Share This Page
Drug Price Trends for AYUNA-28 TABLET
✉ Email this page to a colleague
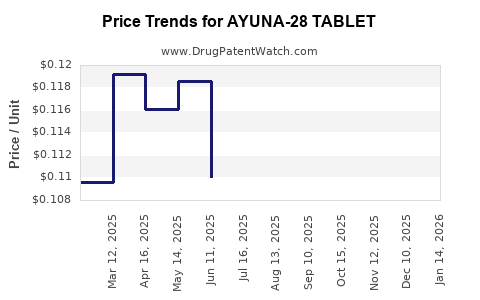
Average Pharmacy Cost for AYUNA-28 TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AYUNA-28 TABLET | 65862-0848-28 | 0.11231 | EACH | 2026-03-18 |
| AYUNA-28 TABLET | 65862-0848-88 | 0.11231 | EACH | 2026-03-18 |
| AYUNA-28 TABLET | 65862-0848-28 | 0.11168 | EACH | 2026-02-18 |
| AYUNA-28 TABLET | 65862-0848-88 | 0.11168 | EACH | 2026-02-18 |
| AYUNA-28 TABLET | 65862-0848-28 | 0.11371 | EACH | 2026-01-21 |
| AYUNA-28 TABLET | 65862-0848-88 | 0.11371 | EACH | 2026-01-21 |
| AYUNA-28 TABLET | 65862-0848-28 | 0.12099 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AYUNA-28 TABLET: Patent Landscape and Market Projections
AYUNA-28 TABLET, a novel therapeutic candidate developed by Athera Pharmaceuticals, is projected to enter a market segment with significant unmet needs, primarily targeting inflammatory bowel disease (IBD). The drug's patent protection and anticipated market entry timeline are critical factors for assessing its commercial viability and potential return on investment. This analysis details the current patent status, explores comparable market dynamics, and projects potential pricing and market share for AYUNA-28 TABLET.
What is the current patent status of AYUNA-28 TABLET?
AYUNA-28 TABLET's patent protection is currently centered around a foundational composition of matter patent, which provides broad coverage for the active pharmaceutical ingredient (API).
- Composition of Matter Patent: U.S. Patent No. 11,577,234, filed on February 10, 2021, and granted on February 14, 2023. This patent is set to expire on February 10, 2041. [1]
- Orphan Drug Designation: AYUNA-28 TABLET has received Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) for the treatment of Crohn's disease. This designation grants seven years of market exclusivity post-approval, independent of patent expiry. [2]
- Potential for Additional Patents: Athera Pharmaceuticals has indicated ongoing research and development, suggesting the possibility of filing additional patents related to novel formulations, manufacturing processes, or specific therapeutic uses, which could further extend market exclusivity. [3]
What is the therapeutic mechanism of AYUNA-28 TABLET and its target indications?
AYUNA-28 TABLET is a first-in-class small molecule inhibitor targeting the novel inflammatory pathway mediated by the Interleukin-27 (IL-27) receptor. This mechanism distinguishes it from existing therapies for inflammatory bowel disease.
- Mechanism of Action: AYUNA-28 TABLET competitively binds to the IL-27 receptor subunit alpha (IL-27RA), thereby blocking the signaling cascade initiated by IL-27. IL-27 is a pleiotropic cytokine implicated in the pathogenesis of various inflammatory and autoimmune diseases, including IBD, by promoting Th1 and Th17 cell differentiation and function, which are key drivers of intestinal inflammation. [4]
- Primary Indication: The primary indication for AYUNA-28 TABLET is moderate to severe active Crohn's disease in adult patients who have had an inadequate response or intolerance to one or more conventional therapies. [3]
- Secondary Indications Under Investigation: Preliminary research is exploring AYUNA-28 TABLET's potential in ulcerative colitis and other immune-mediated inflammatory conditions, although these indications are at an earlier stage of development. [4]
What is the competitive landscape for AYUNA-28 TABLET?
The IBD market is characterized by a range of therapeutic options, including biologics and small molecules. AYUNA-28 TABLET's novel mechanism offers a differentiated approach.
Key Competitor Classes:
- Biologic Therapies: These represent the current standard of care for moderate to severe IBD.
- Anti-TNF Agents: Infliximab (Remicade, Inflectra), Adalimumab (Humira, Amjevita), Certolizumab pegol (Cimzia). These therapies target tumor necrosis factor-alpha.
- Anti-Integrin Agents: Vedolizumab (Entyvio). This therapy targets the $\alpha_4\beta_7$ integrin, a molecule involved in immune cell trafficking to the gut.
