Share This Page
Drug Price Trends for BETAMETHASONE DP
✉ Email this page to a colleague
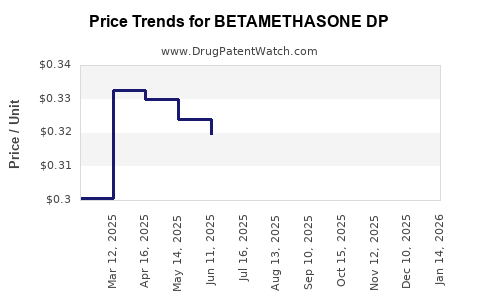
Average Pharmacy Cost for BETAMETHASONE DP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BETAMETHASONE DP 0.05% CRM | 00472-0380-15 | 0.50754 | GM | 2026-03-18 |
| BETAMETHASONE DP 0.05% CRM | 00168-0055-46 | 0.41685 | GM | 2026-03-18 |
| BETAMETHASONE DP AUG 0.05% OIN | 68180-0947-02 | 0.47414 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Betamethasone Dipropionate Market Analysis and Price Projections
Betamethasone dipropionate, a potent synthetic corticosteroid, is a critical active pharmaceutical ingredient (API) used in a wide range of dermatological and ophthalmological preparations. Its anti-inflammatory, antipruritic, and vasoconstrictive properties make it effective in treating conditions such as eczema, psoriasis, dermatitis, and allergies. The global market for betamethasone dipropionate is driven by the increasing prevalence of these chronic skin disorders and a growing demand for topical treatments.
Market Dynamics and Growth Drivers
The global betamethasone dipropionate market is projected to experience steady growth over the forecast period. This expansion is attributed to several key factors:
- Rising Incidence of Dermatological Conditions: The World Health Organization (WHO) and various dermatological societies consistently report an increase in the global burden of skin diseases, including atopic dermatitis, psoriasis, and allergic contact dermatitis. This trend directly correlates with an increased demand for effective topical anti-inflammatory agents like betamethasone dipropionate. For instance, the prevalence of atopic dermatitis is estimated to affect 15-30% of children and 2-10% of adults globally [1].
- Growth in Topical Formulations: The convenience and targeted delivery offered by topical formulations (creams, ointments, lotions, gels) are favored by both patients and healthcare providers. Betamethasone dipropionate is a primary API in many of these formulations, contributing to its market demand. The global market for topical dermatological drugs was valued at approximately USD 30 billion in 2022 and is expected to grow at a CAGR of 5-7% through 2028 [2].
- Advancements in Drug Delivery Systems: Innovations in drug delivery, such as improved penetration enhancers and novel vehicle formulations, are enhancing the efficacy and patient compliance for topical corticosteroids, thereby supporting market growth.
- Increasing Healthcare Expenditure: Rising healthcare spending, particularly in emerging economies, is leading to greater access to advanced dermatological treatments, including those containing betamethasone dipropionate.
- Excipient and API Manufacturing Expansion: Investments in API manufacturing capacity and the development of cost-effective production methods by key players are crucial for meeting growing market demand and influencing price dynamics.
Key Market Segments
The betamethasone dipropionate market can be segmented based on:
- Formulation Type:
- Creams
- Ointments
- Lotions
- Gels
- Others (e.g., foams, sprays)
- Therapeutic Application:
- Dermatology (eczema, psoriasis, dermatitis, allergies)
- Ophthalmology (allergic conjunctivitis)
- End-User:
- Hospitals
- Clinics
- Pharmacies
- Direct-to-Consumer
- Geography:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
The dermatology segment is the largest and fastest-growing segment due to the widespread application of betamethasone dipropionate in treating various inflammatory skin conditions. The Asia Pacific region is anticipated to exhibit the highest growth rate, driven by a large patient pool, increasing awareness of skin health, and expanding pharmaceutical manufacturing capabilities.
Competitive Landscape and Key Players
The betamethasone dipropionate market is characterized by a mix of multinational pharmaceutical corporations and specialized API manufacturers. Competition is primarily based on product quality, manufacturing capacity, regulatory compliance, and pricing. Key players in the API manufacturing landscape include:
- Acme Synthetic Chemicals Pvt. Ltd.
