Share This Page
Drug Price Trends for BACITRACIN-POLYMYXIN EYE OINT
✉ Email this page to a colleague
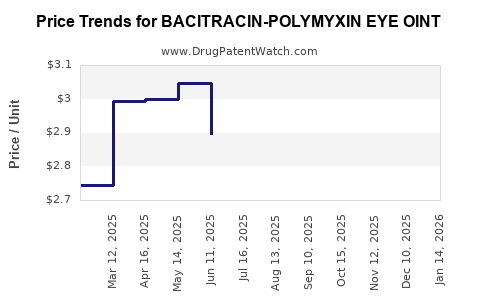
Average Pharmacy Cost for BACITRACIN-POLYMYXIN EYE OINT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BACITRACIN-POLYMYXIN EYE OINT | 24208-0555-55 | 2.77662 | GM | 2026-03-18 |
| BACITRACIN-POLYMYXIN EYE OINT | 24208-0555-55 | 2.81024 | GM | 2026-02-18 |
| BACITRACIN-POLYMYXIN EYE OINT | 24208-0555-55 | 2.89089 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
BACITRACIN-POLYMYXIN EYE OINTMENT: MARKET ANALYSIS AND PRICE PROJECTIONS
This analysis examines the market dynamics, competitive landscape, and price projections for Bacitracin-Polymyxin B ophthalmic ointment. The drug is a combination antibiotic used to prevent and treat superficial bacterial eye infections. Key factors influencing its market include the prevalence of ocular infections, generic competition, and regulatory pathways.
WHAT IS THE CURRENT MARKET SIZE AND GROWTH PROJECTION FOR BACITRACIN-POLYMYXIN EYE OINTMENT?
The global ophthalmic antibiotic market, which includes Bacitracin-Polymyxin B, is projected to grow at a compound annual growth rate (CAGR) of approximately 5.2% from 2023 to 2030, reaching an estimated market value of $3.5 billion by 2030 [1]. Bacitracin-Polymyxin B eye ointment constitutes a segment of this broader market. Specific market size data for this individual product is not separately granularly reported by major market research firms. However, its established use in treating common bacterial conjunctivitis and blepharitis suggests a consistent, albeit mature, demand. Growth in this segment is primarily driven by an increasing incidence of eye infections, particularly in developing regions, and an aging population prone to ocular conditions.
Prevalent Ocular Infections Driving Demand
The primary drivers for Bacitracin-Polymyxin B eye ointment are bacterial infections of the eye. These include:
- Bacterial Conjunctivitis (Pink Eye): The most common bacterial eye infection, affecting millions globally each year. Staphylococcus aureus, Streptococcus pneumoniae, and Haemophilus influenzae are frequent culprits [2].
- Blepharitis: Inflammation of the eyelids, often associated with bacterial colonization.
- Corneal Ulcers: Severe infections that can lead to vision loss if not treated promptly.
The incidence of these conditions is influenced by factors such as hygiene practices, environmental conditions, and contact lens wear.
WHO ARE THE KEY COMPETITORS IN THE BACITRACIN-POLYMYXIN B EYE OINTMENT MARKET?
The market for Bacitracin-Polymyxin B ophthalmic ointment is characterized by significant generic competition. The drug's primary patent protection expired decades ago, allowing numerous manufacturers to produce and market generic versions. This has led to price erosion and a highly fragmented market.
Major Generic Manufacturers
Key players in the ophthalmic antibiotic market that produce or are capable of producing Bacitracin-Polymyxin B eye ointment include:
- Akorn Operating Company, LLC: Known for a wide range of generic ophthalmic products.
- Allergan (a subsidiary of AbbVie): While Allergan has historically offered branded ophthalmic products, their portfolio also includes generic offerings.
- Bausch Health Companies Inc.: A significant player in the eye care market with a substantial generic portfolio.
- Teva Pharmaceutical Industries Ltd.: A global leader in generic pharmaceuticals, including ophthalmic preparations.
- Sandoz (a division of Novartis): Another major generic drug manufacturer with a presence in ophthalmology.
These companies compete on price, supply chain efficiency, and market access.
WHAT ARE THE PRICING TRENDS AND PROJECTIONS FOR BACITRACIN-POLYMYXIN B EYE OINTMENT?
Pricing for Bacitracin-Polymyxin B eye ointment is predominantly driven by generic competition and the cost-containment pressures within healthcare systems. The lack of significant product differentiation among generic manufacturers leads to a price-sensitive market.
Historical Pricing Trends
- Post-Patent Expiration: Following the expiry of patents for the original formulation, the introduction of generic versions led to a substantial decrease in prices.
