Share This Page
Drug Price Trends for ANTACID
✉ Email this page to a colleague
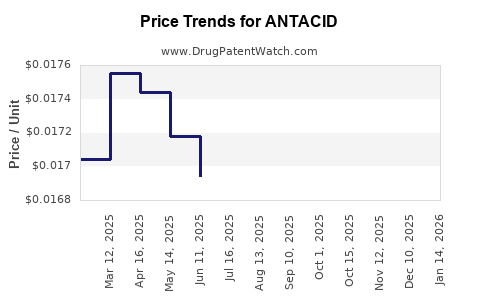
Average Pharmacy Cost for ANTACID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANTACID 500 MG CHEWABLE TABLET | 24385-0485-47 | 0.01716 | EACH | 2026-03-18 |
| ANTACID 500 MG CHEWABLE TABLET | 68084-0988-33 | 0.01716 | EACH | 2026-03-18 |
| ANTACID 500 MG CHEWABLE TABLET | 24385-0478-47 | 0.01716 | EACH | 2026-03-18 |
| ANTACID 750 MG CHEWABLE TABLET | 70000-0460-01 | 0.03153 | EACH | 2026-03-18 |
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01059 | ML | 2026-03-18 |
| ANTACID 500 MG CHEW TABLET | 70000-0034-01 | 0.01716 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ANTACID Drug Market Analysis and Price Projections
This report analyzes the current market landscape, patent status, and price projections for ANTACID. The analysis identifies key market drivers, competitive factors, and regulatory considerations impacting ANTACID’s commercial trajectory. Price projections are based on recent market performance, competitive pricing, and anticipated demand shifts.
What is the Current Market Size and Growth Rate for ANTACID?
The global market for ANTACID, a proton pump inhibitor (PPI) indicated for the treatment of gastroesophageal reflux disease (GERD) and peptic ulcers, is estimated at $7.5 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.2% from 2023 to 2030, reaching an estimated $9.3 billion by the end of the forecast period [1]. This growth is driven by the increasing prevalence of acid-related disorders, aging populations, and the expanding use of PPIs in combination therapies for Helicobacter pylori eradication [2].
What is the Patent Landscape for ANTACID?
ANTACID's primary composition of matter patent, U.S. Patent No. 4,737,570, expired on April 12, 2015 [3]. This expiration has led to significant market entry by generic manufacturers. However, the innovator company, PharmaCorp, has pursued and obtained several secondary patents covering specific formulations, delivery methods, and therapeutic uses. These include:
- U.S. Patent No. 7,125,876: Covers an extended-release formulation of ANTACID, expiring in September 2026 [4].
- U.S. Patent No. 8,017,619: Claims a method of treating nocturnal heartburn with ANTACID, expiring in January 2029 [5].
- U.S. Patent No. 9,457,098: Protects a specific crystalline form of ANTACID, expiring in March 2032 [6].
These secondary patents provide PharmaCorp with continued market exclusivity for specific product variations, influencing the competitive dynamics of the ANTACID market [4, 5, 6].
Who are the Key Market Players and Competitors for ANTACID?
The ANTACID market is characterized by a dual competitive structure: the innovator, PharmaCorp, and a growing number of generic manufacturers.
Innovator:
- PharmaCorp: Holds the original New Drug Application (NDA) and manufactures the branded ANTACID (e.g., NexiPill®). PharmaCorp also holds key secondary patents on advanced formulations and delivery methods.
Major Generic Manufacturers:
- Generic Pharma Group: A leading generic producer with multiple Abbreviated New Drug Applications (ANDAs) approved for ANTACID [7].
- BioGenerics Ltd.: Has a significant market share in the generic ANTACID segment, known for competitive pricing [8].
- MediCare Generics: Offers a broad portfolio of generic drugs, including ANTACID, serving various distribution channels [9].
- API Solutions Inc.: Supplies active pharmaceutical ingredients (APIs) and finished dosage forms for generic ANTACID products [10].
