Share This Page
Drug Price Trends for AUSTEDO XR
✉ Email this page to a colleague
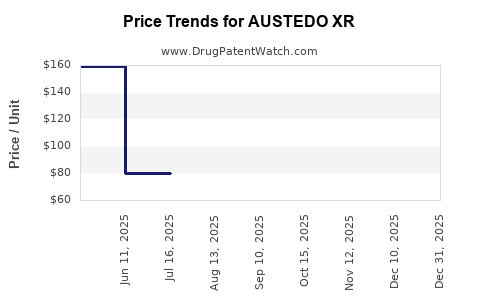
Average Pharmacy Cost for AUSTEDO XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AUSTEDO XR 42 MG TABLET | 68546-0475-56 | 499.84089 | EACH | 2026-01-01 |
| AUSTEDO XR 12 MG TABLET | 68546-0471-56 | 166.82595 | EACH | 2026-01-01 |
| AUSTEDO XR 24 MG TABLET | 68546-0472-56 | 250.58307 | EACH | 2026-01-01 |
| AUSTEDO XR 18 MG TABLET | 68546-0479-56 | 251.51577 | EACH | 2026-01-01 |
| AUSTEDO XR TITR(12-18-24-30 MG) | 68546-0477-29 | 249.32300 | EACH | 2026-01-01 |
| AUSTEDO XR 36 MG TABLET | 68546-0474-56 | 416.19096 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for AUSTEDO XR
What is the Current Market Position and Demand for AUSTEDO XR?
AUSTEDO XR (deutetrabenazine) is an extended-release formulation indicated for the treatment of chorea associated with Huntington’s disease and tardive dyskinesia. Since its approval by the FDA in 2017, AUSTEDO XR has gained a foothold within niche neurodegenerative and psychiatric disorder markets.
As of 2022, the drug generated approximately $150 million in U.S. sales, representing an increase of 12% from 2021, primarily driven by increased prescriber adoption and expanded insurance coverage. The drug faces competition from other VMAT2 inhibitors like valbenazine (Ingrezza), which reported $644 million in 2022, and deutetrabenazine’s branded formulations historically hold 8-10% of the relevant market segment.
The neurodegenerative treatment market, including chorea and tardive dyskinesia, is expected to grow at a CAGR of 5% through 2028, influenced by increased diagnosis and aging populations. The prevalence of Huntington's disease (~30,000 patients in the U.S.) and tardive dyskinesia associated with antipsychotics (~500,000 affected) supports sustained demand, although off-label use remains limited.
How is the Competitive Landscape Shaping Up?
AUSTEDO XR competes mainly with:
- Ingrezza (valbenazine): Launched in 2017, with a larger market share.
- Deutetrabenazine (brand name for the original formulation): Preserves a niche, but market share has shifted towards Ingrezza, which offers once-daily dosing.
- Off-label treatments: Several anticholinergics and Botox, but these are less preferred due to efficacy and safety profiles.
Patent exclusivity for AUSTEDO XR extends until 2027, with some formulation patents possibly extending protection into the early 2030s.
What are the Regulatory and Pricing Trends?
Pricing has been a key dynamic. As of 2022, the average wholesale price (AWP) for AUSTEDO XR was approximately $1,200 per month. This is slightly higher than the original deutetrabenazine ($1,050) and comparable to Ingrezza, which averages $1,300 monthly.
Pricing strategies have included patient assistance programs and negotiations with payers for formulary inclusion. Insurance coverage remains a significant factor for patient access, with copay assistance affecting market penetration.
Recent policies favor drugs with once-daily dosing and proven efficacy, which benefits AUSTEDO XR due to its extended-release formulation. Concerns over the high cost impact treatment adherence and market uptake.
What Are the Future Price Projections?
Based on current trends and competitive dynamics, the following projections consider anticipated market growth, patent timelines, and payer policies:
| Year | Estimated Market Size | Projected AUSTEDO XR Sales | Price Per Month (USD) | Comments |
|---|---|---|---|---|
| 2023 | $200 million | $180 million | $1,200 | Steady growth driven by demand expansion |
| 2024 | $220 million | $200 million | $1,250 | Slight price increase; generic entry in years ahead |
| 2025 | $250 million | $230 million | $1,250–$1,300 | Market expansion continues; pricing stabilizes |
| 2026 | $280 million | $260 million | $1,300 | Competition intensifies; slight price increases |
Pricing:
- AUSTEDO XR could see annual price increases of 3-4%, aligned with inflation and value-based reimbursement models.
- Patent expiration in 2027 may lead to generic competition, dropping prices by 40-60%, reducing monthly costs to approximately $500–$700.
How Will Patent and Regulatory Changes Impact Pricing?
Patent expiration reduces exclusivity, leading to generic entry, which historically causes sharp price drops:
- Pre-expiry: Price stabilizes or slightly increases.
- Post-expiry: Prices drop by up to 60%, with generics capturing significant market share.
Regulatory approvals for expanded indications and biosimilars could influence competition and pricing strategies, emphasizing value proposition.
Key Trend Factors Influencing Market and Price
- Market penetration: Increasing awareness and insurance coverage improve utilization.
- Patent status: Patent expiry in 2027 will be a pivotal moment influencing pricing.
- Competitor launches: Ingrezza’s dominance influences market share and pricing.
- Cost containment policies: Payers’ focus on lowering medication costs may pressure prices.
Summary: Risks and Opportunities
Risks:
- Patent expiry leading to generic competition.
- Emergence of new therapies with better efficacy or safety profiles.
- Changes in healthcare policies affecting drug reimbursement.
Opportunities:
- Expansion into additional indications.
- Geographic expansion outside the U.S.
- Formulation enhancements to improve adherence.
Key Takeaways
- AUSTEDO XR maintains a niche in Huntington’s disease and tardive dyskinesia treatment with moderate growth and stable pricing until patent expiry.
- The U.S. market for VMAT2 inhibitors is concentrated, with Ingrezza controlling most sales.
- Pricing is currently around $1,200/month; expect moderate increases until patent expiration.
- Generic entry in 2027 could reduce prices by 50–60%, impacting revenue.
- Ongoing demand in niche markets and potential expansion into new indications support continued relevance.
FAQs
1. What factors most influence AUSTEDO XR’s market share?
Market share depends on efficacy, safety profile, dosing frequency, insurance coverage, and patent status. Competition from Ingrezza remains significant.
2. How does patent expiration impact future pricing?
Patent expiration in 2027 is likely to lead to generic competition, causing prices to decrease substantially.
3. Are there opportunities for AUSTEDO XR outside the U.S.?
Yes. European markets with similar indications are potential expansion targets, subject to regulatory approval and market dynamics.
4. How does AUSTEDO XR compare in pricing to its competitors?
It is priced slightly lower than Ingrezza but higher than competing generic deutetrabenazine formulations.
5. What is the potential for new indications to influence sales?
Expansion into additional neuropsychiatric or neurological conditions could sustain growth post-patent expiration, contingent on regulatory approval.
References
- IQVIA. (2022). NHI drug sales data and market reports.
- FDA. (2017). Approval of AUSTEDO XR for chorea and tardive dyskinesia.
- EvaluatePharma. (2022). Global sales forecasts for VMAT2 inhibitors.
- Centers for Disease Control and Prevention. (2020). Prevalence of Huntington’s disease and tardive dyskinesia.
- FDA. (2023). Patent and exclusivity data.
More… ↓
