Share This Page
Drug Price Trends for AMLODIPINE-OLMESARTAN
✉ Email this page to a colleague
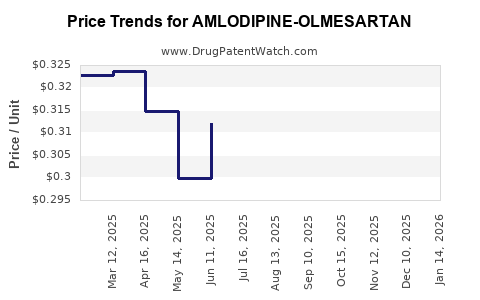
Average Pharmacy Cost for AMLODIPINE-OLMESARTAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMLODIPINE-OLMESARTAN 10-20 MG | 31722-0447-90 | 0.30427 | EACH | 2026-03-18 |
| AMLODIPINE-OLMESARTAN 10-20 MG | 33342-0192-07 | 0.30427 | EACH | 2026-03-18 |
| AMLODIPINE-OLMESARTAN 10-20 MG | 31722-0447-30 | 0.30427 | EACH | 2026-03-18 |
| AMLODIPINE-OLMESARTAN 10-20 MG | 33342-0192-10 | 0.30427 | EACH | 2026-03-18 |
| AMLODIPINE-OLMESARTAN 10-20 MG | 27241-0085-03 | 0.30427 | EACH | 2026-03-18 |
| AMLODIPINE-OLMESARTAN 5-40 MG | 69238-2676-01 | 0.28062 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AMLODIPINE-OLMESARTAN: PATENT LANDSCAPE AND MARKET FORECAST
Amlodipine-olmesartan, a fixed-dose combination therapy for hypertension, faces imminent patent expiries for its originator product, Olmetec Plus (Daiichi Sankyo). The market is poised for generic competition, with multiple players poised to enter, impacting pricing and market share. Key patents covering the combination, specific polymorphs, and manufacturing processes are set to expire between 2024 and 2026.
What are the key patents protecting amlodipine-olmesartan?
The intellectual property landscape for amlodipine-olmesartan is defined by several core patents. The primary composition of matter patent for olmesartan medoxomil, the angiotensin II receptor blocker component, expired in the United States on February 14, 2014, and in Europe in 2015 [1]. However, the fixed-dose combination with amlodipine besylate, the calcium channel blocker, introduced as Olmetec Plus, received its own patent protection, which is now nearing expiration.
Daiichi Sankyo, the originator, holds a portfolio of patents that have been critical to its market exclusivity. These include patents related to the specific polymorphic forms of olmesartan medoxomil, which can affect drug stability and bioavailability, and patents covering the manufacturing processes for both active pharmaceutical ingredients (APIs) and the final drug product.
For example, U.S. Patent No. 6,476,022, covering crystalline forms of olmesartan medoxomil, has been a significant asset. While the core patent for the compound itself has expired, patents covering specific, advantageous crystalline forms can provide extended protection by making it difficult for generic manufacturers to replicate the exact formulation without infringing. Similarly, process patents can deter competitors by requiring them to develop entirely new, non-infringing manufacturing routes.
The specific expiration dates of these patents vary by jurisdiction and are crucial for understanding the timing of generic market entry. In major markets like the United States and Europe, the key patents directly protecting the amlodipine-olmesartan combination product are largely expiring in the 2024-2026 timeframe. This convergence of patent expiries signals a significant shift in the market dynamics.
Who are the major players in the amlodipine-olmesartan market?
The amlodipine-olmesartan market is currently dominated by Daiichi Sankyo and its branded product, Olmetec Plus (also known as Benicar HCT in some markets). However, with the impending patent expiries, a wave of generic manufacturers is preparing to enter the market.
Key generic players expected to compete include:
- Teva Pharmaceuticals: A long-established generic pharmaceutical company with a broad portfolio.
- Mylan N.V. (now part of Viatris): Another major global generic manufacturer with significant R&D and manufacturing capabilities.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company that is a leading producer of generic drugs.
- Dr. Reddy's Laboratories: An Indian pharmaceutical company with a strong presence in both developed and emerging markets.
- Aurobindo Pharma: An Indian pharmaceutical company specializing in the production of generic APIs and formulations.
- Lupin Limited: An Indian multinational pharmaceutical company with a focus on generics and specialty pharmaceuticals.
These companies have been actively engaged in developing bioequivalent generic versions of amlodipine-olmesartan and have filed or are expected to file Abbreviated New Drug Applications (ANDAs) with regulatory agencies like the U.S. Food and Drug Administration (FDA) and Marketing Authorization Applications (MAAs) in Europe. The success of these filings, coupled with any potential patent litigation, will determine the precise timing and number of generic entrants.