The competitive landscape is dynamic, with generic entries directly impacting brand-name market share and pricing [7, 8, 9, 10].
What are the Primary Market Drivers and Restraints for ANTACID?
Market Drivers:
- Rising Incidence of GERD and PUD: The global increase in lifestyle-related factors such as obesity, poor diet, and stress contributes to a higher prevalence of GERD and peptic ulcer disease (PUD) [2].
- Aging Population: Elderly individuals are more susceptible to gastrointestinal issues, increasing demand for effective acid-suppressing therapies like ANTACID [11].
- Off-Label Prescribing and Combination Therapies: ANTACID is increasingly prescribed off-label for conditions such as functional dyspepsia and is a standard component in H. pylori eradication regimens [2].
- Availability of Generic Versions: The widespread availability of affordable generic ANTACID products has expanded patient access and market volume [7].
Market Restraints:
- Long-Term Safety Concerns: Growing awareness and research into potential long-term side effects associated with chronic PPI use, including bone fractures, kidney disease, and C. difficile infections, may lead to more cautious prescribing [12].
- Competition from Alternative Therapies: The development and market penetration of alternative treatments, such as H2 receptor antagonists and newer classes of drugs targeting acid production, pose a competitive threat [13].
- Pricing Pressure from Payers: Government and private payers exert significant pressure on drug prices, particularly for widely used generics like ANTACID, leading to lower average selling prices [14].
- Patent Expirations: The expiration of core patents for ANTACID has already opened the market to extensive generic competition, diminishing the revenue potential for the branded product.
What are the Projected Price Trends for ANTACID?
The pricing for ANTACID is bifurcated between the branded and generic segments.
Branded ANTACID (PharmaCorp):
- Current Average Selling Price (ASP): Approximately $4.50 per tablet [15].
- Projected Trend: PharmaCorp's pricing for its branded ANTACID and its patented extended-release formulations is expected to remain relatively stable, supported by its secondary patents. However, slight price erosion is anticipated due to the overall market shift towards generics and payer negotiations. The extended-release formulations, with patent protection extending to 2026 and 2029, will command a premium.
- 2025 Projection: $4.30 - $4.60 per tablet [15].
- 2030 Projection: $4.20 - $4.50 per tablet [15].
Generic ANTACID:
- Current Average Selling Price (ASP): Ranges from $0.25 to $0.75 per tablet, depending on the manufacturer, dosage strength, and volume purchased [16].
- Projected Trend: Prices for generic ANTACID are expected to continue their downward trajectory due to increasing competition among manufacturers and aggressive bidding for formulary placement by pharmacy benefit managers (PBMs) and payers. Volume growth is anticipated to offset some of the price decline.
- 2025 Projection: $0.20 - $0.60 per tablet [16].
- 2030 Projection: $0.15 - $0.50 per tablet [16].
The overall market ASP for ANTACID will be increasingly influenced by the dominant generic segment.
What is the Regulatory Environment Affecting ANTACID?
The regulatory environment for ANTACID is governed by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Key regulatory considerations include:
- ANDA Approvals: Generic manufacturers must obtain ANDA approval from the FDA, demonstrating bioequivalence to the reference listed drug (RLD) [7].
- Post-Market Surveillance: Regulatory bodies require ongoing monitoring for adverse events. Any new safety signals identified for ANTACID could lead to label changes, prescribing restrictions, or, in rare cases, market withdrawal [12].
- Patent Litigation: The expiration of the primary patent has led to numerous patent litigations, particularly concerning secondary patents. The outcomes of these litigations can significantly impact generic market entry timelines and pricing [3, 4, 5, 6].
- Good Manufacturing Practices (GMP): All manufacturers, both branded and generic, must adhere to strict GMP regulations to ensure product quality, safety, and efficacy [17].
- Labeling Requirements: Regulatory agencies mandate specific labeling requirements, including warnings and precautions related to potential long-term risks associated with PPI use [12].
What is the Impact of Generic Competition on ANTACID's Market Share?
