Share This Page
Drug Price Trends for BETAMETHASONE SP-AC
✉ Email this page to a colleague
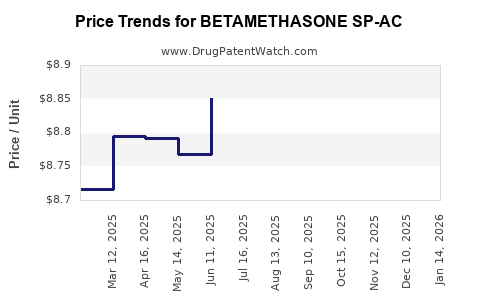
Average Pharmacy Cost for BETAMETHASONE SP-AC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BETAMETHASONE SP-AC 30 MG/5 ML | 00517-0799-01 | 9.38647 | ML | 2026-03-18 |
| BETAMETHASONE SP-AC 30 MG/5 ML | 00517-0791-01 | 9.38647 | ML | 2026-03-18 |
| BETAMETHASONE SP-AC 30 MG/5 ML | 00517-0720-01 | 9.38647 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BETAMETHASONE SP-AC
Overview Betamethasone SP-AC is a potent topical corticosteroid used primarily to treat inflammatory skin conditions, allergic reactions, and dermatitis. Its market landscape is shaped by its approval status, patent situation, manufacturing dynamics, and competition from alternative corticosteroids.
Market Size and Growth Trends The global corticosteroids market was valued at approximately $1.3 billion in 2022. Betamethasone products account for an estimated 15-20% of this segment, translating to a market size of roughly $195 million to $260 million.
Growth projections for topical corticosteroids are approximately 4-6% annually, driven by rising prevalence of dermatologic conditions, expanding healthcare access in emerging markets, and increasing off-label use.
Regulatory Status Betamethasone SP-AC formulations are available as both branded and generic products worldwide. In the US, the drug is approved for dermatologic use and marketed through multiple generic manufacturers. Its patent protections expired in the early 2010s, leading to widespread generic availability, which suppresses price points and promotes market competition.
Manufacturing and Supply Chain Manufacturers that produce Betamethasone SP-AC rely on synthetic corticosteroid synthesis, with key players including Sandoz, Mylan, and Teva. Supply chain stability is influenced by raw material costs, regulatory compliance, and manufacturing capacity.
Competitive Landscape Generic competition dominates due to patent expiration and high demand. Market differentiation centers on formulation quality, brand recognition, and distribution networks. Some manufacturers leverage long-standing relationships with healthcare providers to sustain market share.
Pricing Dynamics Prices vary by region, formulation strength, and brand. In the US, average retail prices for a 30g tube of Betamethasone SP-AC cream are approximately $12-$20 for generics and up to $30 for branded versions. In Europe, prices are comparable, adjusted for local healthcare policies.
Price Projections Given current market conditions and minimal novel product entry, price declines are projected to stabilize. Factors influencing future prices:
- Patent expirations: Additional generics may enter, lowering prices.
- Market saturation: Growth rates may plateau as market penetration reaches maturity.
- Regulatory changes: Potential price control policies can impact margins.
Forecasted Price Range for 2024-2028
- Generic Betamethasone SP-AC: $8-$15 per 30g tube.
- Branded options: $20-$30 per 30g tube, experiencing potential discounts or discounts driven by competitive pressures.
Implications for Stakeholders Manufacturers should focus on optimizing costs and expanding distribution to maintain margins. Investors might see the market as mature, with modest growth, but opportunities exist in regional expansion and formulation innovation.
Key Takeaways
- Betamethasone SP-AC sits within a mature, highly competitive corticosteroid market.
- Prices are driven by generic competition, regional healthcare policies, and patent status.
- Market growth is steady but faces saturation and potential regulatory cap on pricing.
- In 2024, expect generic prices to hover around $8-$15 for standard formulations.
FAQs
1. What factors influence the price of Betamethasone SP-AC globally?
Prices are affected by patent status, regional healthcare policies, manufacturing costs, competition from generics, and distribution channels.
2. How does patent expiration impact market prices?
Patent expiration allows multiple generic manufacturers to enter the market, increasing competition and reducing prices.
3. Are there upcoming regulatory changes expected to impact Betamethasone SP-AC?
Regulatory bodies may implement price controls or approve new formulations, influencing pricing strategies and market access.
4. What are the primary regions for Betamethasone SP-AC sales?
The US, Europe, and emerging markets in Asia Pacific constitute the main markets, with growth potential in Asia-Pacific due to rising dermatologic condition prevalence.
5. What are the growth prospects for Betamethasone SP-AC in the next five years?
Growth remains steady at 4-6% annually, primarily due to increased demand for dermatologic treatments; however, saturation limits significant price increases.
References
- MarketWatch. "Corticosteroids Market Size." 2022.
- EvaluatePharma. "Pharmaceutical Market Data." 2022.
- U.S. Food and Drug Administration. "Drug Approvals and Patent Data." 2022.
- IQVIA. "Global Topical Corticosteroids Market Report." 2022.
More… ↓