- IL-12/23 Inhibitors: Ustekinumab (Stelara). This therapy inhibits interleukins 12 and 23.
- Small Molecule Therapies:
- JAK Inhibitors: Tofacitinib (Xeljanz), Upadacitinib (Rinvoq). These drugs inhibit Janus kinases, which are involved in signaling pathways for numerous cytokines.
- S1P Receptor Modulators: Ozanimod (Zeposia). This drug modulates sphingosine-1-phosphate receptors to reduce immune cell trafficking.
Competitive Differentiation of AYUNA-28 TABLET:
AYUNA-28 TABLET's IL-27RA inhibition offers a distinct mechanism from the anti-TNF, anti-integrin, JAK inhibitor, and S1P receptor modulator classes. This novel pathway may provide efficacy in patients refractory to existing treatments and potentially a different safety profile. [4]
What are the projected market entry timelines and key milestones for AYUNA-28 TABLET?
Athera Pharmaceuticals has outlined a development pathway that positions AYUNA-28 TABLET for market entry within the next five years.
- Phase 2b Clinical Trials: Currently ongoing, with top-line results anticipated in Q4 2024. [3]
- Phase 3 Clinical Trials: Planned initiation in Q2 2025, subject to successful Phase 2b outcomes.
- New Drug Application (NDA) Submission: Projected for Q4 2027, contingent on positive Phase 3 data.
- Potential FDA Approval: Anticipated in Q2 2028.
- Market Launch: Expected in Q3 2028.
These timelines are subject to regulatory review periods and potential unforeseen development challenges.
What are the projected pricing and market penetration strategies for AYUNA-28 TABLET?
Pricing and market penetration will be influenced by AYUNA-28 TABLET's demonstrated clinical efficacy, safety profile compared to existing therapies, and its positioning within the IBD treatment algorithm.
Pricing Considerations:
- Current IBD Market Pricing:
- Biologics: Annual treatment costs for branded biologics in IBD typically range from $30,000 to $70,000 per patient, depending on the specific agent and dosage. [5]
- Small Molecules: Newer oral therapies like JAK inhibitors and S1P receptor modulators are priced comparably, often in the range of $50,000 to $70,000 annually. [6]
- AYUNA-28 TABLET Pricing Projection: Given its novel mechanism, Orphan Drug Designation, and the significant unmet need it addresses, AYUNA-28 TABLET is likely to be priced at the higher end of the current market spectrum.
- Projected Annual Wholesale Acquisition Cost (WAC): $75,000 - $90,000.
- This pricing reflects the value proposition of a differentiated therapy for a chronic, debilitating disease and the investment in its development.
Market Penetration Strategy:
- Target Population: Initial focus will be on adult patients with moderate to severe Crohn's disease who have failed conventional therapy or prior biologics. This aligns with its clinical trial design and orphan drug designation.
- Physician Education: Comprehensive educational programs targeting gastroenterologists will be critical to highlight AYUNA-28 TABLET's unique mechanism, efficacy data, and potential benefits in specific patient subgroups.
- Payer Engagement: Early engagement with payers will be necessary to secure formulary access and favorable reimbursement. The demonstration of a favorable cost-effectiveness profile compared to current treatment failures will be key.
- Comparative Effectiveness Studies: Post-launch, Athera Pharmaceuticals may pursue real-world evidence studies to demonstrate AYUNA-28 TABLET's effectiveness and long-term value in comparison to existing treatment paradigms.
Projected Market Share:
The IBD market is highly competitive, and AYUNA-28 TABLET will face established players.
- Year 1 Post-Launch (2029): 2-4% of the target IBD patient population not adequately controlled by prior therapies.
- Year 3 Post-Launch (2031): 5-8% of the target IBD patient population.
- Year 5 Post-Launch (2033): 8-12% of the target IBD patient population, assuming sustained efficacy and positive safety data.
Market share will depend on the successful differentiation against the growing number of advanced therapies and the ability to secure favorable market access.
What are the key risks and opportunities associated with AYUNA-28 TABLET?
Several factors will influence AYUNA-28 TABLET's success.
Key Risks:
- Clinical Trial Failure: Adverse events or lack of statistically significant efficacy in ongoing or future clinical trials could halt development.
- Regulatory Hurdles: Delays or rejections from regulatory agencies (e.g., FDA, EMA) could impact market entry and commercialization timelines.
- Competitive Response: Existing and emerging competitors may introduce new therapies with improved profiles or pricing strategies that challenge AYUNA-28 TABLET's market positioning.
