary mechanism of action for bimatoprost in treating glaucoma? Bimatoprost enhances the uveoscleral outflow of aqueous humor, thereby reducing intraocular pressure.
-
How has the expiration of bimatoprost patents affected market pricing? Patent expirations have led to a significant increase in generic competition, causing substantial price erosion for bimatoprost ophthalmic solutions.
-
What are the key factors driving future growth in the bimatoprost market? Growth will be driven by an aging global population, increasing prevalence of eye disorders, advancements in drug delivery systems, and the development of novel formulations and potential new indications.
-
Are there any significant risks or challenges facing the bimatoprost market? Key challenges include intense generic competition, reimbursement pressures from payers, the development of alternative therapies, and potential ocular surface toxicity concerns with certain formulations.
-
What is the projected difference in price between branded bimatoprost and its generic versions? Branded bimatoprost typically ranges from \$80 to \$150 per 3ml bottle, while generic versions commonly sell for \$20 to \$50 per 3ml bottle, reflecting a substantial price difference.
Citations
[1] World Health Organization. (n.d.). Blindness and vision impairment. Retrieved from [WHO Website]
[2] United States Patent and Trademark Office. (n.d.). Patent Number 5,760,055. Retrieved from USPTO Patent Database
More… ↓