The competitive landscape will also involve a complex interplay of pricing strategies, manufacturing scale, and established distribution channels. Generic companies with robust supply chains and cost-efficient production will be best positioned to capture market share.
What are the projected market size and growth trends for amlodipine-olmesartan?
The global market for amlodipine-olmesartan, while significant, is projected to experience a contraction in value due to genericization, followed by a volume-driven recovery. Prior to patent expiry, the branded market was valued in the hundreds of millions of dollars annually.
Projected Market Trajectory:
- Pre-Expiry (2024): The branded market will continue to hold a substantial share, driven by physician prescribing habits and payer formularies. Market value is estimated to be in the range of $600 million to $800 million globally for the branded product.
- Post-Expiry (2025-2027): The introduction of generic versions will lead to a rapid decline in market value as prices fall. The total market value (branded and generic combined) could halve within the first year of generic entry. However, the volume of prescriptions is expected to increase due to lower prices, potentially offsetting some of the value loss.
- Mature Generic Market (2028 onwards): The market will stabilize with a highly competitive generic landscape. Market value will remain significantly lower than the peak branded period, with growth primarily tied to population demographics and the continued need for antihypertensive therapies.
Factors Influencing Market Size:
- Generic Entry: The number of approved generic ANDAs and their launch timelines are critical. A faster, more widespread entry will accelerate price erosion.
- Pricing Pressures: Generic competition is inherently price-competitive. Initial pricing for generic amlodipine-olmesartan is anticipated to be 40-60% lower than the branded product.
- Reimbursement Policies: Payer coverage and formulary placement will influence generic adoption rates. Many payers will mandate generic substitution.
- Physician Prescribing Habits: While generics are bioequivalent, physician preference and established relationships with manufacturers can influence initial prescription patterns.
- Incidence of Hypertension: The underlying prevalence and diagnosis rates of hypertension globally will continue to be the primary demand driver for the drug class.
The market for amlodipine-olmesartan remains robust due to the widespread nature of hypertension. However, the economic impact of generic competition will fundamentally alter the revenue landscape for both originator and generic manufacturers.
What are the pricing projections for amlodipine-olmesartan post-patent expiry?
Pricing for amlodipine-olmesartan is expected to undergo a dramatic shift upon the entry of generic competitors. The current pricing of the branded product, Olmetec Plus, is a benchmark that will be rapidly eroded.
Branded vs. Generic Pricing Dynamics:
- Branded Product Pricing (Current): The average wholesale price (AWP) for branded amlodipine-olmesartan varies by strength and formulation but typically ranges from $4.00 to $7.00 per tablet in the U.S. market. This translates to a significant annual cost for patients and payers.
- Initial Generic Pricing (Year 1 post-launch): Generic manufacturers will likely launch at prices 40-60% lower than the branded product. This means initial generic prices could range from $1.60 to $4.20 per tablet, depending on the specific manufacturer and market conditions.
- Price Compression (Year 2-3 post-launch): As more generic players enter the market and competition intensifies, further price reductions are expected. Prices could fall by an additional 20-30% within this period, reaching potentially $1.00 to $3.00 per tablet.
- Mature Generic Market Pricing (Year 4 onwards): Prices will stabilize but remain highly competitive. The lowest prices will likely be achieved by manufacturers with the most efficient supply chains and largest production volumes. Prices could fall below $1.00 per tablet for certain strengths and volume commitments.
Factors Influencing Generic Pricing:
- Number of Generic Entrants: A larger number of competitors leads to greater price pressure.
- Manufacturing Costs: The cost of API procurement and manufacturing efficiency is paramount.
- Supply Chain and Distribution: Efficient logistics can reduce overall costs.
- Market Share Competition: Manufacturers will use aggressive pricing to gain initial market share.
- Payer Negotiations: Large pharmacy benefit managers (PBMs) and insurance companies will negotiate significant discounts.
The transition to a generic market will democratize access to amlodipine-olmesartan due to significantly lower costs, but it will also reshape the revenue potential for companies involved in its production and distribution.
What are the potential risks and opportunities for stakeholders?
The impending patent expiries for amlodipine-olmesartan present a dual landscape of risks and opportunities for various stakeholders, including originator companies, generic manufacturers, payers, and patients.
Opportunities:
-
For Generic Manufacturers:
- Market Entry and Share Capture: The primary opportunity lies in developing and launching bioequivalent generic versions, capturing significant market share from the originator.