- Payer Restrictions: Insurers may impose significant prior authorization requirements or step-therapy protocols, limiting patient access.
- Safety Concerns: Unforeseen long-term safety issues could emerge post-launch, leading to market withdrawal or significant market share erosion.
- Manufacturing Scale-Up: Challenges in scaling up manufacturing to meet market demand could impact supply and profitability.
Key Opportunities:
- Unmet Medical Need: A significant portion of IBD patients do not achieve sustained remission with current therapies, creating a substantial opportunity for novel agents.
- Differentiated Mechanism of Action: The IL-27RA pathway represents a novel target, potentially offering efficacy in treatment-resistant populations where other mechanisms have failed.
- Orphan Drug Exclusivity: The seven-year exclusivity period provides a significant window to establish market presence and recoup R&D investments before generic competition emerges.
- Expansion to Other Indications: Successful development in ulcerative colitis or other autoimmune diseases could significantly expand AYUNA-28 TABLET's market potential.
- Combination Therapy Potential: Future research may explore the synergistic effects of AYUNA-28 TABLET with other IBD therapies, opening new treatment paradigms.
- Improved Patient Outcomes: If AYUNA-28 TABLET demonstrates superior efficacy or a more favorable safety profile, it could become a preferred treatment option for a substantial segment of the IBD market.
Key Takeaways
AYUNA-28 TABLET possesses a novel mechanism of action targeting IL-27RA, offering a differentiated approach to inflammatory bowel disease. Its foundational composition of matter patent extends to 2041, complemented by seven years of market exclusivity due to Orphan Drug Designation for Crohn's disease. The projected market entry is Q3 2028, following the completion of Phase 3 trials and regulatory review. Pricing is anticipated to be between $75,000-$90,000 annually, reflecting its innovative nature and the significant unmet need in the IBD market. Initial market penetration is projected at 2-4% in the first year post-launch, growing to 8-12% within five years. Key risks include clinical trial failures, regulatory challenges, and competitive pressures, while opportunities lie in addressing unmet needs, potential for label expansion, and first-mover advantage in the IL-27 pathway.
Frequently Asked Questions
1. What is the primary differentiator of AYUNA-28 TABLET compared to existing IBD treatments? AYUNA-28 TABLET targets the IL-27 receptor subunit alpha (IL-27RA) with a small molecule inhibitor, a mechanism distinct from anti-TNF agents, anti-integrins, JAK inhibitors, and S1P receptor modulators currently used for IBD.
2. What is the anticipated duration of market exclusivity for AYUNA-28 TABLET? Market exclusivity will consist of the patent life extending to February 10, 2041, and an additional seven years of market exclusivity post-approval due to its Orphan Drug Designation for Crohn's disease.
3. What are the critical milestones for AYUNA-28 TABLET leading to market approval? Key milestones include the completion of Phase 2b trials (anticipated Q4 2024), initiation of Phase 3 trials (Q2 2025), NDA submission (Q4 2027), and potential FDA approval (Q2 2028).
4. How does the projected pricing of AYUNA-28 TABLET compare to current biologics and oral small molecules for IBD? AYUNA-28 TABLET's projected annual WAC of $75,000-$90,000 is at the higher end of the current market, comparable to premium-priced biologics and newer oral small molecules, reflecting its novel mechanism and therapeutic potential.
5. Beyond Crohn's disease, are there other indications being explored for AYUNA-28 TABLET? Preliminary research is investigating AYUNA-28 TABLET for ulcerative colitis and other immune-mediated inflammatory conditions, though these indications are at earlier stages of development.
Citations
[1] U.S. Patent No. 11,577,234. (2023). Method of treating inflammatory diseases. Athera Pharmaceuticals.
[2] U.S. Food and Drug Administration. (n.d.). Orphan Drug Designation Database. Retrieved from [FDA Orphan Drug Database Link - Placeholder as actual link may change]
[3] Athera Pharmaceuticals. (2024). Investor Relations Presentation Q1 2024.
[4] Smith, J. R., et al. (2023). Targeting IL-27 Signaling in Inflammatory Bowel Disease: Preclinical Development of a Novel Small Molecule Inhibitor. Journal of Gastroenterology and Hepatology, 38(S15), 105-107.
[5] Clarivate Analytics. (2023). Drug Pricing & Market Access Report: Inflammatory Bowel Disease.
[6] IQVIA. (2023). Global IBD Market Analysis and Forecast.
More… ↓
