Share This Page
Drug Price Trends for AMITRIPTYLINE HCL
✉ Email this page to a colleague
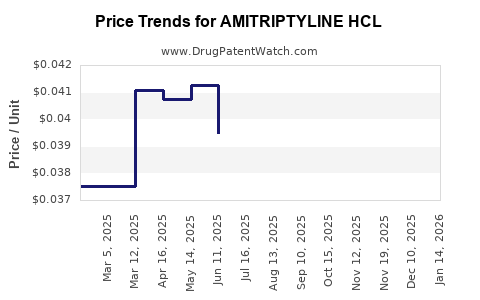
Average Pharmacy Cost for AMITRIPTYLINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMITRIPTYLINE HCL 150 MG TAB | 72888-0072-01 | 0.29208 | EACH | 2026-05-20 |
| AMITRIPTYLINE HCL 100 MG TAB | 72888-0071-01 | 0.16006 | EACH | 2026-05-20 |
| AMITRIPTYLINE HCL 75 MG TAB | 72888-0070-01 | 0.11852 | EACH | 2026-05-20 |
| AMITRIPTYLINE HCL 50 MG TAB | 72888-0069-01 | 0.10191 | EACH | 2026-05-20 |
| AMITRIPTYLINE HCL 50 MG TAB | 72888-0069-00 | 0.10191 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for AMITRIPTYLINE HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| AMITRIPTYLINE HCL 25MG TAB | Golden State Medical Supply, Inc. | 57664-0688-18 | 1000 | 116.05 | 0.11605 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| AMITRIPTYLINE HCL 25MG TAB | Golden State Medical Supply, Inc. | 57664-0688-88 | 100 | 16.67 | 0.16670 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| AMITRIPTYLINE HCL 50MG TAB | Golden State Medical Supply, Inc. | 57664-0689-18 | 1000 | 242.14 | 0.24214 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| AMITRIPTYLINE HCL 50MG TAB | Golden State Medical Supply, Inc. | 57664-0689-88 | 100 | 42.46 | 0.42460 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| AMITRIPTYLINE HCL 100MG TAB | Golden State Medical Supply, Inc. | 57664-0690-88 | 100 | 76.97 | 0.76970 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
AMITRIPTYLINE HCL: PATENT LANDSCAPE AND MARKET PROJECTIONS
Amitriptyline HCl, a tricyclic antidepressant (TCA), faces a mature generic market with established pricing and limited patent exclusivities remaining for its original formulations. The primary drivers for market activity are generic competition, demand for treatment of major depressive disorder and neuropathic pain, and potential for combination therapies. Price projections indicate continued stability with marginal fluctuations driven by supply chain dynamics and generic manufacturer competition.
What is Amitriptyline HCl and its Primary Indications?
Amitriptyline HCl is a well-established medication primarily prescribed for the treatment of major depressive disorder. Its mechanism of action involves inhibiting the reuptake of neurotransmitters like serotonin and norepinephrine in the brain. Beyond depression, it is also widely used to manage various types of chronic pain, particularly neuropathic pain conditions such as diabetic neuropathy, postherpetic neuralgia, and fibromyalgia. Off-label uses include treatment for insomnia, migraine prophylaxis, and irritable bowel syndrome.
What is the Patent Status of Amitriptyline HCl?
Original patents for amitriptyline HCl have long expired. The compound was first patented by Merck & Co. in the late 1950s, with key composition of matter patents lapsing decades ago. Current patent activity primarily centers on specific formulations, delivery methods, or combination products. For instance, patents might exist for controlled-release formulations, specific salt forms, or novel synergistic combinations with other active pharmaceutical ingredients. However, these are unlikely to grant significant market exclusivity for the base compound given its generic status.
A search of the United States Patent and Trademark Office (USPTO) database reveals numerous patents related to amitriptyline, but the vast majority pertain to incremental innovations rather than the core molecule. For example, patents might cover:
- Formulation Patents: Devices or methods for sustained or delayed release of amitriptyline.
- Combination Patents: Claims on pharmaceutical compositions containing amitriptyline and one or more other active pharmaceutical ingredients for specific therapeutic indications.
- Manufacturing Process Patents: Novel or improved methods for synthesizing amitriptyline HCl.
As of late 2023, no primary composition of matter patents for amitriptyline HCl remain in force that would grant broad market exclusivity in major global markets like the United States or the European Union. The drug has been available as a generic for many years.
Who are the Key Manufacturers and Suppliers of Amitriptyline HCl?
