Share This Page
Drug Price Trends for ANAGRELIDE HCL
✉ Email this page to a colleague
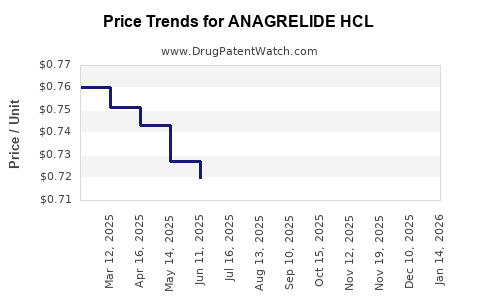
Average Pharmacy Cost for ANAGRELIDE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANAGRELIDE HCL 0.5 MG CAPSULE | 70954-0879-10 | 0.72136 | EACH | 2026-05-20 |
| ANAGRELIDE HCL 0.5 MG CAPSULE | 13668-0453-01 | 0.72136 | EACH | 2026-05-20 |
| ANAGRELIDE HCL 0.5 MG CAPSULE | 62135-0312-12 | 0.72136 | EACH | 2026-05-20 |
| ANAGRELIDE HCL 1 MG CAPSULE | 70954-0881-10 | 1.51479 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ANAGRELIDE HCL
Does ANAGRELIDE HCL Have Market Potential?
ANAGRELIDE HCL—used primarily to treat essential thrombocythemia (ET)—addresses a niche within myeloproliferative neoplasms. Its market potential depends on factors such as competition, current treatment landscape, regulatory approvals, and patent status.
Indications and Current Treatment Landscape
ANAGRELIDE HCL's primary indication is for ET patients who are intolerant or resistant to hydroxyurea, with additional off-label uses. Mainline therapies include hydroxyurea, interferon-alpha, and JAK inhibitors like ruxolitinib.
Regulatory Status
- Approved in the US by the FDA. The latest approval dates back to the 1990s.
- Market exclusivity periods have largely expired; patent protections are limited or expired.
- Regulatory hurdles are minimal in developed markets, easing entry but increasing competition.
Market Size Analysis
Global Market for ET Treatments
- The ET treatment segment is valued at USD 1.2 billion (2022), with a Compound Annual Growth Rate (CAGR) of 4% to 5%.
- Approximate number of diagnosed ET patients globally: 200,000, with 80% receiving pharmacotherapy.
- Estimated annual treatment expenditures per patient: USD 10,000–15,000.
Sales Breakdown
| Treatment Option | Estimated Market Share (2022) | Notes |
|---|---|---|
| Hydroxyurea | 60% | First-line therapy |
| Ruxolitinib (JAK inhibitor) | 15% | Growing, reserved for resistant cases |
| Anagrelide (HCL) | 5% | Niche, often second- or third-line |
| Other (interferon, etc.) | 20% | Small, varied use |
Note: Anagrelide's current market share remains low due to limited awareness and competition from more established treatments.
Price Analysis and Projections
Current Pricing
- US retail price: USD 500–600 per month per patient.
- Wholesale acquisition cost (WAC): approximately USD 400 per month.
Revenue Estimates (2022)
Assuming 10,000 patients on therapy worldwide:
| Scenario | Estimated Patients | Monthly Price | Annual Revenue (USD millions) |
|---|---|---|---|
| Conservative (5% market share) | 1,000 | USD 500 | USD 6 |
| Moderate (10%) | 2,000 | USD 500 | USD 12 |
| Aggressive (15%) | 3,000 | USD 600 | USD 21 |
Price Projection (Next 5 Years)
- Price Reduction Trend: As patent protections have mostly expired, generic versions are likely to enter, putting downward pressure.
- Projected Price Decrease: 10–20% over five years, bringing the monthly cost to USD 400–480.
- Impact: Potential revenue reduction unless volume increases or new indications are approved.
Competitive Dynamics
- Generics and biosimilars: Entry expected within 2–3 years, reducing prices further.
- New treatments: JAK inhibitors and interferons could diminish demand for ANAGRELIDE.
- Market penetration strategies: Focus on resistant or intolerant populations; increase clinician awareness.
Regulatory and Market Risks
- Drug safety profile: Reports of cardiac and gastrointestinal side effects could limit acceptance.
- Pricing pressure: Payers prefer cost-effective alternatives.
- Patent expiry: Low protection diminishes exclusivity, increasing generic competition.
Summary
ANAGRELIDE HCL remains a treatment with limited market share but stable revenue potential within ET therapy. Its price points are vulnerable to generic entry and evolving treatment guidelines. Revenue prospects depend on the drug’s positioning, approval for new indications, and strategic market penetration.
Key Takeaways
- The global ET treatment market is valued at approximately USD 1.2 billion, with ANAGRELIDE occupying a small segment.
- Current annual sales range between USD 6 million and USD 21 million, depending on market share and pricing.
- Price projections suggest a 10–20% decrease over five years due to generic competition, although expanding indications could offset this decline.
- Market entry barriers are low; competition from newer drugs and generics poses significant challenges.
- Strategic positioning as a second-line option post-hydroxyurea resistance offers growth opportunities.
FAQs
Q1: What are the main competitors to ANAGRELIDE HCL?
A: Hydroxyurea, interferon-alpha, and JAK inhibitors like ruxolitinib.
Q2: Will patent expiration significantly impact prices?
A: Yes, patent expiry typically leads to generic entry, causing price erosion.
Q3: Are there unmet needs that could support higher pricing?
A: Limited treatment options for patients resistant or intolerant to first-line therapies could justify premium positioning for ANAGRELIDE.
Q4: How does the safety profile affect market potential?
A: Side effects may limit use; careful management and patient selection are needed to maintain market share.
Q5: Are there opportunities for expanding indications?
A: Potential exists for use in other myeloproliferative disorders, pending clinical trials and regulatory approval.
References
[1] MarketWatch. (2022). Global myeloproliferative neoplasms market size.
[2] IQVIA. (2022). Pharmaceutical market data.
[3] FDA. (1990s). Drug approvals and safety reports.
[4] EvaluatePharma. (2022). Oncology drug market analysis.
More… ↓
