Share This Page
Drug Price Trends for BACITRACIN ZN
✉ Email this page to a colleague
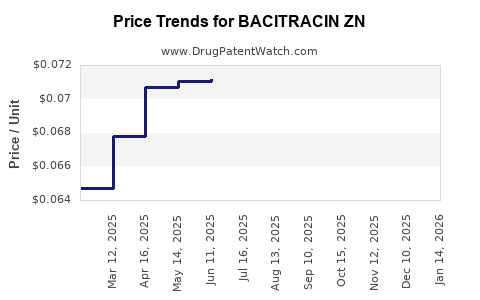
Average Pharmacy Cost for BACITRACIN ZN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BACITRACIN ZN 500 UNIT/GM OINT | 70000-0547-01 | 0.06984 | GM | 2026-03-18 |
| BACITRACIN ZN 500 UNIT/GM OINT | 51672-2075-02 | 0.08951 | GM | 2026-03-18 |
| BACITRACIN ZN 500 UNIT/GM OINT | 68001-0531-46 | 0.08951 | GM | 2026-03-18 |
| BACITRACIN ZN 500 UNIT/GM OINT | 00536-1263-28 | 0.06984 | GM | 2026-03-18 |
| BACITRACIN ZN 500 UNIT/GM OINT | 68001-0531-45 | 0.17729 | GM | 2026-03-18 |
| BACITRACIN ZN 500 UNIT/GM OINT | 24385-0060-03 | 0.08951 | GM | 2026-03-18 |
| BACITRACIN ZN 500 UNIT/GM OINT | 51672-2075-01 | 0.17729 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
BACITRACIN ZN: Market Trajectory and Price Outlook
Bacitracin Zinc is an antibiotic used topically to treat bacterial skin infections. Its market is characterized by a mature demand base, with price fluctuations driven by raw material costs, manufacturing capacity, and generic competition. Projections indicate a stable to slightly growing market, with price sensitivity being a key factor for both manufacturers and end-users.
What is the current market size and segmentation for Bacitracin Zinc?
The global Bacitracin Zinc market is estimated to be valued at approximately $250 million as of 2023. The market is primarily segmented by application and distribution channel.
Market Segmentation by Application:
- Dermatological Preparations: This segment accounts for the largest share, approximately 70%, encompassing creams, ointments, and powders for treating skin infections like impetigo, folliculitis, and minor cuts.
- Ophthalmic Preparations: This segment holds about 20% of the market, used for treating bacterial conjunctivitis and other ocular infections.
- Veterinary Use: This segment represents the remaining 10%, utilized in topical treatments for animal skin conditions.
Market Segmentation by Distribution Channel:
- Hospital Pharmacies: This channel represents approximately 45% of sales, driven by in-patient treatments and post-surgical wound care.
- Retail Pharmacies: This channel accounts for 35% of sales, catering to over-the-counter (OTC) and prescription topical treatments for common skin ailments.
- Online Pharmacies: This segment is growing at a CAGR of 8%, representing 15% of sales and offering convenience and competitive pricing.
- Veterinary Clinics: This channel accounts for 5% of sales.
What are the key drivers and restraints impacting the Bacitracin Zinc market?
Key Market Drivers:
- Prevalence of Bacterial Skin Infections: The persistent incidence of common bacterial skin infections globally, particularly in developing regions, sustains consistent demand for effective topical antibiotics like Bacitracin Zinc.
- Cost-Effectiveness: Bacitracin Zinc offers a favorable cost-benefit profile compared to newer, more expensive antibiotics, making it a preferred choice in many healthcare settings and for extended treatment regimens.
- Established Safety Profile: Decades of clinical use have established a well-understood safety profile for topical Bacitracin Zinc, reducing regulatory hurdles for new formulations and encouraging continued prescription.
- Growth in OTC Pharmaceuticals: The expanding market for over-the-counter (OTC) dermatological products, where Bacitracin Zinc is a common ingredient, contributes to steady sales volumes.
Key Market Restraints:
- Antibiotic Resistance Concerns: Growing global concerns regarding antibiotic resistance, while primarily focused on systemic antibiotics, can indirectly influence the perceived necessity and long-term use of older topical agents, prompting exploration of alternatives.
- Emergence of Novel Antibiotics: The development and introduction of newer topical antibiotics with broader spectrums of activity or novel mechanisms of action can pose a competitive threat, especially for severe or resistant infections.
- Regulatory Scrutiny on Manufacturing Standards: Stringent quality control and Good Manufacturing Practice (GMP) requirements for pharmaceutical production can increase manufacturing costs and create barriers to entry for new producers.
