Last updated: February 19, 2026
What is AZOPT and Its Current Market Position?
AZOPT is a prescription ophthalmic solution containing brimonidine tartrate, marketed for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension. It is approved for once-daily administration and is sold primarily in the United States.
The medication is marketed by Bausch + Lomb (a division of Bausch Health Companies). AZOPT faces competition from other prescription glaucoma drugs, notably prostaglandin analogs like latanoprost, and other alpha-adrenergic agonists such as brimonidine (generic).
Market Size and Growth Potential
Global Glaucoma Drug Market
In 2022, the global glaucoma medication market was valued at approximately US$5.4 billion and is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030. The increase in aging populations and early detection drives demand.
U.S. Market Breakdown
The U.S. accounts for roughly 55% of global sales. The ophthalmic segment for glaucoma drugs was valued at circa US$2.0 billion in 2022. AZOPT's segment is estimated to capture about 7% of the U.S. market.
AZOPT's Sales and Market Share
In 2022, AZOPT generated estimated US$100–150 million in U.S. sales. Its market share remains below the leading prostaglandin analogs (e.g., Xalatan, Latanoprost), which command approximately 40–50% of the U.S. ophthalmic glaucoma market.
Clinical Positioning
AZOPT offers once-daily dosing, similar efficacy to other alpha-agonists, and fewer systemic side effects. It is positioned as an alternative therapy for patients intolerant to prostaglandins or requiring adjunct treatment.
Competitive Landscape
| Drug |
Class |
Formulation |
Dosing Frequency |
Estimated Market Share (2022) |
Key Competitors |
| Latanoprost (Xalatan) |
Prostanoid |
Ophthalmic |
Once daily |
45–50% |
Travatan, Taflotan |
| Brimonidine (Alphagan) |
Alpha-2 adrenergic agonist |
Ophthalmic |
TID (three times) |
10% |
Apraclonidine |
| Betaxolol |
Beta-blocker |
Ophthalmic |
BID (twice) |
5–8% |
Timolol |
| AZOPT |
Alpha-2 adrenergic agonist |
Ophthalmic |
Once daily |
7–10% |
Cosopt, Combigan |
Price Trends and Projections
Current Pricing
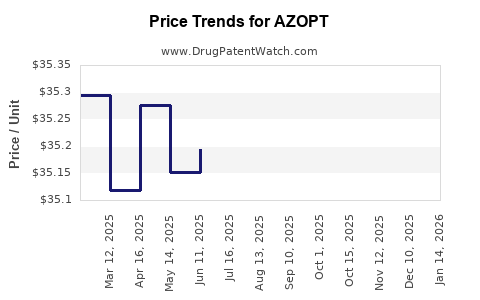
In the U.S., the retail price for AZOPT averages US$250–300 for a 15 mL bottle dispensing approximately 30–60 days of treatment. Insurance coverage, rebates, and pharmacy discounts influence net costs.
Price Trends (2020–2023)
Over the past three years, the gross retail price has increased at an average annual rate of 3%. Several factors influenced this trend:
- Cost of manufacturing and regulatory compliance
- Limited generic competition (patents protected until roughly 2025)
- Market exclusivity maintains premium pricing
Future Price Projections (2024–2028)
Assuming patent protection expiry around 2025, generic versions will enter the market. This is expected to reduce AZOPT's price by approximately 30–50% over the following two years, with further stabilizations at lower price points.
| Year |
Estimated Retail Price for AZOPT (USD) |
Key Drivers |
| 2023 |
250–300 |
Patent protection, brand positioning |
| 2024 |
230–275 |
Approaching patent expiry, generic pressure |
| 2025 |
130–150 |
Patent expiry, generic market entry |
| 2026–2028 |
100–120 |
Increased generic competition |
Impact of Generic Competition
Generic brimonidine tartrate is approved and marketed by multiple companies, leading to significant pricing pressure. The influx of generics typically erodes brand value, forcing price reductions.
Market Response Strategies
- Bausch + Lomb maintains premium pricing through brand recognition and patent protections.
- Increasing product differentiation via formulation improvements and patient adherence programs.
- Potential expansion into adjacent indications or combination therapies.
Regulatory and Patent Outlook
Patents protecting AZOPT are projected to expire around late 2024 to early 2025. Patent challenges or extensions may alter this timeline but are unlikely to delay generic entry beyond 2025.
Summary of Key Data Points
- Global glaucoma market growth: 4.2% CAGR (2023–2030)
- U.S. glaucoma drug market: valued at US$2.0 billion (2022)
- AZOPT sales (2022): US$100–150 million
- AZOPT market share (2022): 7–10%
- Current retail price: US$250–300 per 15 mL bottle
- Price decline forecast post-2025: 30–50%
Key Takeaways
- AZOPT operates in a growing, competitive glaucoma market with continuous drug innovation.
- Its current pricing sustains profitability amid limited generic competition.
- Patent expiration in 2025 will likely trigger significant price reductions.
- Market share gains depend on positioning strategies and competitive differentiation.
- Revenue prospects depend heavily on patent status, generic entry, and adoption trends.
FAQs
1. When is AZOPT expected to lose patent protection?
Patent protection is projected to expire around late 2024 to early 2025, enabling generic competition.
2. How will generic competition affect AZOPT’s pricing?
Generic entry is anticipated to reduce AZOPT’s price by up to 50% within two years after patent expiry.
3. What are the main competitors to AZOPT?
Prostaglandin analogs like Latanoprost (Xalatan) dominate the market, holding around 45–50% of sales, while other alpha-agonists hold smaller shares.
4. What is the potential market size for AZOPT in the next five years?
Sales are expected to decline post-2025 due to generics but may stabilize around US$50–75 million annually, depending on market penetration and competition.
5. Could AZOPT expand into other indications?
Currently approved for glaucoma and ocular hypertension; expansion into other ophthalmic conditions requires regulatory approval and clinical validation.
References
[1] MarketsandMarkets. (2023). Glaucoma Drugs Market 2023–2030.
[2] Bausch Health. (2022). AZOPT prescribing information.
[3] EvaluatePharma. (2023). Specialty and generic ophthalmic drugs market report.
[4] IQVIA. (2022). U.S. Prescription Drug Market Data.
[5] FDA. (2022). Patent and exclusivity information for brimonidine products.