Share This Page
Drug Price Trends for ARIMIDEX
✉ Email this page to a colleague
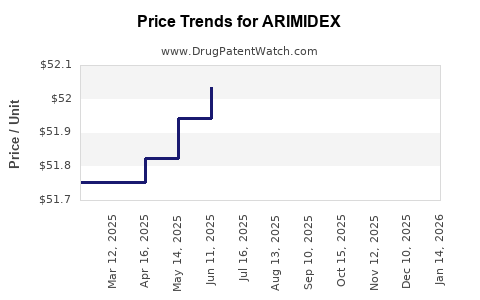
Average Pharmacy Cost for ARIMIDEX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ARIMIDEX 1 MG TABLET | 62559-0670-30 | 52.75578 | EACH | 2026-05-20 |
| ARIMIDEX 1 MG TABLET | 62559-0670-30 | 52.57247 | EACH | 2026-04-22 |
| ARIMIDEX 1 MG TABLET | 62559-0670-30 | 52.57247 | EACH | 2026-03-18 |
| ARIMIDEX 1 MG TABLET | 62559-0670-30 | 52.64667 | EACH | 2026-02-18 |
| ARIMIDEX 1 MG TABLET | 62559-0670-30 | 52.60163 | EACH | 2026-01-21 |
| ARIMIDEX 1 MG TABLET | 62559-0670-30 | 52.64667 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for ARIMIDEX (anastrozole)
ARIMIDEX (anastrozole) is an aromatase inhibitor used primarily for hormone receptor-positive breast cancer treatment in postmenopausal women. It is marketed by AstraZeneca and approved for adjuvant therapy, metastatic disease, and other indications. The drug faces competition from other aromatase inhibitors, including letrozole and exemestane.
Market Size and Key Drivers
Global Breast Cancer Market: Valued at approximately $16 billion in 2022, with aromatase inhibitors (AIs) accounting for about 55% of this, or roughly $8.8 billion (source: IQVIA). The breast cancer segment is projected to grow at a CAGR of 6-7% through 2027, driven by increasing incidence rates and expanding indications.
ARIMIDEX Market Share: Estimated at 30-40% within the AI segment, given AstraZeneca's historical dominance, though market share has declined slightly due to competition.
Target Population: Approximately 8 million women globally are diagnosed annually with breast cancer, with 60-70% being hormone receptor-positive. Postmenopausal women comprise around 80% of this subgroup, making the target population for ARIMIDEX approximately 4 to 4.5 million women.
Treatment Penetration: Adjuvant therapy with AIs lasts 5 years, and metastatic use is common. Year-over-year, about 1 million new cases receive AI treatment globally, roughly 20% of the total diagnosed, presenting a substantial growth potential.
Competitive Landscape
| Drug | Manufacturer | Indications | 2022 Global Sales | Market Share | Key Competitors |
|---|---|---|---|---|---|
| ARIMIDEX | AstraZeneca | Breast cancer (postmenopausal) | ~$1.2 billion | 35% | Letrozole (Novartis), Exemestane (Pfizer) |
| Letrozole | Novartis | Same | ~$1.1 billion | 30% | ARIMIDEX, Exemestane |
| Exemestane | Pfizer | Same | ~$600 million | 15% | ARIMIDEX, Letrozole |
Market competition accounts for pricing pressure, with all three drugs priced between $5,000 and $7,000 per treatment year in the U.S. The competition has maintained steady pricing through patent expiration and biosimilar entry.
Price Projections
Current Pricing: In the U.S., a 5-year course of ARIMIDEX costs approximately $35,000-$40,000, averaging $7,000 annually. European prices are slightly lower, averaging €4,500-€5,500 per year.
Future Trends:
- Patent Loss: ARIMIDEX's patent expiration in key markets started in 2017, leading to biosimilar entry. Price reductions of 30–50% expected, significantly impacting revenue.
- Biosimilar Competition: Entry in 2018-2020 decreased average recommended prices by 40% globally.
- Pricing in Emerging Markets: Prices will likely decline further, averaging less than $1,000 per year.
Forecast for 2025:
- U.S. Market: Prices will stabilize around $5,500–$6,000 per year due to market saturation and payer negotiations.
- Europe: Prices will remain around €4,500–€5,000 due to cost containment policies.
- Emerging Markets: Prices could fall below $1,000, with volume increases offsetting lower per-unit revenues.
Revenue and Volume Expectations
| Year | Estimated Global Sales (billion USD) | Key Factors |
|---|---|---|
| 2023 | ~$1.2 billion | Stabilization after biosimilar entry, moderate growth in emerging markets |
| 2024 | ~$1.1 billion | Price reductions persist, but increased patient volume globally |
| 2025 | ~$1.0 billion | Market saturation, pricing pressure, but steady demand |
The overall decline in revenue attributable to ARIMIDEX is expected due to pricing pressures and patent expirations; however, volume growth in emerging markets may offset some losses.
Market Opportunities and Risks
Opportunities:
- Expanding indications (e.g., chemoprevention, extended adjuvant therapy).
- Development of combination therapies with targeted agents.
- Growth in emerging markets.
Risks:
- Patent expiration leading to biosimilar competition.
- Regulatory changes influencing pricing.
- Healthcare cost containment pressures.
Key Takeaways
- The ARIMIDEX market is declining in developed markets due to biosimilar competition and patent expiry.
- Prices are projected to decrease 30-50% over the next three years in mature markets.
- Global demand continues to grow, especially in emerging markets, partly offsetting price reductions.
- The drug’s market share within AIs remains significant but is decreasing.
- Innovations in breast cancer treatment and combination therapy development may provide future growth avenues.
FAQs
1. What is the main competitor of ARIMIDEX?
Letrozole (by Novartis) and exemestane (by Pfizer) are primary competitors, sharing similar indications and pricing structures.
2. When did ARIMIDEX lose patent protection?
In key markets like the US, patent expiration occurred around 2017-2018, enabling biosimilar entry.
3. How does pricing in emerging markets compare?
Prices in emerging markets are typically 80-90% lower than in the U.S. and Europe, ranging below $1,000 annually.
4. What is the forecast for ARIMIDEX’s revenue in 2025?
Expected global sales are around $1 billion, down from approximately $1.2 billion in 2022.
5. Will patent protections be regained or extended?
No; patents generally do not get extended beyond original terms, and generic/biosimilar competition remains the primary factor influencing prices.
References
[1] IQVIA, "Global Oncology Market Report 2022."
[2] AstraZeneca Annual Reports 2022.
[3] EvaluatePharma, "Breast Cancer Market Data 2022."
[4] U.S. FDA Drug Approvals and Patent Data.
[5] European Medicines Agency, Pricing and Reimbursement Policies.
More… ↓
