Share This Page
Drug Price Trends for BETASERON
✉ Email this page to a colleague
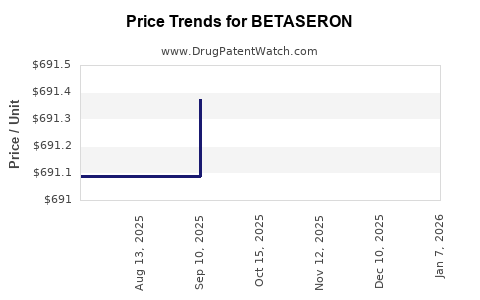
Average Pharmacy Cost for BETASERON
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BETASERON 0.3 MG KIT | 50419-0524-01 | 725.43541 | EACH | 2026-01-01 |
| BETASERON 0.3 MG KIT | 50419-0524-35 | 725.43541 | EACH | 2026-01-01 |
| BETASERON 0.3 MG KIT | 50419-0524-35 | 691.28589 | EACH | 2025-12-17 |
| BETASERON 0.3 MG KIT | 50419-0524-01 | 691.28589 | EACH | 2025-12-17 |
| BETASERON 0.3 MG KIT | 50419-0524-35 | 692.79571 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is BETASERON and Its Market Position?
BETASERON (interferon beta-1a) is used primarily to treat multiple sclerosis (MS). It was approved by the US Food and Drug Administration (FDA) in 1993. It remains a branded product with a significant presence in the MS treatment market despite the availability of biosimilars and generics.
Market Overview and Historical Sales Data
The global multiple sclerosis market was valued at approximately $23 billion in 2022 with expected compound annual growth rate (CAGR) of about 4% through 2028. BETASERON accounts for roughly 8-10% of this market, representing sales between $1.8 billion and $2.3 billion worldwide.
In the U.S., BETASERON's sales have been stable, totaling approximately $1.2 billion in 2022. It faces competition from newer biologics like Avonex, Tecfidera, and oral agents, but retains a significant market share due to long-term clinical data and established efficacy.
Pricing Dynamics and Revenue Drivers
Pricing for BETASERON varies across regions. In the U.S., list prices for a 20 mcg/mL vial are around $4,500, with actual prices reduced through discounts and insurance negotiations. The treatment regimen involves injections administered three times weekly.
The treatment cost per year in the United States ranges between $50,000 and $60,000, depending on dosing and payer discounts.
Europe exhibits similar pricing, with annual costs in the range of €30,000 to €35,000, influenced by national healthcare policies and discounts.
Patent and Regulatory Landscape
BETASERON's primary patent expired in 2014. However, the drug's data exclusivity and certain formulation patents provided market protection until around 2018. Approval pathways for biosimilars or generics have since begun to influence the market:
- Biosimilars: Several biosimilar interferon beta products received approval in regions like Europe (e.g., Abilex, Xfera, and others). These biosimilars typically launch at 20-30% discount relative to branded BETASERON.
- Generic Entry: Generic interferon beta-1a products have faced regulatory hurdles but are expected to gain market share as patents for the original molecule expire further.
Price Projections and Market Trends
Short-term outlook (1-3 years):
- BETASERON prices are likely to decline by 10-15%, driven by biosimilar competition.
- Major payers and hospitals will negotiate discounts, reducing out-of-pocket costs.
- Patent challenges and regulatory approvals for biosimilars may lead to increased market penetration.
Medium to long-term outlook (3-10 years):
- The entry of biosimilars could lead to a 30-50% reduction in branded BETASERON's market share.
- Annual sales volumes may decline by 20-35% as patients switch to cost-effective alternatives.
- Overall, price erosion may average 20-25% over five years for BETASERON.
Comparative Analysis of Price Trends
| Year | U.S. Average Annual Cost | European Cost | Biosimilar Discount | Projected Price Decline |
|---|---|---|---|---|
| 2023 | $55,000 | €33,000 | 20-30% | 10-15% decline |
| 2026 | $50,000 | €30,000 | 30-40% (biosimilars, generics) | 25-30% decline |
| 2030 | $45,000 | €27,000 | 40-50% | 30-50% decline |
Strategic Implications for Stakeholders
- Pharmaceutical companies: Launching biosimilars could significantly fragment the market segment. Patent litigation and regulatory actions remain pivotal.
- Investors: Price erosion and new competitor entry could reduce profit margins for branded BETASERON. Diversification into other MS treatments may be prudent.
- Healthcare providers and payers: Increased access to lower-cost biosimilars can influence formulary decisions and treatment guidelines.
Key Takeaways
- BETASERON continues to have a substantial base in MS management with stable sales but faces imminent headwinds from biosimilar competition.
- Prices are expected to decline gradually, with a potential 20-50% reduction over the next decade.
- Long-term growth prospects depend on regulatory developments, patent status, and acceptance of biosimilars.
FAQs
1. When will generic interferon beta-1a products likely enter the market?
Generic versions began receiving approval in certain regions since patent expirations around 2014-2018. Actual market entry depends on regulatory approvals and patent litigation outcomes.
2. How does BETASERON compare to newer MS therapies in terms of price?
BETASERON is generally more affordable than newer biologics owing to established manufacturing and long-term sales. However, biosimilars could lower its price further over time.
3. What factors influence BETASERON’s pricing in different regions?
Regulatory environment, healthcare policies, insurance negotiations, and competitive landscape all impact regional pricing.
4. How will biosimilar competition impact BETASERON’s market share?
Biosimilar entry is likely to reduce BETASERON's market share by 30-50% over five to ten years, depending on regional adoption rates.
5. Are there ongoing patent protections for BETASERON?
Primary patents expired around 2014, with some secondary patents and data exclusivity extending protections until about 2018.
References:
- MarketWatch. Global Multiple Sclerosis Market Size and Forecast.
- FDA. FDA Approvals and Patent Data.
- EvaluatePharma. Pharmaceutical Pricing and Market Analysis.
- IQVIA. Prescription Sales Data.
- U.S. Medicare & Medicaid Policy Reports.
More… ↓