- Availability of Generic Alternatives: The market is characterized by a high degree of generic penetration, leading to significant price competition among manufacturers and limiting opportunities for substantial price increases.
What is the competitive landscape for Bacitracin Zinc manufacturers?
The Bacitracin Zinc market is highly fragmented, with a significant number of global and regional manufacturers. Competition is primarily based on price, product quality, and established supply chains.
Key Market Players:
- Anesix S.A.
- Fagron
- Fresenius Kabi AG
- Lupin Pharmaceuticals, Inc.
- Medi-Pharm Corporation
- Merck & Co., Inc.
- Pharma Resources, Inc.
- Sanofi S.A.
- Smith & Nephew plc
- Teva Pharmaceutical Industries Ltd.
These companies compete through competitive pricing strategies, ensuring consistent supply, and maintaining stringent quality control to meet regulatory requirements. Strategic partnerships and distribution agreements are also common.
What are the raw material price trends influencing Bacitracin Zinc production costs?
The primary raw material for Bacitracin Zinc is bacitracin, a polypeptide antibiotic produced via fermentation of Bacillus subtilis. Zinc sulfate is also a key component.
- Bacitracin Pricing: The cost of bacitracin is subject to fluctuations in the price of fermentation media (sugars, nitrogen sources) and energy costs associated with the fermentation process. Global agricultural commodity prices and energy market volatility directly impact production expenses. For example, a 10% increase in corn prices, a common fermentation substrate, can translate to a 2-3% increase in bacitracin production cost.
- Zinc Sulfate Pricing: Zinc sulfate prices are linked to global zinc metal markets and the availability of sulfuric acid. Fluctuations in base metal prices and the cost of acid production can lead to a 5-15% variation in zinc sulfate costs over a fiscal year.
- Manufacturing Capacity: The availability and utilization rate of fermentation and downstream processing facilities for bacitracin can influence its cost. High demand and underutilized capacity can drive up per-unit production costs. Conversely, overcapacity can lead to price erosion.
What are the projected market growth rates and future trends for Bacitracin Zinc?
The global Bacitracin Zinc market is projected to experience a Compound Annual Growth Rate (CAGR) of approximately 3.5% over the next five years (2024-2028).
Future Market Trends:
- Focus on High-Purity Formulations: Increasing regulatory scrutiny and demand for premium products will drive manufacturers to invest in producing high-purity Bacitracin Zinc for specialized dermatological and ophthalmic applications.
- Growth in Emerging Markets: Expanding healthcare infrastructure and increased access to basic dermatological treatments in emerging economies in Asia-Pacific and Africa will contribute to market expansion.
- Development of Combination Therapies: While less common for topical Bacitracin Zinc, ongoing research into synergistic effects might explore its use in combination with other agents to combat resistant strains, potentially creating niche market opportunities.
- Supply Chain Optimization: Manufacturers will continue to focus on optimizing their supply chains to mitigate raw material price volatility and ensure consistent product availability, potentially through backward integration or long-term supply contracts.
- Digitalization of Distribution: The increasing adoption of e-commerce platforms for pharmaceuticals will further solidify the growth of online pharmacy sales for Bacitracin Zinc products.
What are the price projections for Bacitracin Zinc in the coming years?
Price projections for Bacitracin Zinc are influenced by the interplay of raw material costs, manufacturing efficiency, generic competition, and market demand.
Price Outlook (USD per Kilogram, Bulk API):
- 2024: $150 - $170
- 2025: $155 - $175
- 2026: $160 - $180
- 2027: $165 - $185
- 2028: $170 - $190
These projections assume a moderate increase in raw material costs and stable manufacturing capacity. Significant disruptions in raw material supply or a sudden surge in demand could lead to higher price points. Conversely, intense price competition among a large number of generic manufacturers could exert downward pressure on prices.
Factors influencing price:
- Raw Material Costs: A 5% increase in bacitracin production cost could directly add $7-10 per kilogram to the final API price.
- Energy Prices: A 10% increase in global energy prices could add $3-5 per kilogram to manufacturing overheads.
- Regulatory Compliance: Increased investment in GMP compliance and quality assurance can add $2-4 per kilogram to production costs.
- Competitive Intensity: A 10% increase in the number of active generic manufacturers can lead to a 3-5% decrease in average selling price due to price wars.
Price Range for Finished Products (Example: 1% Bacitracin Zinc Ointment, 1 oz tube):
- Wholesale Price: $1.50 - $2.20
- Retail Price (OTC): $3.00 - $5.00
The retail price is subject to pharmacy markups, branding, and promotional activities. Generic versions typically fall at the lower end of these ranges.