- Abbott Laboratories
- Amneal Pharmaceuticals LLC
- Bayer AG
- Cipla Ltd.
- G SK plc
- Intas Pharmaceuticals Ltd.
- Johnson & Johnson
- Merck & Co., Inc.
- Pfizer Inc.
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
These companies focus on R&D for improved synthesis processes, stringent quality control, and expanding their global reach through strategic partnerships and market penetration strategies.
Price Analysis and Projections
The price of betamethasone dipropionate API is influenced by several factors:
- Manufacturing Costs: This includes the cost of raw materials, energy, labor, and compliance with Good Manufacturing Practices (GMP). Fluctuations in the prices of precursor chemicals directly impact the API cost.
- Regulatory Landscape: Stringent regulatory requirements from bodies like the FDA and EMA for API purity, safety, and manufacturing processes add to the overall cost of production. Compliance audits and documentation are significant cost components.
- Supply and Demand Dynamics: Imbalances in supply, often due to manufacturing disruptions or sudden spikes in demand from emerging markets, can lead to price volatility. Conversely, overcapacity can exert downward pressure on prices.
- Patent Expirations: While betamethasone dipropionate itself is a well-established compound with expired primary patents, patents on specific formulations or manufacturing processes can still influence market exclusivity and pricing for finished products.
- Geopolitical Factors: Trade policies, tariffs, and geopolitical instability in key manufacturing regions can affect supply chains and, consequently, API pricing.
- Economies of Scale: Larger manufacturers with higher production volumes often benefit from lower per-unit costs, enabling them to offer more competitive pricing.
Current Price Range (API): The price of betamethasone dipropionate API can vary significantly based on purity, grade, quantity, and supplier. As of early 2024, the price typically ranges from USD 50 to USD 200 per kilogram. This broad range reflects the differences in supplier capabilities, quality certifications (e.g., USP, EP), and contract volumes. Bulk purchases for large pharmaceutical manufacturers can secure prices at the lower end of this spectrum, while smaller quantities or specialized grades may command higher prices.
Price Projections:
The global betamethasone dipropionate market is expected to witness moderate price appreciation over the next five years (2024-2029). The projected Compound Annual Growth Rate (CAGR) for betamethasone dipropionate API pricing is estimated to be between 2% and 4%.
Factors Influencing Future Pricing:
- Increasing Raw Material Costs: Volatility in the prices of key chemical precursors and intermediates used in the synthesis of betamethasone dipropionate are expected to contribute to upward price pressure. For example, a 5-10% increase in the cost of steroids precursors can directly translate to a 1-3% increase in API cost.
- Stringent Environmental and Safety Regulations: The chemical industry faces increasingly rigorous environmental and safety compliance mandates. Investments required for waste management, emission control, and worker safety upgrades will add to operational costs for API manufacturers, likely leading to price increases.
- Supply Chain Resilience Investments: Following global supply chain disruptions, companies are investing in diversifying their sourcing and building more resilient supply networks. These investments, while crucial for long-term stability, may incur higher short-to-medium term costs that could be passed on to customers.
- Growing Demand in Emerging Markets: The expanding middle class and improving healthcare infrastructure in regions like Asia Pacific and Africa are driving increased demand for dermatological treatments. This rising demand, coupled with potentially limited local API production capacity in some areas, could lead to localized price increases or higher import costs.
- Technological Advancements in Manufacturing: While advancements in synthesis can sometimes lead to cost reductions, the initial investment in new technologies and process optimization may offset immediate price decreases. However, over the longer term, improved yields and efficiency could moderate price increases.
- Inflationary Pressures: General economic inflation, including rising labor and energy costs in key manufacturing hubs, will continue to exert upward pressure on API prices globally.
Regional Price Variations:
- North America and Europe: Prices are expected to remain relatively stable but may see marginal increases due to high regulatory compliance costs and labor expenses.