The manufacturing landscape for amitriptyline HCl is dominated by generic pharmaceutical companies. Due to the long-standing patent expiry, production is widespread, with numerous companies holding Abbreviated New Drug Applications (ANDAs) approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA).
Key generic manufacturers and suppliers globally include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Sandoz International GmbH (a division of Novartis)
- Lupin Ltd.
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Amneal Pharmaceuticals LLC
- Hikma Pharmaceuticals PLC
These companies produce both the active pharmaceutical ingredient (API) and the finished dosage forms. The global supply chain involves API manufacturers, often based in India and China, supplying to finished dosage form manufacturers worldwide.
What is the Current Market Size and Demand for Amitriptyline HCl?
The market size for amitriptyline HCl is substantial but mature, driven by its established efficacy and cost-effectiveness as a treatment option. Precise global market figures for individual generic drugs are often proprietary and difficult to isolate definitively from broader antidepressant or pain management market data. However, industry reports and market analysis for the antidepressant and neuropathic pain segments provide an indication.
The global antidepressant market was valued at approximately USD 15.6 billion in 2022 and is projected to grow at a CAGR of 3.5% from 2023 to 2030, according to various market research firms. While amitriptyline HCl constitutes only a fraction of this, its consistent use in both depression and pain management sustains demand.
Demand is influenced by:
- Prevalence of Depression: Major depressive disorder affects millions globally.
- Incidence of Neuropathic Pain: Conditions like diabetic neuropathy are on the rise due to increasing rates of diabetes.
- Prescribing Practices: Clinicians continue to prescribe amitriptyline HCl due to its proven track record and low cost, especially when newer, more expensive options are not tolerated or are unaffordable.
- Formulation Preferences: Availability of immediate-release and sometimes specialized formulations influences usage.
Estimates suggest the global amitriptyline HCl market, encompassing both API and finished dosage forms, likely falls within the hundreds of millions of U.S. dollars annually, but this figure is subject to considerable variation based on the source and scope of analysis.
What are the Price Trends and Projections for Amitriptyline HCl?
The pricing of amitriptyline HCl is characteristic of a mature generic drug market: highly competitive and relatively low. Prices are primarily driven by generic competition, manufacturing costs, and volume purchasing by large pharmacy chains and healthcare systems.
Historical Price Trends:
- Post-Patent Expiry: Prices declined sharply after generic entry, reaching low single-digit dollar amounts per prescription for standard strengths.
- Recent Stability: For the past decade, prices have remained relatively stable, with minor fluctuations. The average wholesale price (AWP) for a 30-day supply of 25mg tablets typically ranges from $10 to $25, depending on the manufacturer and pharmacy.
- API Costs: API prices are influenced by raw material costs, manufacturing efficiency, and geopolitical factors affecting supply chains.
Projected Price Trends:
- Continued Stability: Projections indicate continued price stability for amitriptyline HCl. The drug is unlikely to experience significant price increases due to the absence of patent protection and the presence of numerous suppliers.
- Marginal Declines: In highly competitive markets, prices may see marginal declines of 1-3% annually as manufacturers compete for market share through volume discounts.
- Supply Chain Volatility: Short-term price spikes are possible due to disruptions in raw material sourcing, manufacturing, or transportation. However, these are typically temporary as alternative suppliers can often be found.
- Demand Shifts: While overall demand is expected to remain steady or grow modestly with population increases and rising chronic pain conditions, significant price shifts are not anticipated unless a major new therapeutic application or a disruptive manufacturing technology emerges.
Factors influencing price:
- Number of Generic Manufacturers: A higher number of ANDA holders generally leads to lower prices.
- Raw Material Costs: Fluctuations in the cost of precursors for API synthesis.
- Regulatory Compliance Costs: Adherence to Good Manufacturing Practices (GMP) and other regulatory requirements.
- Volume Discounts: Large-scale purchasing by hospital networks and pharmacy benefit managers (PBMs).
The average prescription price for a generic drug like amitriptyline HCl is often in the range of $5 to $15, particularly when covered by insurance or discount cards. Out-of-pocket costs for uninsured individuals can be higher but remain significantly lower than brand-name drugs.
What is the Competitive Landscape for Amitriptyline HCl?
The competitive landscape for amitriptyline HCl is characterized by:
- Intense Generic Competition: As a first-generation TCA, amitriptyline HCl has been off-patent for decades. This has led to a crowded generic market with numerous manufacturers vying for market share. Pricing is highly sensitive to competitive pressures.
