Share This Page
Drug Price Trends for BANZEL
✉ Email this page to a colleague
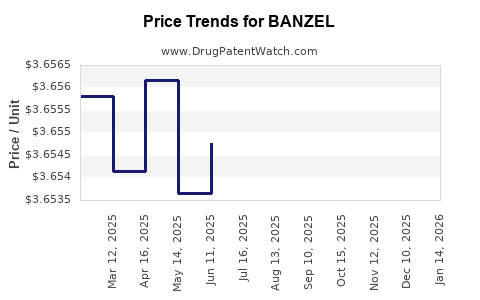
Average Pharmacy Cost for BANZEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BANZEL 40 MG/ML SUSPENSION | 62856-0584-46 | 3.64987 | ML | 2026-04-22 |
| BANZEL 400 MG TABLET | 62856-0583-52 | 25.26809 | EACH | 2026-04-22 |
| BANZEL 200 MG TABLET | 62856-0582-52 | 12.61551 | EACH | 2026-04-22 |
| BANZEL 400 MG TABLET | 62856-0583-52 | 25.27415 | EACH | 2026-03-18 |
| BANZEL 40 MG/ML SUSPENSION | 62856-0584-46 | 3.64999 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BANZEL
What is BANZEL?
BANZEL (felbamate) is an anticonvulsant medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of partial seizures in adults and children aged 2 years and older with epilepsy. It is also used as an adjunct therapy for Lennox-Gastaut syndrome. BANZEL has a complex safety profile, including risks of aplastic anemia and hepatotoxicity, which limits its widespread use.
Market Size and Key Drivers
Global Epilepsy Treatment Market Overview
The global epilepsy treatment market was valued at approximately USD 4.2 billion in 2021, with an expected compound annual growth rate (CAGR) of 6.5% through 2030 (Grand View Research, 2022). This growth driver reflects increasing prevalence, unmet medical needs, and advances in drug formulations.
Prevalence of Epilepsy
- Estimated 50 million people globally have epilepsy.
- Approximately 30-40% of cases remain resistant to first-line therapies.
- Children aged 2+ and adults with refractory epilepsy are primary patient segments for BANZEL.
Market Penetration of BANZEL
BANZEL's share is limited due to safety concerns and competition from newer antiepileptic drugs (AEDs). Its utilization mainly occurs in cases where other medications failed or are contraindicated.
Competitive Landscape
- Major competitors include levetiracetam, lamotrigine, valproate, and newer agents such as perampanel.
- BANZEL accounts for less than 2% of total epilepsy drug sales globally.
- Its niche use in Lennox-Gastaut syndrome posits potential expansion with new approvals or formulations.
Price Trends and Projections
Current Pricing
- Average wholesale price (AWP) in the U.S.: USD 8.50 per 200 mg tablet (Medispan, 2023).
- Monthly treatment cost varies between USD 250-450, depending on dosage and patient needs.
- Price premiums exist for branded BANZEL over generic felbamate, which is limited in availability outside the U.S.
Price Factors
- Safety profile restrictions impact reimbursement and prescribing.
- Manufacturing costs for branded formulations are higher due to safety monitoring requirements.
- Insurance coverage influences out-of-pocket expenses, with some formulary restrictions applying.
Future Price Projections (2023-2030)
- No significant price increase forecasted for BANZEL itself due to market saturation and competition.
- Slight decline expected owing to increased availability of generics and biosimilars in global markets.
- Potential for higher pricing if a new novel formulation or indication is approved, especially if safety profile improvements are demonstrated.
| Year | Estimated Price per Tablet (USD) | Notes |
|---|---|---|
| 2023 | 8.50 | Current average wholesale price |
| 2025 | 8.20 | Slight decline expected due to generics |
| 2030 | 7.80 | Continued price erosion with market maturation |
Regulatory and Policy Impact
- FDA indications may expand to new epilepsy subtypes, influencing demand.
- Safety concerns restrict off-label use, limiting market expansion.
- Emerging global policies favor genericization, pressuring branded prices downward.
Key Challenges and Opportunities
Challenges
- Safety concerns restrict prescribing.
- Limited new formulations or indications.
- Competition from newer, safer AEDs.
Opportunities
- Development of safer, extended-release formulations.
- Expansion into emerging markets with high epilepsy prevalence.
- Potential approval for additional indications.
Key Takeaways
- The global epilepsy market remains grow-oriented but faces stiff competition.
- BANZEL holds a niche segment, primarily for refractory epilepsy and Lennox-Gastaut syndrome.
- Price stability is likely trending downward owing to generic competition.
- Future growth depends significantly on market penetration strategies, safety profile improvements, and regulatory approvals.
FAQs
1. How does BANZEL compare to newer AEDs in terms of price?
BANZEL generally costs less per tablet than newer AEDs like brivaracetam or perampanel but has lower market share due to safety concerns.
2. Are there plans to develop safer formulations of BANZEL?
No current FDA-approved formulations aim to improve safety; future development is uncertain but could enhance market viability.
3. What is the impact of generics on BANZEL pricing?
Generics are limited outside the U.S., leading to stable but potentially higher branded pricing domestically. Global markets see downward pressure due to generic availability.
4. How might regulatory changes influence BANZEL's market size?
Expansion of approved indications or safety profile improvements could increase demand, positively impacting price and sales.
5. What regions offer the most growth potential for BANZEL?
Emerging markets with high epilepsy prevalence and limited drug options present growth opportunities, contingent on regulatory access and affordability measures.
References
[1] Grand View Research. (2022). Epilepsy treatment market size, share & trends analysis report.
[2] Medispan. (2023). Wholesale Drug Pricing Data.
More… ↓