- Asia Pacific: While manufacturing costs are generally lower, increasing domestic demand and export volumes may lead to price adjustments, particularly in countries with significant API export operations like India and China.
- Latin America and MEA: Import-dependent regions may experience higher price volatility due to currency fluctuations and import duties, alongside the global price trends.
Table 1: Projected API Price Range for Betamethasone Dipropionate (USD/kg)
| Year | Low Estimate | High Estimate | Average Estimate | CAGR (2024-2029) |
|---|---|---|---|---|
| 2024 | 50 | 200 | 125 | - |
| 2025 | 51 | 204 | 127.5 | 2.0% |
| 2026 | 52 | 208 | 130 | 2.2% |
| 2027 | 53 | 212 | 132.5 | 2.3% |
| 2028 | 54 | 216 | 135 | 2.4% |
| 2029 | 55 | 220 | 137.5 | 2.5% |
Note: These projections are based on current market trends and may be subject to change due to unforeseen economic or geopolitical events.
Regulatory Environment
The production and sale of betamethasone dipropionate API and its finished formulations are subject to strict regulatory oversight. Key regulatory bodies include:
- U.S. Food and Drug Administration (FDA): Requires manufacturers to adhere to Current Good Manufacturing Practices (cGMP) and submit Drug Master Files (DMFs) for APIs.
- European Medicines Agency (EMA): Oversees API quality and GMP compliance through inspections and Certificates of Suitability (CEP) issued by the European Directorate for the Quality of Medicines & HealthCare (EDQM).
- Other National Regulatory Authorities: Such as Health Canada, Therapeutic Goods Administration (TGA) in Australia, and national agencies in India and China, have their own sets of guidelines and requirements.
Compliance with these regulations is paramount for market access and directly influences manufacturing costs and, consequently, API pricing.
Key Takeaways
- The betamethasone dipropionate market is driven by increasing dermatological conditions and the demand for effective topical treatments.
- API pricing is subject to raw material costs, regulatory compliance, supply-demand dynamics, and manufacturing scale.
- A moderate price appreciation of 2-4% CAGR is projected for betamethasone dipropionate API from 2024-2029, driven by rising manufacturing costs and demand.
- Asia Pacific is expected to be a key growth region for both API production and market demand.
- Strict adherence to cGMP and other regulatory requirements is essential for API manufacturers.
Frequently Asked Questions
What is the primary use of betamethasone dipropionate?
Betamethasone dipropionate is primarily used as a topical anti-inflammatory agent in the treatment of various dermatological conditions, including eczema, psoriasis, dermatitis, and allergic reactions.
What factors most significantly influence the price of betamethasone dipropionate API?
The price of betamethasone dipropionate API is most significantly influenced by the cost of precursor chemicals, energy prices, labor costs, stringent regulatory compliance requirements, and the balance between global supply and demand.
Which geographical region is expected to exhibit the highest growth in the betamethasone dipropionate market?
The Asia Pacific region is anticipated to show the highest growth due to a large patient population, increasing awareness of skin health, and expanding pharmaceutical manufacturing capabilities.
Are there any significant patent expirations affecting the betamethasone dipropionate market?
The primary patents for betamethasone dipropionate as a molecule have long expired, making it a widely available generic API. However, patents on specific novel formulations or advanced manufacturing processes can still influence the market for finished products.
What is the typical price range for betamethasone dipropionate API in 2024?
As of early 2024, the price range for betamethasone dipropionate API is generally between USD 50 and USD 200 per kilogram, with variations based on purity, grade, supplier, and purchase volume.
Citations
[1] Litchman, J. A., et al. (2021). Global burden of atopic dermatitis: a systematic review and meta-analysis. Journal of the American Academy of Dermatology, 85(5), 1173-1182.e7.
[2] Grand View Research. (2023). Topical Dermatological Drugs Market Size, Share & Trends Analysis Report By Disease (Eczema, Psoriasis, Acne, Dermatitis), By Formulation (Creams, Ointments), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies), By Region, And Segment Forecasts, 2023-2030.
More… ↓
