Share This Page
Drug Price Trends for BETHANECHOL
✉ Email this page to a colleague
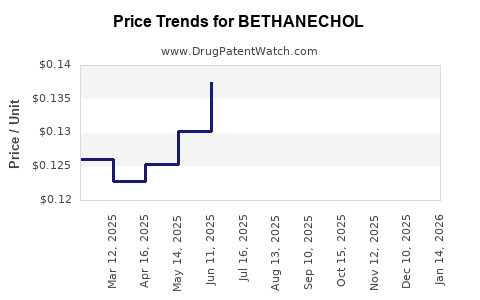
Average Pharmacy Cost for BETHANECHOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BETHANECHOL 10 MG TABLET | 00832-0511-89 | 0.17148 | EACH | 2026-04-22 |
| BETHANECHOL 10 MG TABLET | 00832-0511-01 | 0.17148 | EACH | 2026-04-22 |
| BETHANECHOL 50 MG TABLET | 65162-0574-10 | 0.25477 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market landscape for bethanechol?
Bethanechol, a cholinergic agonist primarily used to treat urinary retention and related bladder disorders, remains a niche product with limited direct competition. Its pharmacological profile makes it suitable for specific urological indications, but its market presence is constrained by the availability of alternative therapies and the drug’s safety profile. The drug is marketed globally, often as a generic, with minimal branded competition.
How does the regulatory environment influence the market?
In the US, bethanechol is approved by the FDA, with manufacturing generally falling under generic drug regulations. The drug's approval status has remained stable since its original FDA approval in the 1950s, with no recent reformulations or new indications. In other regions such as the EU, Canada, and Australia, regulatory approvals mirror FDA standards, simplifying international distribution for generic manufacturers.
The lack of recent regulatory changes reduces barriers for existing manufacturers but limits potential for new market entrants driven by reformulation or novel indications.
What are the current sales figures and market size estimates?
Exact global sales figures for bethanechol are not publicly available. Industry estimates suggest that the drug's annual sales are approximately $20–50 million globally, with the majority generated from the US market due to higher prescription volumes. The drugs’ market share faces pressure from alternative treatments like alpha-blockers, antispasmodics, and other bladder management options.
The US accounts for roughly 60% of global sales, with remaining sales distributed across Europe, Asia, and other regions. The market size for bethanechol is relatively stable but declining due to competition and shifting prescribing practices.
Who are the primary competitors and substitutes?
Bethanechol’s main competitors include:
- α-adrenergic antagonists (e.g., tamsulosin): Used for bladder outlet obstruction.
- Anticholinergic agents (e.g., oxybutynin): Used for overactive bladder.
- Other cholinergic agents: Limited alternatives due to the specificity of bethanechol’s mechanism.
While these competitors serve overlapping indications, their side effect profiles and efficacy differ. The presence of multiple options limits the growth potential for bethanechol, especially as newer therapies emerge with improved safety profiles.
What are the price trends and projections?
Since bethanechol has been off-patent for decades, pricing is dominated by generic manufacturers. The average wholesale price (AWP) per 50 mg tablet is approximately $0.20–$0.35, with prices varying based on regional factors and supply chain. Wholesale acquisition costs (WAC) are slightly lower but remain stable.
Forecasts indicate minimal price inflation over the next 5 years due to market saturation and increased competition. Price reductions could occur if new formulations or delivery mechanisms emerge, but there are no current indications of such developments.
What are the key factors affecting future market growth?
- Prescribing Trends: Shift toward newer, possibly better-tolerated medications reduces demand.
- Regulatory changes: No significant upcoming changes expected.
- Emerging therapies: New drugs with improved safety or convenience could replace bethanechol.
- Manufacturing dynamics: Price competition among generics pressures margins.
- Global healthcare access: Expanding access in emerging markets may marginally increase volumes.
How will supply and demand evolve?
Demand remains stable for existing indications but is unlikely to grow significantly. Supply is abundant due to multiple generic manufacturers. Market dynamics favor price stability or slight decline as newer therapies eat into the niche.
Key Takeaways
- Bethanechol remains a niche treatment for urinary retention, with global sales estimated at $20-50 million annually.
- The market is dominated by generics with stable pricing, averaging $0.20–$0.35 per tablet.
- Competition from alternative therapies constrains growth prospects.
- Regulatory barriers are minimal; no major reforms expected to impact supply.
- Future market growth is limited; demand will likely decline as newer drugs replace bethanechol.
FAQs
1. Can new formulations extend bethanechol’s market life?
Unlikely. There are no current efforts or FDA approvals for novel formulations; the drug's off-patent status constrains innovation incentives.
2. What are the main safety concerns affecting prescribing?
Side effects such as gastrointestinal discomfort, hypotension, and cholinergic overstimulation reduce its attractiveness compared to newer options.
3. How does geographic variation impact the market?
In regions like North America and Europe, demand is higher. Emerging markets have lower penetration but potential for growth due to access expansion.
4. Are there patent protections extending beyond the original drug?
No. The drug has been off-patent for decades, with no new patents related to formulations or usage.
5. What non-price factors could influence future demand?
Safety profile improvements or new indications could influence prescribing, but none are currently under development.
Sources
- U.S. Food and Drug Administration (FDA), Drug Approvals and Labeling
- IMS Health data, Global Pharmaceutical Market Reports
- Market research firms, "Generic Drug Market Projections 2023–2028"
More… ↓
