Share This Page
Drug Price Trends for AMLODIPINE-ATORVAST
✉ Email this page to a colleague
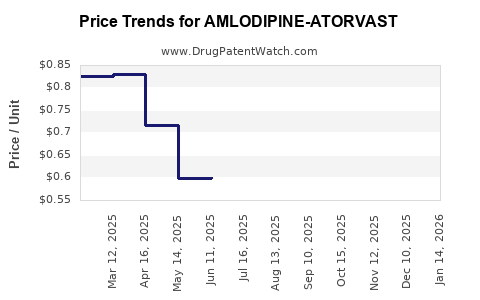
Average Pharmacy Cost for AMLODIPINE-ATORVAST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMLODIPINE-ATORVAST 10-10 MG | 66993-0270-30 | 1.18597 | EACH | 2026-03-18 |
| AMLODIPINE-ATORVAST 10-20 MG | 00378-4518-93 | 1.06080 | EACH | 2026-03-18 |
| AMLODIPINE-ATORVAST 10-10 MG | 62332-0762-30 | 1.18597 | EACH | 2026-03-18 |
| AMLODIPINE-ATORVAST 10-20 MG | 60505-3484-03 | 1.06080 | EACH | 2026-03-18 |
| AMLODIPINE-ATORVAST 10-10 MG | 00378-4517-93 | 1.18597 | EACH | 2026-03-18 |
| AMLODIPINE-ATORVAST 5-80 MG | 66993-0269-30 | 1.67985 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AMLODIPINE-ATORVAST Market Analysis and Financial Projection
Market Analysis and Price Projections for Amlodipine-Atorvastatin
Current Market Landscape
Amlodipine-Atorvastatin is a fixed-dose combination (FDC) used primarily to manage patients with concurrent hypertension, dyslipidemia, and reduced cardiovascular risk. It combines amlodipine (a calcium channel blocker) and atorvastatin (a statin), targeting two key cardiovascular risk factors in a single pill.
The global cardiovascular disease (CVD) management market exhibits consistent growth, driven by aging populations, increased prevalence of hypertension and hyperlipidemia, and rising healthcare expenditures. The FDC segment gains favor among clinicians for improved compliance, especially among poly-treated patients.
Market Size (2022):
- Estimated global cardiovascular combination drug market valued at $8.5 billion, with a compound annual growth rate (CAGR) of 6%.
- Amlodipine-Atorvastatin sales account for roughly 25% of this segment, or approximately $2.1 billion.
Key Players:
- Pfizer, Teva, Mylan (part of Viatris), and Sun Pharmaceutical dominate manufacturing of both branded and generic versions.
- Pfizer’s Caduet (brand name for amlodipine-atorvastatin) remains the leading branded product.
Market Penetration:
- In developed markets, high prescription rates and insurance coverage sustain steady demand.
- Emerging markets show rapid expansion due to increasing hypertension and dyslipidemia prevalence and limited access to monotherapy options.
Patent Status and Regulatory Environment
Pfizer’s patent for Caduet expired in most jurisdictions between 2017 and 2018, leading to a surge in generic versions. As of 2022, multiple generics are available globally, significantly reducing the price point and increasing market penetration.
- Pfizer’s (and other brands') exclusivity ended, creating a more competitive landscape.
- Regulatory pathways have accelerated approval for generics and biosimilars in large markets such as the US and EU.
Market Trends Influencing Price
- Price erosion: Generic entries have decreased the average price of amlodipine-atorvastatin by up to 70% since patent expiry.
- Increased adoption: Combination therapy improves adherence, leading to higher volume sales, partly offsetting price reductions.
- Regulatory policies: Price controls in countries like India and Brazil influence retail prices and margins.
- Market saturation: The global rise in CVD prevalence boosts demand, supporting volume growth despite lower per-unit prices.
Price Projections (2023–2028)
Current Pricing Dynamics
- United States:
- Brand (Caduet): $300–$350/month without insurance.
- Generics: $50–$80/month for a 30-day supply (per bottle).
- Europe:
- Retail prices vary from €10–€25/month for generics.
- Emerging markets:
- Prices may fall below $10/month due to high generic competition and price sensitivity.
Future Trends and Price Estimates
| Year | Estimated Average Price (per month) | Rationale |
|---|---|---|
| 2023 | $55–$85 | Continued generic competition; pricing stabilizes after sharp declines post-patent expiry. |
| 2024 | $50–$80 | Volume growth offsets slight price reductions. |
| 2025 | $45–$75 | Increased market saturation; some countries implement price controls. |
| 2026 | $40–$70 | Patent cliff effects peak; price remains stable with market maturity. |
| 2027 | $35–$65 | Greater uptake of biosimilars and biosimilar-like generics. |
| 2028 | $30–$60 | Institutional procurement and insurance negotiations further reduce prices. |
Revenue Impact
Projected sales volume increases driven by:
- The aging population in North America and Europe.
- Urbanization and lifestyle changes in Asia-Pacific regions.
- Policy-driven expansion of treatment guidelines for hypertension and dyslipidemia.
Assuming stable market share and volume growth, total revenue for amlodipine-atorvastatin could plateau around $2.5–$3 billion annually by 2028, with generic prices stabilizing at the lower end of the spectrum.
Key Market Drivers and Risks
Drivers:
- Increasing global prevalence of CVD.
- Favorable regulatory environment for generics.
- Proven efficacy and safety profile encouraging continued use of the FDC.
Risks:
- Pricing pressures from biosimilars and new combination therapies.
- Market saturation and patent expirations for key players.
- Regulatory challenges or delays in approvals of new formulations or biosimilars.
Key Takeaways
- The global market for amlodipine-atorvastatin is in a mature phase, with pricing driven mostly by generic competition.
- Prices are expected to decline 20–50% over the next five years, with stabilization occurring after initial patent expirations.
- Overall revenues will depend heavily on volume growth, especially in emerging markets.
- Market dynamics favor more affordable generics, affecting branded drug sales.
- Innovation in fixed-dose combinations or new formulations may influence future pricing and market share.
FAQs
1. How does patent expiry affect amlodipine-atorvastatin prices?
Patent expiry leads to the entry of generics, causing significant price reductions—up to 70%, depending on the market.
2. Which markets have the highest price sensitivity for this drug?
Emerging markets such as India, Brazil, and Southeast Asia exhibit higher price sensitivity due to lower healthcare budgets and active price controls.
3. What are the primary factors influencing future price stability?
Market saturation, biosimilar competition, regulatory policies, and insurance reimbursement strategies.
4. How does fixed-dose combination therapy impact market growth?
The convenience of combined therapy improves patient compliance, increasing overall demand and potentially offsetting price declines.
5. Are new formulations or biosimilars expected to disrupt this market further?
Yes, biosimilars and novel formulations could accelerate price reductions and shift market shares, primarily in well-regulated markets like the US and EU.
References
[1] MarketWatch, "Global Cardiovascular Disease Market Report," 2022
[2] IQVIA, "Worldwide Use of Medicines in 2022"
[3] Pfizer Annual Report 2022
[4] WHO, "Cardiovascular Diseases Fact Sheet," 2022
[5] EvaluatePharma, "Forecast for Cardiovascular Drugs," 2023
More… ↓