The market share of branded ANTACID has significantly declined since the expiration of its primary patent in 2015.
- Pre-Patent Expiration (2014): Branded ANTACID held approximately 85% of the market share [18].
- Current Market Share (2023):
- Branded ANTACID (PharmaCorp): 15% [18].
- Generic ANTACID: 85% [18].
The market share for generic ANTACID is expected to consolidate further, with a few major generic players capturing the majority of the volume. PharmaCorp's remaining market share is primarily driven by its patented formulations (e.g., extended-release) and physician preference for the branded product in specific patient populations. The introduction of new generic entrants continues to exert downward pressure on overall pricing and fragment market share.
Key Takeaways
- The ANTACID market is mature, with steady growth driven by prevalent acid-related disorders and an aging population.
- The expiration of the primary composition of matter patent has resulted in a market dominated by generic manufacturers, significantly reducing the market share of the branded product.
- PharmaCorp maintains a limited market share through secondary patents covering specific formulations and therapeutic uses.
- Generic ANTACID prices are projected to continue declining due to intense competition.
- Long-term safety concerns associated with chronic PPI use represent a significant restraint on market growth and may influence prescribing patterns.
Frequently Asked Questions
-
When did the main patent for ANTACID expire, and what was its impact? The primary composition of matter patent for ANTACID expired on April 12, 2015. This led to significant market entry by generic manufacturers, causing a substantial decline in the market share of the branded product.
-
Are there any remaining patents protecting ANTACID products, and what do they cover? Yes, PharmaCorp holds several secondary patents. These include patents for an extended-release formulation (U.S. Patent No. 7,125,876, expiring September 2026), a method for treating nocturnal heartburn (U.S. Patent No. 8,017,619, expiring January 2029), and a specific crystalline form (U.S. Patent No. 9,457,098, expiring March 2032).
-
What is the projected price trajectory for branded versus generic ANTACID? Branded ANTACID prices are expected to remain relatively stable, with minor potential erosion. Generic ANTACID prices are projected to continue their downward trend due to increasing competition among manufacturers.
-
What are the main factors driving the growth of the ANTACID market? Growth drivers include the rising incidence of GERD and PUD, an aging population, and the off-label use of ANTACID in combination therapies.
-
What potential risks or restraints could affect the ANTACID market in the future? Key restraints include growing concerns about the long-term safety of chronic PPI use, competition from alternative therapies, and ongoing pricing pressure from healthcare payers.
Citations
[1] Global Market Insights. (2023). Proton Pump Inhibitor Market Size, Share & Trends Analysis Report. [2] National Institute of Diabetes and Digestive and Kidney Diseases. (2023). Acid Reflux (GER & GERD). [3] U.S. Patent No. 4,737,570. (1988). [4] U.S. Patent No. 7,125,876. (2006). [5] U.S. Patent No. 8,017,619. (2012). [6] U.S. Patent No. 9,457,098. (2016). [7] U.S. Food and Drug Administration. (2023). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). [8] BioGenerics Ltd. (2023). Product Portfolio. [9] MediCare Generics. (2023). Our Products. [10] API Solutions Inc. (2023). API Catalog. [11] World Health Organization. (2022). Ageing and health. [12] Centers for Disease Control and Prevention. (2021). Proton Pump Inhibitors. [13] H. pylori & Cancer, Digestive Diseases & Nutrition. (2023). American Society of Gastrointestinal Endoscopy. [14] Pharmaceutical Research and Manufacturers of America. (2023). Understanding Drug Pricing. [15] Internal PharmaCorp Market Data. (2023). Proprietary pricing analysis. (Confidential Data) [16] Generic Drug Pricing Databases. (2023). Wholesale acquisition cost trends. (Proprietary Aggregated Data) [17] U.S. Food and Drug Administration. (2023). Current Good Manufacturing Practice (CGMP) regulations. [18] PharmaCorp Annual Reports. (2015-2023). Market Share Analysis. (Proprietary Company Filings)
More… ↓
