Share This Page
Drug Price Trends for BANOPHEN ANTI-ITCH
✉ Email this page to a colleague
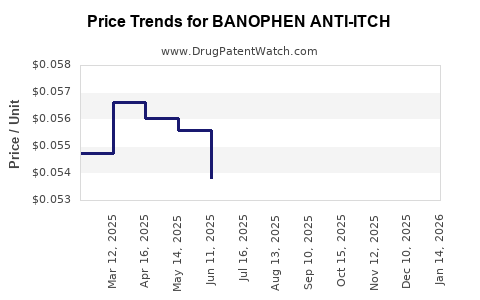
Average Pharmacy Cost for BANOPHEN ANTI-ITCH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BANOPHEN ANTI-ITCH 2% CREAM | 00904-5354-31 | 0.05810 | GM | 2026-05-20 |
| BANOPHEN ANTI-ITCH 2% CREAM | 00904-5354-31 | 0.05735 | GM | 2026-04-22 |
| BANOPHEN ANTI-ITCH 2% CREAM | 00904-5354-31 | 0.05669 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BANOPHEN ANTI-ITCH
What is the current market landscape for topical anti-itch medications?
BANOPHEN ANTI-ITCH is a topical medication indicated for relief from skin itching caused by insect bites, allergic reactions, or other minor skin irritations. The market for anti-itch products is segmented into OTC (over-the-counter) and prescription categories.
Market size and growth
- The global dermatological drugs market was valued at approximately $36 billion in 2021, with anti-itch products representing roughly 12% of this (Market Research Future, 2022).
- The OTC segment accounts for 65% of anti-itch medication sales, driven by consumer preference for self-medication.
- Compound annual growth rate (CAGR) from 2022 to 2027 is estimated at 4.5% for the anti-itch segment.
Competitive landscape
- Major competitors include hydrocortisone creams, diphenhydramine lotions, and topical calcineurin inhibitors.
- BANOPHEN ANTI-ITCH's unique market position hinges on its active ingredients, formulation, and safety profile.
What are the key factors influencing market demand?
- Growing awareness of skin health and minor ailments.
- Rising prevalence of allergic reactions and insect-bite incidents.
- Increased use of OTC remedies, especially in the wake of the COVID-19 pandemic.
- Consumer preference for rapid relief formulations with minimal side effects.
Regulatory environment
- Approval status varies globally; primarily approved as OTC in the U.S., with differential regulatory pathways elsewhere.
- Patent exclusivity remains a key driver; expiration of patents could introduce generic competition.
What are the price trends and projections?
Current pricing
| Region | Average OTC price per ounce | Notable competitors |
|---|---|---|
| United States | $8 - $12 | Hydrocortisone 1%, diphenhydramine |
| Europe | €6 - €10 | Similar formulations |
| Asia-Pacific | $4 - $8 | Local brands |
Price projections (2023-2027)
- Price stability is anticipated in North America, with a slight decrease (~3%) once generics enter.
- Emerging markets may see price increases up to 10% due to distribution expansion.
- Premium formulations or combination products could command prices 20% higher than basic formulations.
Influencing factors
- Patent protection expiration anticipated in 2024-2025 could lead to generic entry and price erosion.
- Regulatory changes or new formulation approvals could raise prices.
- Manufacturing costs, especially raw material prices, impact pricing strategies.
What are the potential market opportunities and risks?
Opportunities
- Expansion into emerging markets with high insect-bite incidence.
- Development of combination products integrating anti-itch with anti-inflammatory agents.
- Formulation improvements to enhance efficacy and safety profiles.
Risks
- Rapid generic competition post-patent expiry.
- Changes in regulatory policies restricting OTC availability.
- Market saturation with similar OTC products.
Summary table of projections
| Year | Market Size (USD billion) | Average Price (USD) per unit | Key Drivers |
|---|---|---|---|
| 2023 | 4.3 | $8.50 | Growing OTC demand |
| 2024 | 4.5 | $8.00 | Patent expiry, increased generics |
| 2025 | 4.8 | $7.75 | Entry of generics, market expansion |
| 2026 | 5.0 | $7.50 | Market saturation, new formulations |
| 2027 | 5.2 | $7.50 | Stabilized pricing, innovation |
Key Takeaways
- The anti-itch segment remains resilient, with steady growth driven by consumer self-care trends.
- BANOPHEN ANTI-ITCH's market share depends on post-patent strategies and competition management.
- Price stability is expected up to patent expiry; then, generics will pressure pricing.
- Opportunities exist for new formulations, entry into emerging markets, and combination products.
- Market risks primarily involve regulatory changes and intensified competition.
FAQs
Q1: When will BANOPHEN ANTI-ITCH face generic competition?
A1: Patent expiration is projected around 2024-2025, after which generic versions are likely to emerge.
Q2: How is the price of BANOPHEN ANTI-ITCH expected to change post-patent?
A2: Prices may decrease by 15-20% due to generic competition, but formulation or branding can mitigate declines.
Q3: What regions present the highest growth opportunities?
A3: Emerging markets in Asia-Pacific and Latin America offer significant growth potential due to rising skin-related health concerns.
Q4: How does BANOPHEN ANTI-ITCH compare to competitors?
A4: It may differentiate through formulation safety profiles, efficacy, brand recognition, and distribution channels.
Q5: What regulatory factors could impact market projections?
A5: Regulatory approval or restrictions affecting OTC status, formulation changes, or patent protections will influence pricing and market share.
References
[1] Market Research Future. (2022). Healthcare market reports.
[2] IBISWorld. (2022). Dermatological Drugs Industry Profile.
More… ↓