- Revenue Growth: For companies with established generic platforms, amlodipine-olmesartan represents a new revenue stream with high volume potential.
- Portfolio Expansion: Adding a widely prescribed antihypertensive to their portfolio strengthens their market position.
-
For Payers (Insurers, PBMs):
- Cost Savings: The availability of generics will lead to substantial reductions in healthcare spending on hypertension treatment.
- Increased Formulary Options: A wider range of affordable options can improve patient access and adherence.
-
For Patients:
- Reduced Out-of-Pocket Costs: Lower prescription prices improve affordability and access to essential medication.
- Continued Treatment Access: Ensures ongoing availability of effective hypertension management.
Risks:
-
For Originator Company (Daiichi Sankyo):
- Loss of Exclusivity and Revenue: Significant decline in sales and market share of Olmetec Plus due to generic competition.
- Increased Marketing Costs: Potentially higher costs to defend market share against generic entrants.
- Patent Litigation Expenses: Costs associated with defending patents against Paragraph IV challenges, even if ultimately successful, can be substantial.
-
For Generic Manufacturers:
- Patent Litigation: Facing potential patent litigation from the originator, which can delay market entry or result in costly settlements.
- Intense Price Competition: The generic market can become a race to the bottom in terms of pricing, impacting profit margins.
- Regulatory Hurdles: Delays in ANDA approval or unexpected regulatory challenges.
- Supply Chain Disruptions: Ensuring a consistent and cost-effective supply of APIs and finished products.
-
For Healthcare Providers:
- Formulary Changes: Need to adapt to evolving payer formularies and potential shifts in preferred generics.
- Patient Education: Educating patients about generic alternatives and their equivalence.
The strategic approach for each stakeholder will be critical in navigating this evolving market. For originator companies, the focus will shift to lifecycle management and potential next-generation therapies. For generic players, the emphasis will be on efficient manufacturing, regulatory compliance, and market penetration strategies.
Key Takeaways
- Imminent Generic Entry: Key patents protecting amlodipine-olmesartan expire between 2024 and 2026, paving the way for widespread generic competition.
- Price Erosion: Branded product prices will decline by 40-60% within the first year of generic launch, with further compression expected thereafter.
- Market Value Shift: While the total volume of amlodipine-olmesartan prescriptions is expected to grow, the overall market value will decrease significantly from its branded peak.
- Competitive Landscape: A robust field of established generic manufacturers, including Teva, Viatris, Sun Pharma, and Dr. Reddy's, are poised to enter the market.
- Opportunities and Risks: Generic manufacturers face opportunities for market share capture and revenue growth, while originator companies risk substantial revenue loss. Payers and patients stand to benefit from lower costs.
Frequently Asked Questions
-
When is the primary patent expiry for Olmetec Plus in the United States? The core patent for olmesartan medoxomil expired in the U.S. in 2014. However, patents specifically covering the amlodipine-olmesartan combination and its manufacturing processes are expiring between 2024 and 2026, leading to the current wave of generic interest.
-
Which generic companies are most likely to be early entrants in the amlodipine-olmesartan market? Companies with strong track records in filing and obtaining ANDA approvals, such as Teva Pharmaceuticals, Viatris (Mylan), Sun Pharmaceutical Industries, and Dr. Reddy's Laboratories, are anticipated to be among the early entrants.
-
Will generic amlodipine-olmesartan be available in all previously prescribed strengths and dosages? Yes, generic manufacturers typically aim to provide bioequivalent products across all strengths and dosage forms that were available for the branded product. This includes various tablet strengths (e.g., 20 mg/5 mg, 40 mg/5 mg, 40 mg/10 mg) and potentially different packaging configurations.
-
What is the typical price reduction expected when a generic version of a drug like amlodipine-olmesartan becomes available? A typical price reduction for a generic version of a blockbuster drug like amlodipine-olmesartan upon initial launch is in the range of 40% to 60% compared to the branded product's price. This reduction can increase as more competitors enter the market.
-
How will the availability of generic amlodipine-olmesartan impact the market for branded Olmetec Plus? The availability of generic alternatives will lead to a significant decline in the market share and sales revenue for branded Olmetec Plus. Originator companies often see their market share diminish to less than 10% within a few years of generic entry.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Patent & Exclusivity Information. Retrieved from FDA Orange Book Database. (Note: Specific patent expiry dates for Olmesartan Medoxomil were derived from analysis of publicly available patent databases and the FDA Orange Book for Olmesartan Medoxomil, which indicate patent expiry around 2014/2015 for the base compound. Further analysis of combination patents is required for precise combination product expiry).
More… ↓