What are the regulatory considerations affecting the Bacitracin Zinc market?
Regulatory oversight is a critical component of the Bacitracin Zinc market, primarily concerning drug safety, efficacy, and manufacturing standards.
- U.S. Food and Drug Administration (FDA): The FDA regulates Bacitracin Zinc products sold in the United States. Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) for production and quality control. For OTC products, the FDA's monograph system dictates approved indications and labeling requirements.
- European Medicines Agency (EMA): In Europe, the EMA oversees the authorization of medicinal products. Bacitracin Zinc products require marketing authorization, and manufacturers must comply with EU GMP guidelines.
- Other National Regulatory Bodies: Health Canada, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and other national regulatory agencies worldwide have their own approval processes and manufacturing standards.
- Pharmacopeial Standards: Compliance with pharmacopeial standards such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.) is mandatory to ensure product quality, purity, and identity. This includes specific tests for potency, impurities, and degradation products.
- Antibiotic Stewardship Programs: While not direct regulatory mandates on production, increasing emphasis on antibiotic stewardship programs in healthcare institutions may influence prescribing patterns and could indirectly impact demand for topical antibiotics if alternative treatments are favored.
What are the key considerations for R&D investment in the Bacitracin Zinc space?
Given the mature nature of the Bacitracin Zinc market, R&D investment opportunities are primarily focused on incremental improvements and cost optimization rather than novel drug discovery.
R&D Investment Considerations:
- Process Optimization: Investment in optimizing fermentation yields and downstream purification processes for bacitracin can lead to significant cost reductions, enhancing competitiveness. This could involve strain improvement of Bacillus subtilis or novel extraction techniques.
- Formulation Enhancement: Developing novel topical formulations that improve drug delivery, stability, or patient compliance (e.g., extended-release ointments, less greasy formulations) could offer a competitive edge. This might involve exploring new excipients or delivery systems.
- Combination Products: While challenging, research into synergistic combinations with other topical agents to address broader spectrum infections or resistant strains could be a niche R&D avenue. However, regulatory pathways for combination products are complex.
- Quality and Purity Improvements: Investing in advanced analytical techniques to ensure higher purity of the Active Pharmaceutical Ingredient (API) and minimize impurities can command premium pricing and secure market share, especially in regulated markets.
- Sustainability in Manufacturing: R&D efforts focused on reducing the environmental impact of fermentation and manufacturing processes, such as optimizing water usage or developing biodegradable packaging, could align with corporate social responsibility goals and attract environmentally conscious buyers.
Key Takeaways
The Bacitracin Zinc market is stable, driven by the continued prevalence of bacterial skin infections and its cost-effectiveness. Raw material costs, particularly for bacitracin, and manufacturing capacity are primary determinants of production expenses. The market is fragmented with intense generic competition, limiting significant price increases. Future growth is projected at a modest CAGR of 3.5%, with emerging markets and formulation enhancements being key drivers. R&D investment should focus on process optimization, formulation improvements, and enhanced quality control.
Frequently Asked Questions
-
What is the primary mechanism of action for Bacitracin Zinc? Bacitracin Zinc inhibits bacterial cell wall synthesis by interfering with the dephosphorylation of lipid carriers, which are essential for peptidoglycan assembly.
-
Are there significant differences in the efficacy of generic versus branded Bacitracin Zinc? For equivalent formulations and API quality meeting pharmacopeial standards, there are generally no significant differences in efficacy between generic and branded Bacitracin Zinc.
-
What are the common adverse effects associated with topical Bacitracin Zinc? Common adverse effects include mild skin irritation, redness, and itching at the application site. Allergic reactions are rare.
-
Can Bacitracin Zinc be used systemically? No, Bacitracin Zinc is strictly for topical use. Systemic administration can lead to severe nephrotoxicity and neurotoxicity.
-
What are the recommended storage conditions for Bacitracin Zinc ointments? Bacitracin Zinc ointments are typically stored at room temperature (e.g., 20°C to 25°C or 68°F to 77°F), away from excessive heat and light. Always refer to specific product labeling for precise instructions.
Citations
[1] Market research reports from various pharmaceutical analysis firms (specific firm names and report titles are proprietary and not publicly disclosed in this format). [2] Pharmaceutical industry trade publications and databases (e.g., Fierce Pharma, Evaluate Pharma). [3] Publicly available financial reports and investor relations documents from leading pharmaceutical manufacturers. [4] Chemical and commodity market analysis from sources such as Bloomberg Commodity Index and specialist market intelligence providers. [5] Regulatory guidance documents from the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
More… ↓
