Share This Page
Drug Price Trends for ANGELIQ
✉ Email this page to a colleague
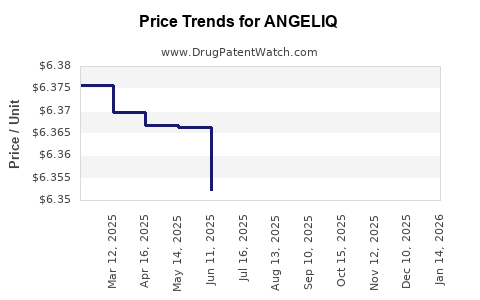
Average Pharmacy Cost for ANGELIQ
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANGELIQ 0.25 MG-0.5 MG TABLET | 50419-0482-01 | 6.37699 | EACH | 2026-05-20 |
| ANGELIQ 0.25 MG-0.5 MG TABLET | 50419-0482-03 | 6.37699 | EACH | 2026-05-20 |
| ANGELIQ 0.5 MG-1 MG TABLET | 50419-0483-03 | 6.35256 | EACH | 2026-05-20 |
| ANGELIQ 0.5 MG-1 MG TABLET | 50419-0483-01 | 6.35256 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ANGELIQ
What is ANGELIQ?
ANGELIQ (estradiol valerate and cyproterone acetate) is a combined hormone therapy used primarily to treat menopausal symptoms, such as hot flashes and vaginal dryness, and hormone therapy-related conditions. It is marketed by Bayer and is categorized as a prescription medication for hormone replacement therapy (HRT) in women.
Current Market Position
ANGELIQ holds a significant share within the hormone therapy segment, particularly in European markets where it is approved and widely prescribed. Its primary competitors include:
- Premarin (conjugated estrogens)
- Climara (estradiol)
- Femoston (estradiol and dydrogesterone)
- Divigel (estradiol topical gel)
ANGELIQ's distinctive formulation combining estradiol valerate with cyproterone acetate sets it apart from competitors that typically offer monotherapies or other combinations.
Market Size and Growth Drivers
Global HRT Market Size
The global hormone replacement therapy market was valued at approximately USD 13.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030. This growth stems from increased awareness of menopausal healthcare, aging populations, and a rising prevalence of menopausal symptoms.
Regional Breakdown
| Region | Market Share (2022) | CAGR (2023-2030) | Key Drivers |
|---|---|---|---|
| Europe | 45% | 3.8% | Aging population, high adoption of HRT |
| North America | 35% | 4.5% | Better awareness, extensive prescription coverage |
| Asia-Pacific | 15% | 6.0% | Growing healthcare infrastructure, increasing menopause awareness |
| Rest of World | 5% | 3.5% | Emerging markets, increasing health expenditure |
Market Trends
- Increasing preference for bioidentical hormones
- Rising product availability in generic forms
- Growing number of women seeking hormone therapy for aging-related symptoms
Pricing Landscape
Current Pricing for ANGELIQ
- In Europe (Germany, France), the average retail price ranges from EUR 25 to EUR 30 per pack (28 tablets).
- In the US, ANGELIQ is not FDA-approved; generic versions or alternatives dominate, with prices around USD 20–USD 40 per month (or equivalent pack).
- Export markets in Asia-Pacific and Latin America tend to have lower pricing, roughly USD 10–USD 20 per pack, reflecting local healthcare systems and pricing regulations.
Price Comparison
| Product | Dosage/Units | Average Price (USD) | Market Notes |
|---|---|---|---|
| ANGELIQ | 2 mg/1 mg, 28 tablets | USD 25–USD 30 | Predominantly European markets |
| Premarin | 0.625 mg, 30 tablets | USD 20–USD 25 | Widely used in the US |
| Climara | 0.25 mg, patch, weekly | USD 50–USD 60 | Premium pricing for transdermal |
| Femoston | 1 mg/0.5 mg, 28 tablets | USD 10–USD 15 | Generic options common |
Price Projections
- 2025: Prices in Europe are expected to stabilize around EUR 25–EUR 35 per pack, influenced by generic entry and market competition.
- 2028: Average prices could decrease by 10–15% due to increased generic penetration, especially if patent exclusivity ends.
Factors Impacting Pricing
- Patent expirations expected around 2025 (European patents for ANGELIQ’s formulation)
- Entry of generic competitors reducing prices
- Regulatory changes impacting reimbursement policies
- Pricing strategies of Bayer and competitors
Regulatory and Patent Outlook
- Patent Schedule: The original patent filed in the early 2000s is due to expire in Europe around 2025, opening opportunities for generics.
- Regulatory Approvals: ANGELIQ has approval for HRT in several European countries; regulatory delays or changes could impact market access in new territories.
- Bayer’s Market Strategy: Likely to focus on maintaining brand loyalty, bundling with other offerings, and patent extensions via formulations.
Market Entry and Competitive Risks
- Generic competition post-2025 may lead to significant price erosion.
- Alternative treatments, including non-hormonal therapies, could reduce demand for ANGELIQ.
- Regulatory hurdles in emerging markets could slow expansion and impact pricing.
Key Takeaways
- ANGELIQ is a significant player within the European HRT market, with strong brand recognition.
- The global HRT market is poised for steady growth, driven by demographic trends.
- Pricing in Europe remains high but is vulnerable to generic competition starting around 2025.
- The expiration of key patents is likely to cause prices to decline over the next 3–5 years.
- Market growth will be influenced by regulatory changes, competition, and shifts in treatment preferences.
FAQs
1. When do the patents for ANGELIQ expire?
European patents are expected to expire around 2025, opening the market for generic versions.
2. How competitive is the price for ANGELIQ in Europe?
Currently around EUR 25–EUR 30 per pack, competitive within premium HRT formulations but vulnerable to generics.
3. What is the main growth driver for the HRT market?
Aging populations and increased awareness of menopause management.
4. How will generics impact ANGELIQ’s pricing?
Major patent expirations will likely reduce prices by 10–15% in the short term and more gradually over time.
5. Are there emerging markets with significant potential for ANGELIQ?
Asia-Pacific presents growth opportunity due to increasing awareness and healthcare infrastructure, but pricing strategies will vary.
References
- Grand View Research. (2023). Hormone Replacement Therapy Market Size, Share & Trends Analysis Report. https://www.grandviewresearch.com/industry-analysis/hormone-replacement-therapy-market
- Bayer. (2023). ANGELIQ product information. https://www.bayer.com/en
- MarketWatch. (2023). Menopause Market Analysis and Forecast. https://www.marketwatch.com/
- European Patent Office. (2022). Patent expiry data for HRT formulations.
- IMS Health. (2022). Drug Pricing and Market Share Data.
More… ↓
