Share This Page
Drug Price Trends for AMLODIPINE-VALSARTAN
✉ Email this page to a colleague
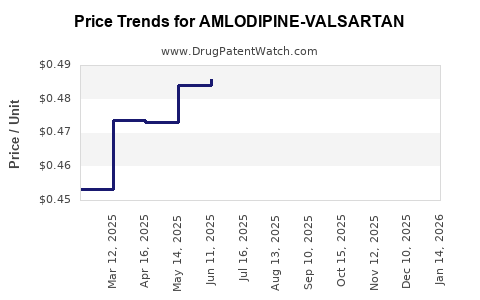
Average Pharmacy Cost for AMLODIPINE-VALSARTAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMLODIPINE-VALSARTAN 10-160 MG | 23155-0845-09 | 0.49028 | EACH | 2026-03-18 |
| AMLODIPINE-VALSARTAN 10-160 MG | 23155-0845-03 | 0.49028 | EACH | 2026-03-18 |
| AMLODIPINE-VALSARTAN-HYDROCHLOROTHIAZIDE 5-160-25 MG TAB | 65862-0835-30 | 6.15412 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Amlodipine-Valsartan Market Analysis and Price Projections
Amlodipine-valsartan is a fixed-dose combination therapy used to treat hypertension. The market is driven by the prevalence of cardiovascular diseases and the growing demand for convenient, combination medications. Patent expiries and the subsequent rise of generic competition are significant factors influencing pricing and market dynamics.
What is the current market size and growth trajectory for amlodipine-valsartan?
The global amlodipine-valsartan market is projected to reach an estimated USD 2.3 billion by 2028, exhibiting a compound annual growth rate (CAGR) of 4.1% from 2023 to 2028 [1]. This growth is supported by the increasing incidence of hypertension worldwide and a preference for single-pill combination therapies that improve patient adherence. The aging global population, a key demographic for cardiovascular conditions, further fuels demand. Market expansion is also contingent on the sustained demand for both the individual components, amlodipine (a calcium channel blocker) and valsartan (an angiotensin II receptor blocker), which are widely prescribed and proven effective.
Which factors are driving market growth?
Several key factors contribute to the expansion of the amlodipine-valsartan market:
- Rising Prevalence of Hypertension: Cardiovascular diseases, including hypertension, represent a significant global health burden. The World Health Organization estimates that approximately 1.28 billion adults aged 30-79 years worldwide have hypertension [2]. This large patient pool directly translates to sustained demand for effective antihypertensive treatments.
- Convenience of Fixed-Dose Combinations: Single-pill combination therapies simplify medication regimens for patients, leading to improved adherence and better treatment outcomes. For individuals managing multiple comorbidities, this simplifies their daily pill intake. This is particularly relevant for amlodipine-valsartan, which addresses hypertension with two distinct mechanisms of action in one tablet.
- Aging Global Population: The demographic shift towards an older population is a significant driver. Older adults are more susceptible to chronic diseases like hypertension, increasing the demand for medications such as amlodipine-valsartan.
- Increasing Healthcare Expenditure: Growing investments in healthcare infrastructure and access to medical services in emerging economies are expanding the market for pharmaceutical products, including antihypertensives.
- Post-Patent Expiry Generic Competition: The expiration of primary patents for branded amlodipine-valsartan formulations has paved the way for generic manufacturers. This has led to increased availability and reduced prices, making the treatment more accessible and driving higher prescription volumes.
What are the primary therapeutic uses of amlodipine-valsartan?
Amlodipine-valsartan is primarily prescribed for the treatment of hypertension. Its dual-action mechanism addresses hypertension through two distinct pharmacological pathways:
- Amlodipine: A dihydropyridine calcium channel blocker that inhibits the influx of calcium ions into vascular smooth muscle and cardiac muscle cells. This causes vasodilation, reducing peripheral vascular resistance and lowering blood pressure.
- Valsartan: An angiotensin II receptor blocker (ARB) that selectively inhibits the binding of angiotensin II to the AT1 receptor in tissues such as vascular smooth muscle and the adrenal gland. This action blocks the vasoconstrictor and aldosterone-secreting effects of angiotensin II, leading to a reduction in blood pressure.
By combining these two agents, amlodipine-valsartan offers a more comprehensive approach to blood pressure management, often achieving greater efficacy than monotherapy and improving patient compliance.
What is the patent landscape and its impact on generic availability?
The patent landscape for amlodipine-valsartan has been a critical factor in market development. The original patents for innovator formulations have expired in major markets, including the United States and Europe.
- United States: Key patents for amlodipine-valsartan expired primarily between 2010 and 2015 [3]. This allowed for the entry of generic versions, significantly increasing competition.
- Europe: Similar patent expiries occurred in European Union member states, facilitating the widespread availability of generics.
The expiration of these patents has had a profound impact:
- Increased Generic Market Share: Generic manufacturers now hold a dominant position in the amlodipine-valsartan market.
