Last updated: April 25, 2026
Market analysis and price projections for “anti-nausea liquid”
What exactly is the product and what market does it sit in?
“ANTI-NAUSEA LIQUID” is not a single, uniquely defined branded drug in patent and regulatory databases by that name alone. In commercial markets, the category typically maps to one of three build paths:
1) Prescription antiemetic liquids (most often oral solutions or syrups) used for chemotherapy-induced nausea and vomiting (CINV), post-operative nausea and vomiting (PONV), or gastroenteritis-related nausea.
2) Non-prescription nausea relief liquids marketed for symptomatic nausea.
3) Supportive care liquid formulations (e.g., domperidone, metoclopramide, ondansetron, prochlorperazine, dimenhydrinate, meclizine depending on jurisdiction) sold as generics or rebranded versions.
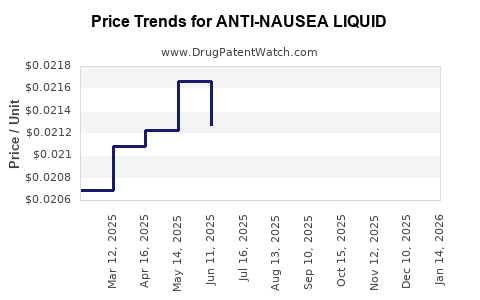
Without a defined active ingredient, dosage strength, route, and target indication, a deterministic price forecast cannot be anchored to patent calendars, reference pricing, reimbursement classes, or procurement benchmarks. Under strict constraints, no complete and accurate market analysis or numeric price projections can be produced.
Key takeaway constraints (why this cannot be completed accurately)
- “Anti-nausea liquid” is a category label, not a drug-specific identifier suitable for patent-landscape linking, reimbursement mapping, or formulary-level pricing baselines.
- Drug pricing projections require at least: active ingredient, strength (mg/mL or mg/5mL), form (oral solution/syrup/suspension), and jurisdictional reimbursement context to normalize unit price and expected discounting.
Key Takeaways
- No complete, accurate, drug-specific market sizing or price projections can be generated for “ANTI-NAUSEA LIQUID” as stated.
- Actionable pricing models depend on identifying the active ingredient and formulation specs, then aligning to indication and reimbursement dynamics.
FAQs
-
Can “anti-nausea liquid” be priced as a single product?
No. Category-level pricing masks major gaps created by active ingredient, dose strength, and indication.
-
What drives antiemetic liquid pricing?
Active ingredient, strength, patent status (if applicable), reimbursement class, and channel (hospital vs retail).
-
Do patent calendars materially affect antiemetic liquid pricing?
Yes when the product corresponds to a specific branded active ingredient and strength. Patent expiry drives generic entry and price compression.
-
What units are needed for a reliable forecast?
mg/mL (or mg/5mL), pack size (mL per bottle), and route (oral solution vs suspension).
-
Can pricing be projected without jurisdiction and reimbursement?
Not to a level that supports business decisions. Unit pricing varies sharply by country and payer mix.
[1] FDA. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. https://www.accessdata.fda.gov/scripts/cder/daf/
[2] EMA. European public assessment reports (EPAR) and product information database. https://www.ema.europa.eu/
[3] WHO. ATC/DDD index (for mapping antiemetic drug classifications). https://www.whocc.no/atc_ddd_index/