Share This Page
Drug Price Trends for ANTISEPTIC SKIN CLEANSER
✉ Email this page to a colleague
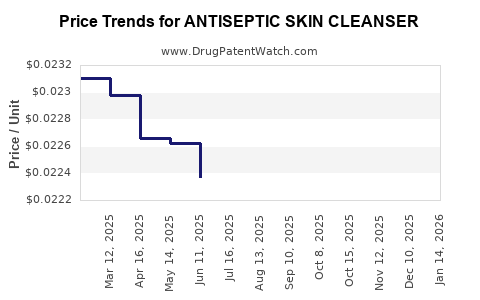
Average Pharmacy Cost for ANTISEPTIC SKIN CLEANSER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANTISEPTIC SKIN CLEANSER 4% | 46122-0137-34 | 0.03727 | ML | 2026-03-18 |
| ANTISEPTIC SKIN CLEANSER 4% | 68599-5401-02 | 0.04723 | ML | 2026-03-18 |
| ANTISEPTIC SKIN CLEANSER 4% | 68599-5401-03 | 0.03727 | ML | 2026-03-18 |
| ANTISEPTIC SKIN CLEANSER 4% | 68599-5401-04 | 0.01787 | ML | 2026-03-18 |
| ANTISEPTIC SKIN CLEANSER 4% | 68599-5401-04 | 0.01787 | ML | 2026-02-18 |
| ANTISEPTIC SKIN CLEANSER 4% | 68599-5401-02 | 0.04741 | ML | 2026-02-18 |
| ANTISEPTIC SKIN CLEANSER 4% | 46122-0137-34 | 0.03874 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ANTISEPTIC SKIN CLEANSER Market Analysis and Financial Projection
What is the Market Size and Its Growth Trajectory for Antiseptic Skin Cleansers?
The global antiseptic skin cleanser market was valued at approximately $2.4 billion in 2022. It is projected to grow at a Compound Annual Growth Rate (CAGR) of around 5.1% from 2023 to 2030, reaching an estimated $3.5 billion by 2030.
Key drivers include increased demand for hygiene products due to heightened awareness about personal cleanliness, especially following the COVID-19 pandemic. The rise in hygiene-related infections and the expansion of healthcare settings also boost market growth.
Regionally, North America holds the largest market share (around 40%), driven by higher consumer health awareness and established healthcare infrastructure. Asia-Pacific shows the fastest growth, with a CAGR near 6.8%, fueled by population density and rising urbanization.
How Are Market Dynamics Shaping Demand?
Market Drivers
- Increased public health initiatives emphasizing hand hygiene.
- Rising prevalence of skin infections and hygiene-related diseases.
- Expansion of hospital and clinical settings requiring antiseptic products.
- Elevated consumer awareness about personal skin care.
Market Restraints
- Regulatory complexities concerning active ingredients.
- Consumer concern over skin irritation and allergic reactions.
- Competition from alternative antiseptic and disinfectant products.
Competitive Landscape
Major players include Johnson & Johnson, Reckitt Benckiser, 3M, and Safetec of America. These firms invest in marketing and product innovation, including alcohol-based formulations and products with natural ingredients.
What Are Current Price Points and Factor Influences?
Pricing varies depending on formulation, branding, and packaging. Typical retail prices range from $3 to $8 per 250 ml bottle. Premium products with organic or dermatologist-recommended claims sometimes command prices exceeding $10 per bottle.
Wholesale prices to retailers hover around $1.50 to $4 per unit for standard 250 ml bottles, influenced by manufacturing costs, distribution logistics, and brand positioning.
Factors influencing price include:
- Ingredient costs: Alcohol-based antiseptics are sensitive to volatility in alcohol prices.
- Regulatory compliance costs: More strict regulations increase certification expenses.
- Packaging: Larger bottles or specialized packaging increase per-unit costs.
- Market segment: Consumer brands compete primarily on price and brand trust; professional-grade products emphasize efficacy and safety.
How Do Regulatory and Policy Environments Impact Pricing?
Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) enforce guidelines on active ingredients, claims, and labeling. These regulations influence manufacturing costs and can create barriers to entry, constraining price competition.
The EPA regulates environmental exposure, affecting formulations utilizing certain chemicals. Stricter regulations can increase manufacturing costs, impacting retail prices.
In jurisdictions with high regulatory compliance costs, manufacturers may pass expenses onto consumers, elevating prices. Conversely, deregulation in emerging markets may lower barriers, influencing local pricing strategies.
What Are Price Projections for 2025 and 2030?
Based on current growth trends, retail prices for standard antiseptic skin cleansers are expected to increase modestly, approximately 2% annually, reflecting inflation and raw material costs.
Price forecast summary:
| Year | Average Price Range (per 250 ml bottle) | Notes |
|---|---|---|
| 2023 | $3.00 – $8.00 | Current range |
| 2025 | $3.20 – $8.50 | Slight increase expected |
| 2030 | $3.50 – $9.50 | Inflation and ingredient costs factored in |
Premium products with organic or specialty claims may maintain higher price points, projecting increases exceeding 3% annually due to consumer willingness to pay for perceived safety and efficacy.
What Are Market Opportunities and Challenges?
Opportunities
- Increasing demand in emerging markets.
- Innovation in formulations, such as alcohol-free or skin-friendly products.
- Expansion into non-medical sectors, including hospitality and consumer retail.
Challenges
- Regulatory barriers that increase time-to-market.
- Consumer skepticism regarding efficacy versus traditional soaps.
- Competition from natural disinfectants and alternative skincare products.
Key Takeaways
- The antiseptic skin cleanser market is expanding, driven by increased hygiene awareness.
- Growth is strongest in Asia-Pacific, with North American dominance maintained through consumer preferences.
- Prices range widely, influenced by ingredients, packaging, and regulatory costs.
- Projected prices will increase modestly through 2030, with premium segments maintaining higher price points.
- Companies must navigate regulatory landscapes and innovate to capture market share.
FAQs
Q1: What active ingredients are standard in antiseptic skin cleansers?
Alcohol (ethanol or isopropanol) is predominant, with concentrations typically between 60-70%. Chlorhexidine and benzalkonium chloride are also used but face regulatory scrutiny.
Q2: How does regulatory status differ across regions?
In the U.S., the FDA regulates antiseptic claim safety and efficacy, with recent updates requiring more rigorous testing. EU regulations focus on ingredient safety and labeling. Emerging markets often have less comprehensive regulation, impacting product composition and pricing.
Q3: Are natural or organic antiseptic skin cleansers gaining market share?
Yes, consumer demand for natural products has increased, influencing formulations and pricing strategies. These products often command premiums and are marketed as safer alternatives.
Q4: What factors could limit market growth?
Stringent regulation, consumer concerns over chemical safety, and competition from alternative disinfectants (e.g., antimicrobial wipes) could slow growth.
Q5: How do pricing strategies differ between consumer and professional markets?
Consumer products emphasize affordability and brand recognition. Professional or medical-grade products prioritize efficacy, safety, and regulatory compliance, resulting in higher prices.
Sources
[1] MarketsandMarkets. "Antiseptic Skin Cleansers Market by Product, Application, and Region — Global Forecast to 2030."
[2] Statista. "Hand Sanitizer and Antiseptic Market Revenue Forecast."
[3] U.S. Food and Drug Administration. "Regulation of Over-the-Counter Skin Products."
[4] European Medicines Agency. "Guidelines on disinfectants and antiseptics."
More… ↓