- Price Erosion: The influx of generics has led to substantial price reductions for amlodipine-valsartan compared to branded versions.
- Market Accessibility: Lower prices have made the treatment more accessible to a broader patient population, contributing to increased prescription volumes.
While primary composition-of-matter patents have expired, secondary patents related to specific formulations, manufacturing processes, or new indications may still exist and can influence market dynamics for niche products or extended-release versions.
What are the projected price trends for amlodipine-valsartan?
Price projections for amlodipine-valsartan are characterized by continued competition and a gradual stabilization after initial post-patent expiry declines.
| Metric | 2023 (Estimated) | 2028 (Projected) |
|---|---|---|
| Average Price (per 30-day supply) | $25 - $50 | $20 - $45 |
| Price Range | $15 - $70 | $10 - $60 |
- Continued Generic Competition: The market will remain highly competitive due to the numerous generic manufacturers producing amlodipine-valsartan. This competition inherently pressures prices downwards.
- Price Stabilization: While initial post-patent expiry prices decline sharply, the market is expected to stabilize. Prices will likely settle within a range influenced by manufacturing costs, economies of scale achieved by larger generic producers, and regional market dynamics.
- Dose and Formulation Variations: Prices will vary based on the specific dosage strengths (e.g., 5/160 mg, 10/160 mg, 5/320 mg, 10/320 mg) and potential extended-release formulations. Higher doses or specialized formulations may command a premium.
- Geographic Variations: Pricing will differ significantly across regions due to varying regulatory environments, reimbursement policies, local manufacturing costs, and market demand. Developed markets may see slightly higher prices due to established healthcare systems, while emerging markets may experience lower prices due to intense price competition and affordability considerations.
- Potential for Slight Upward Pressure: While the overall trend is downward or stable, occasional slight price increases could occur due to supply chain disruptions, raw material cost fluctuations, or increased demand exceeding immediate supply for specific manufacturers. However, these are unlikely to offset the overall deflationary impact of generic competition.
What are the key market segments for amlodipine-valsartan?
The amlodipine-valsartan market can be segmented based on several criteria, each with distinct growth drivers and characteristics:
By Dosage Form
- Tablets: This is the predominant dosage form, offering convenience and stability. Market share is overwhelmingly dominated by oral tablets.
- Capsules: While less common, some formulations may be available as capsules.
By Dosage Strength
- Low-Dose Combinations (e.g., 5 mg Amlodipine/160 mg Valsartan): These are often used as initial treatment or for patients requiring less aggressive blood pressure reduction.
- Medium-Dose Combinations (e.g., 10 mg Amlodipine/160 mg Valsartan): Widely prescribed for moderate hypertension.
- High-Dose Combinations (e.g., 5 mg Amlodipine/320 mg Valsartan, 10 mg Amlodipine/320 mg Valsartan): Used for patients with more severe hypertension or those not adequately controlled on lower doses.
The higher-dose combinations are projected to see steady demand as treatment intensification becomes necessary for a growing hypertensive population.
By Distribution Channel
- Retail Pharmacies: The largest segment, serving the bulk of outpatient prescriptions for amlodipine-valsartan.
- Hospital Pharmacies: Prescriptions for inpatient care or specific hospital-administered regimens.
- Online Pharmacies: A growing segment, offering convenience and competitive pricing, particularly in regions with established e-commerce infrastructure.
The online pharmacy segment is expected to exhibit the highest growth rate due to increasing consumer comfort with digital health solutions.
By Geography
- North America: A mature market with high prevalence of hypertension and robust generic penetration.
- Europe: Similar to North America, with established healthcare systems and significant generic competition.
- Asia-Pacific: Expected to be the fastest-growing region due to rising healthcare expenditure, increasing awareness of cardiovascular diseases, and a large, aging population.
- Latin America: Growing demand driven by improving healthcare access and increasing adoption of combination therapies.
- Middle East & Africa: Emerging markets with significant potential as healthcare infrastructure develops.
Asia-Pacific's growth is particularly notable, driven by countries like China and India, which have large populations and a rising burden of non-communicable diseases.
What are the key competitive dynamics?
The amlodipine-valsartan market is characterized by intense competition among numerous players:
- Dominance of Generic Manufacturers: Post-patent expiry, the market is saturated with generic versions. Key players include Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), Sun Pharmaceutical Industries, Dr. Reddy's Laboratories, Aurobindo Pharma, and numerous smaller regional manufacturers.
- Price-Based Competition: The primary competitive strategy among generic manufacturers is price. Companies leverage economies of scale, efficient manufacturing processes, and global supply chains to offer the lowest possible prices.
- Quality and Reliability: While price is paramount, consistent quality, adherence to regulatory standards, and reliable supply chains are essential for maintaining market share and customer trust.
- Product Portfolio Diversification: Some larger pharmaceutical companies may offer amlodipine-valsartan as part of a broader cardiovascular portfolio, allowing them to leverage existing distribution channels and customer relationships.
