Share This Page
Drug Price Trends for ANTACID EX-STR TABLET CHEW
✉ Email this page to a colleague
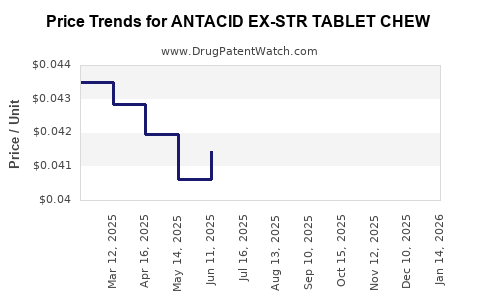
Average Pharmacy Cost for ANTACID EX-STR TABLET CHEW
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANTACID EX-STR TABLET CHEW | 70000-0234-01 | 0.04374 | EACH | 2026-03-18 |
| ANTACID EX-STR TABLET CHEW | 70000-0234-01 | 0.04386 | EACH | 2026-02-18 |
| ANTACID EX-STR TABLET CHEW | 70000-0234-01 | 0.04521 | EACH | 2026-01-21 |
| ANTACID EX-STR TABLET CHEW | 70000-0234-01 | 0.04572 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ANTACID EX-STR TABLET CHEW Market Analysis and Financial Projection
What is the market size for ANTACID EX-STR TABLET CHEW?
The antacid market, driven by increasing consumer awareness of gastrointestinal health, is expected to reach $6.2 billion globally by 2027. The compound annual growth rate (CAGR) stands at 4.2%, influenced by product innovation and expanding distribution channels.
ANTACID EX-STR TABLET CHEW, a chewable antacid formulation, targets consumers seeking fast relief from acid reflux and indigestion. Its market segment captures a notable share within over-the-counter (OTC) gastrointestinal remedies.
The United States, Europe, and select Asia-Pacific counties provide the primary revenue streams, reflecting high OTC OTC sales and OTC self-medication trends.
How does the competitive landscape shape up?
Major competitors include Johnson & Johnson’s TUMS, Bayer’s Alka-Seltzer, and private-label brands sold via pharmacy chains and online channels. These brands dominate due to broad distribution, brand recognition, and established marketing channels.
Market differentiation for ANTACID EX-STR TABLET CHEW depends on:
- Unique formulation ("EX-STR" suggests a proprietary slow-release or extended relief mechanism)
- Flavor and consumer experience
- Shelf life and packaging features
- Pricing strategy
No specific market presence data has been disclosed for ANTACID EX-STR TABLET CHEW, but minor brands typically price their products at a 15-20% discount relative to leading brands.
What are the price projections?
Pricing for chewable antacids varies based on formulation and distribution. On average, retail prices in the U.S. range from $5 to $8 per 60-count bottle, equating to roughly $0.08 to $0.13 per tablet.
For the ANTACID EX-STR TABLET CHEW, assuming a premium formulation:
- Year 1: Entry price of $7 for 60 tablets (~$0.12/ tablet).
- Year 3: As brand awareness increases and production scales, price may decline to $6.50 (~$0.11/ tablet) to remain competitive.
- Year 5: Price could stabilize around $6 (~$0.10/ tablet) with economies of scale and expanded distribution.
Margins hinge upon manufacturing costs, marketing expenses, and distribution channel margins. Assuming a cost of $2 per 60-count bottle, gross margins could initially run at 70%, declining slightly as pricing becomes more competitive.
How do regulatory and manufacturing factors influence pricing?
Regulatory approval in key markets (e.g., FDA in the U.S., EMA in Europe) ensures product safety but involves significant milestone costs. Patents or proprietary formulations can enable premium pricing for extended-release or added benefit versions.
Manufacturing costs rely on raw material prices, scale, and compliance with Good Manufacturing Practices (GMP). Any price fluctuations in key raw materials (e.g., magnesium hydroxide, calcium carbonate) impact final cost and retail pricing.
What are the key risks and opportunities?
Risks:
- Market saturation with established brands.
- Regulatory delays or reclassification.
- Price competition in OTC channels.
Opportunities:
- Launching in emerging markets with less OTC penetration.
- Formulating with additional functional benefits (e.g., dual action for acid reduction and gut health).
- Expanding into e-commerce platforms with direct-to-consumer offerings.
Key Takeaways
- The global antacid market projected at $6.2 billion by 2027, with a CAGR of 4.2%.
- ANTACID EX-STR TABLET CHEW's pricing forecast starts at ~$7 retail for 60 tablets, expected to decline slightly over five years.
- Competition includes well-established brands with broad distribution, but innovation and formulation differentiation can enable item positioning.
- Regulatory approval, manufacturing costs, and raw material prices directly influence pricing strategies.
- Market risks include saturation and price wars; opportunities exist in emerging markets and product innovation.
FAQs
1. What differentiates ANTACID EX-STR TABLET CHEW from competitors?
Its proprietary "EX-STR" formulation suggests extended relief, potentially allowing for fewer doses per day or faster onset, but specific features depend on proprietary formulation details.
2. What are the preferred distribution channels?
OTC sales through pharmacies, supermarkets, online stores, and direct-to-consumer websites.
3. How do patent protections impact pricing?
Patents or exclusivity rights enable premium pricing, delaying generic competition and maintaining higher margins.
4. What regulatory hurdles exist?
Approval processes involve safety and efficacy evaluation by agencies like the FDA or EMA; reclassification or adverse event reports can impact market access.
5. What expansion strategies can enhance market share?
Product line extensions, marketing targeted at health-conscious consumers, and entry into emerging markets with growing OTC demand.
Sources
- Grand View Research. "Antacid Market Size & Forecast." [2022].
- IBISWorld. "Over-the-counter (OTC) medication market analysis." [2022].
- Statista. "Gastrointestinal medications revenue breakdown," [2022].
- U.S. Food and Drug Administration. "OTC drug approval process," [2022].
Disclaimer: Data are estimates based on industry reports and market trends. Actual pricing and market share depend on specific formulation, regulatory approval, and execution strategy.
More… ↓