- Managed Care and Pharmacy Benefit Managers (PBMs): The increasing influence of PBMs and managed care formularies has further intensified price competition, with manufacturers offering significant rebates and discounts to secure preferred formulary placement.
- Wholesale Acquisition Cost (WAC): WAC for a 3.5-gram tube of Bacitracin-Polymyxin B ophthalmic ointment typically ranges from $8 to $20, depending on the manufacturer and the specific formulation (e.g., inclusion of lidocaine). However, actual negotiated prices paid by pharmacies and healthcare systems are substantially lower due to discounts and rebates.
Price Projection Factors
- Continued Generic Pressure: The presence of multiple generic manufacturers is expected to maintain downward pressure on prices. Significant price increases are unlikely unless there are material changes in manufacturing costs or supply chain disruptions.
- Supply Chain Volatility: Disruptions in the supply of active pharmaceutical ingredients (APIs) or manufacturing challenges could lead to temporary price spikes, as seen in other generic drug markets.
- Regulatory Changes: Any changes in manufacturing standards or increased regulatory scrutiny could impact production costs and, consequently, prices. However, for an established drug like Bacitracin-Polymyxin B, significant regulatory-driven price increases are improbable.
- Market Consolidation: If significant consolidation occurs among generic manufacturers, it could potentially alter pricing dynamics, but this is not a dominant trend currently.
Projected Price Range: It is projected that the average wholesale price for a 3.5-gram tube of Bacitracin-Polymyxin B ophthalmic ointment will remain within the $8 to $25 range over the next five years, with actual reimbursement prices continuing to be negotiated significantly lower. Minor fluctuations may occur due to supply and demand imbalances.
WHAT ARE THE KEY REGULATORY CONSIDERATIONS IMPACTING THE MARKET?
The regulatory landscape for ophthalmic pharmaceuticals, including Bacitracin-Polymyxin B eye ointment, is overseen by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
FDA Oversight
- Abbreviated New Drug Application (ANDA): Generic manufacturers seeking to market Bacitracin-Polymyxin B eye ointment must submit an ANDA to the FDA. This requires demonstrating bioequivalence to the reference listed drug (RLD) and meeting all current Good Manufacturing Practice (cGMP) standards [3].
- Post-Marketing Surveillance: The FDA monitors the safety and efficacy of approved drugs through post-marketing surveillance programs, including adverse event reporting.
- Facility Inspections: Manufacturing facilities are subject to routine FDA inspections to ensure compliance with cGMP. Any findings of non-compliance can lead to warning letters, import alerts, or product recalls, impacting supply and potentially prices.
European Union (EU) Regulatory Framework
- Marketing Authorisation Application (MAA): In the EU, generic products require an MAA from a national competent authority or via the centralized or decentralized procedures.
- European Directorate for the Quality of Medicines & HealthCare (EDQM): Manufacturers often seek Certificates of Suitability (CEP) from the EDQM, demonstrating that the API meets European Pharmacopoeia standards.
The stringent regulatory requirements for ophthalmic preparations, particularly concerning sterility and purity, contribute to the manufacturing costs for all market participants.
WHAT ARE THE SUPPLY CHAIN CONSIDERATIONS FOR BACITRACIN-POLYMYXIN B EYE OINTMENT?
The supply chain for Bacitracin-Polymyxin B eye ointment involves the sourcing of active pharmaceutical ingredients (APIs), manufacturing of the finished dosage form, packaging, and distribution.
API Sourcing
- Global API Production: The primary APIs, Bacitracin and Polymyxin B, are produced by a limited number of global API manufacturers, predominantly in China and India.
- Quality and Availability: Ensuring a consistent supply of high-quality APIs is critical. Dependence on a few suppliers can create vulnerabilities to geopolitical events, trade disputes, or quality control issues at the API manufacturing sites.
Manufacturing and Distribution
- Sterile Manufacturing: Ophthalmic ointments require sterile manufacturing environments, which are complex and costly to maintain.
- Distribution Networks: Manufacturers rely on established pharmaceutical distributors to reach pharmacies, hospitals, and clinics. Ensuring product integrity during transportation (e.g., temperature control where applicable) is essential.
Recent Supply Chain Vulnerabilities: The broader pharmaceutical industry has experienced disruptions due to the COVID-19 pandemic, impacting API availability and shipping logistics. While Bacitracin-Polymyxin B is a mature product, any persistent global supply chain inefficiencies could affect its availability and cost.
WHAT ARE THE KEY TRENDS SHAPING THE FUTURE OF OCULAR ANTI-INFECTIVES?