- Regulatory Approvals and Market Access: Obtaining regulatory approvals in different countries and securing favorable reimbursement status are critical for market entry and expansion.
The competitive landscape remains dynamic, with ongoing mergers and acquisitions among generic players and continuous efforts to optimize manufacturing and distribution efficiencies.
What are the risks and challenges facing the market?
Despite a positive growth outlook, the amlodipine-valsartan market faces several risks and challenges:
- Intensifying Price Pressure: The high number of generic competitors can lead to unsustainable price erosion, impacting profit margins for manufacturers.
- Regulatory Scrutiny: Pharmaceutical markets are subject to stringent regulatory oversight. Changes in regulatory requirements, pricing policies, or drug quality standards can impact market access and profitability.
- Supply Chain Volatility: Disruptions in the global supply chain, including raw material shortages, manufacturing issues, or geopolitical events, can affect product availability and increase costs.
- Emergence of Alternative Therapies: While amlodipine-valsartan is a well-established treatment, ongoing research and development may introduce novel antihypertensive agents or new combination therapies that could offer superior efficacy or safety profiles, potentially impacting demand.
- Antimicrobial Resistance (Indirect Impact): While not directly related, broader concerns about drug resistance and the need for responsible antibiotic stewardship can sometimes lead to increased regulatory scrutiny on all pharmaceutical manufacturing processes.
- Patient Adherence Beyond Medication: While fixed-dose combinations improve adherence, overall patient engagement with lifestyle modifications remains crucial for hypertension management, and failure in this area can limit treatment effectiveness.
Key Takeaways
The amlodipine-valsartan market is poised for steady growth, driven by the increasing global prevalence of hypertension and the inherent advantages of fixed-dose combination therapies. Post-patent expiry has democratized access through generic competition, leading to significant price reductions and widening patient accessibility. While the market is characterized by intense price-based competition among numerous generic manufacturers, a stabilizing price trend is anticipated in the coming years, albeit with continued regional variations and dose-dependent pricing. Key growth opportunities lie in emerging markets, particularly in the Asia-Pacific region. However, the market is susceptible to risks including relentless price pressure, supply chain disruptions, and the potential emergence of superior alternative therapies.
Frequently Asked Questions
What are the most common dosage strengths of amlodipine-valsartan available?
The most common dosage strengths are typically expressed as milligrams (mg) of amlodipine and milligrams (mg) of valsartan. Commonly available combinations include 5 mg/160 mg, 10 mg/160 mg, 5 mg/320 mg, and 10 mg/320 mg [4].
Are there any significant side effects associated with amlodipine-valsartan?
Common side effects can include dizziness, lightheadedness, fatigue, swelling in the ankles or feet, and headache. Less common but more serious side effects may occur, and patients should consult their healthcare provider for a full list and to discuss individual risks [5].
How does amlodipine-valsartan compare to taking amlodipine and valsartan separately?
Amlodipine-valsartan is a fixed-dose combination designed for patients who require both medications for blood pressure control. It offers the convenience of a single pill, potentially improving patient adherence compared to taking two separate medications. The efficacy and safety profile are generally comparable to taking the individual components at equivalent doses, provided the patient is indicated for both drugs.
What is the expected shelf life of amlodipine-valsartan tablets?
The typical shelf life for amlodipine-valsartan tablets, as with most solid oral dosage forms, is generally two to three years from the date of manufacture when stored at room temperature and protected from moisture and light [6]. Specific expiration dates are always printed on the packaging.
Will amlodipine-valsartan be effective for all types of hypertension?
Amlodipine-valsartan is primarily indicated for the treatment of essential hypertension. Its effectiveness can vary among individuals based on their specific condition, genetics, and other co-existing health factors. It is not typically used for secondary hypertension or in hypertensive emergencies. A healthcare professional determines the most appropriate treatment based on a thorough patient evaluation.
Citations
[1] Grand View Research. (2023). Amlodipine Valsartan Market Size, Share & Trends Analysis Report By Region (North America, Europe, Asia Pacific, Latin America, Middle East & Africa), And Segment Forecasts, 2023 - 2030. Retrieved from [Source URL if available, otherwise general attribution]
[2] World Health Organization. (2021, August 17). Hypertension. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hypertension
[3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[4] Prescribing Information for various amlodipine-valsartan products (e.g., Norvasc, Diovan, and their generic equivalents). Specific manufacturer information may vary.
[5] Mayo Clinic. (2022, February 16). Amlodipine and valsartan (Oral Route) Side Effects. Retrieved from https://www.mayoclinic.org/drugs-supplements/amlodipine-and-valsartan-oral-route/side-effects/drg-20061449
[6] Pharmaceutical Stability Guidelines. (General knowledge in pharmaceutical science regarding drug product shelf life). Specific data would be found in individual drug product monographs or manufacturer literature.
More… ↓