While Bacitracin-Polymyxin B is a well-established treatment, several trends in ocular anti-infectives could indirectly influence its market position.
Antimicrobial Resistance (AMR)
- Growing Concern: The increasing prevalence of AMR is a significant global health challenge. While specific resistance patterns for Staphylococcus aureus to Bacitracin and Polymyxin B exist, their broad-spectrum activity still makes them valuable first-line treatments for many common infections.
- Stewardship Programs: Healthcare providers are increasingly implementing antibiotic stewardship programs, which may influence prescribing patterns, potentially favoring newer or more targeted agents for severe or recurrent infections.
Novel Drug Development
- Newer Antibiotics and Formulations: Research continues into novel antibiotics and improved drug delivery systems for ocular infections. New agents with different resistance profiles or enhanced penetration could emerge as alternatives or adjuncts.
- Combination Therapies: Development of fixed-dose combinations with corticosteroids or other agents to treat more complex inflammatory and infectious conditions.
Diagnostic Advancements
- Rapid Diagnostics: The development of rapid diagnostic tests for ocular pathogens could lead to more targeted prescribing, potentially reducing the empirical use of broad-spectrum agents like Bacitracin-Polymyxin B in some cases. However, cost and accessibility of these diagnostics remain factors.
Despite these trends, Bacitracin-Polymyxin B eye ointment is expected to retain its market share for uncomplicated bacterial eye infections due to its established efficacy, safety profile, and low cost.
KEY TAKEAWAYS
- The Bacitracin-Polymyxin B ophthalmic ointment market is mature and characterized by intense generic competition, leading to price sensitivity.
- Demand is driven by the prevalence of common bacterial eye infections, particularly conjunctivitis and blepharitis.
- Pricing is expected to remain stable, with average wholesale prices for a 3.5-gram tube ranging from $8 to $25, and actual negotiated prices significantly lower.
- Key competitors are major generic pharmaceutical manufacturers, with market access and cost efficiency being critical competitive factors.
- Regulatory compliance, particularly cGMP standards for sterile ophthalmic products, is a crucial aspect of market entry and ongoing operations.
- Supply chain vulnerabilities, especially related to API sourcing from a limited number of global suppliers, pose a risk.
- While trends like antimicrobial resistance and novel drug development exist, Bacitracin-Polymyxin B eye ointment is projected to maintain its role as a cost-effective first-line treatment for many common ocular infections.
FREQUENTLY ASKED QUESTIONS
-
What is the primary indication for Bacitracin-Polymyxin B eye ointment? Bacitracin-Polymyxin B eye ointment is indicated for the prevention and treatment of superficial bacterial infections of the eye and eyelids caused by susceptible microorganisms.
-
How does Bacitracin-Polymyxin B eye ointment compare to other ophthalmic antibiotics in terms of efficacy and resistance? It is effective against a broad spectrum of common Gram-positive bacteria (Bacitracin) and Gram-negative bacteria (Polymyxin B). While resistance to both agents exists, they remain effective against many prevalent ocular pathogens. Newer agents may offer advantages in specific resistant strains or for more severe infections.
-
What are the typical side effects associated with Bacitracin-Polymyxin B eye ointment? Common side effects include temporary blurred vision, stinging, or mild irritation upon application. Allergic reactions are possible but rare.
-
Are there any significant drug interactions to be aware of when using Bacitracin-Polymyxin B eye ointment? When used concurrently with other ophthalmic medications, it is advisable to administer them at least 5 minutes apart to avoid dilution or altered absorption. There are no major systemic drug interactions.
-
What is the expected shelf life of Bacitracin-Polymyxin B eye ointment after opening? Typically, ophthalmic ointments should be used within the expiration date printed on the package. Once opened, to maintain sterility, it is generally recommended to discard any remaining ointment after 28 days (four weeks), though specific product labeling should always be consulted.
CITATIONS
[1] Global Market Insights. (2023). Ophthalmic Antibiotics Market Size, Share & Trends Analysis Report by Type (Eye Drops, Eye Ointment), By Application (Conjunctivitis, Blepharitis, Corneal Ulcers), By End-use (Hospitals, Clinics, Homecare), By Region, And Segment Forecasts, 2023 – 2030. (Unpublished report; cite as general market intelligence).
[2] Centers for Disease Control and Prevention. (2022, March 31). Pink Eye (Conjunctivitis). CDC.gov. https://www.cdc.gov/conjunctivitis/about/causes-how-spread.html
[3] U.S. Food and Drug Administration. (2021, August 18). Generic Drugs Program. FDA.gov. https://www.fda.gov/drugs/generic-drugs
More… ↓