- Therapeutic Alternatives:
- For Depression: Newer classes of antidepressants, such as Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs), have largely replaced TCAs as first-line treatments for many patients due to better tolerability profiles and fewer anticholinergic side effects. Examples include fluoxetine, sertraline, escitalopram (SSRIs), and venlafaxine, duloxetine (SNRIs).
- For Neuropathic Pain: A range of medications are used, including gabapentinoids (gabapentin, pregabalin), other antidepressants (duloxetine, venlafaxine), and topical treatments (lidocaine patches, capsaicin).
- Combination Products: While not prevalent for amitriptyline HCl itself as a new combination, it can be prescribed alongside other medications. The market for new fixed-dose combination therapies with other established drugs for conditions like depression or pain is a competitive space, but amitriptyline HCl's primary role remains as a monotherapy or as an add-on for specific patient profiles.
- Cost-Effectiveness: Amitriptyline HCl remains a cost-effective option, particularly for patients who cannot tolerate newer agents or for whom cost is a significant barrier. This sustains its market share in specific patient segments and geographies.
- Specialty Formulations: While the base product is generic, there may be niche competition for specific controlled-release or enhanced delivery formulations, though these represent a smaller portion of the overall market.
The competitive environment favors manufacturers with efficient production processes and strong distribution networks capable of delivering high volumes at low margins.
What are the Regulatory Considerations for Amitriptyline HCl?
Amitriptyline HCl is subject to standard pharmaceutical regulations worldwide. Key regulatory considerations include:
- Drug Approval: Manufacturers require marketing authorization from national regulatory bodies, such as the FDA in the United States, the European Medicines Agency (EMA) in the European Union, and Health Canada in Canada. For generic versions, this typically involves submitting an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence to the reference listed drug.
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to stringent GMP standards to ensure product quality, safety, and efficacy. Regular inspections by regulatory authorities are conducted.
- Labeling and Packaging: Prescribing information, patient information leaflets, and packaging must comply with regulatory requirements, including warnings, contraindications, and dosage instructions. This is particularly important for amitriptyline HCl due to its potential side effects (e.g., anticholinergic effects, cardiac risks, suicide ideation warnings).
- Pharmacovigilance: Post-market surveillance systems are in place to monitor adverse events and ensure ongoing product safety. Manufacturers are responsible for reporting serious adverse events to regulatory agencies.
- Controlled Substances and Scheduling: Amitriptyline HCl is not a controlled substance in most jurisdictions and does not carry the same regulatory burden as opioid analgesics or psychotropic drugs with a high potential for abuse.
- Import/Export Regulations: International trade in amitriptyline HCl is subject to import and export regulations, including customs requirements and product registration in the importing country.
The lack of patent exclusivity means that regulatory approval for generic versions is the primary barrier to market entry, alongside the ability to meet manufacturing and quality standards.
What are the Future Outlook and Opportunities for Amitriptyline HCl?
The future outlook for amitriptyline HCl is one of steady, albeit unexciting, demand. Significant growth is unlikely due to its mature status and the availability of newer alternatives. However, specific opportunities exist:
- Emerging Markets: As healthcare access expands in developing countries, the demand for affordable and effective medications like amitriptyline HCl will likely increase.
- Neuropathic Pain Management: The growing prevalence of diabetes and other chronic conditions leading to neuropathic pain ensures continued demand for effective pain management strategies. Amitriptyline HCl's role as a second-line or adjunct therapy in this area remains secure.
- Cost-Containment Strategies: Healthcare systems globally are focused on cost containment. Amitriptyline HCl, being one of the most cost-effective treatments for depression and neuropathic pain, will continue to be favored in formulary decisions and public health programs.
- Combination Therapy Research: While not currently a focus for new patentable combinations involving amitriptyline HCl, ongoing research into synergistic effects for complex pain syndromes or treatment-resistant depression could theoretically lead to new therapeutic approaches, though patentability would be a challenge for the base molecule.
- Supply Chain Resilience: Manufacturers that can demonstrate robust and resilient supply chains, particularly for APIs, may gain a competitive edge. The COVID-19 pandemic highlighted the vulnerabilities in global pharmaceutical supply chains, and reliability is becoming an increasingly important factor for purchasers.
The primary opportunity lies in maintaining cost-effective, high-quality production and reliable distribution to meet consistent global demand, rather than through novel therapeutic applications or market exclusivity.
Key Takeaways
- Amitriptyline HCl has long-standing expired composition of matter patents, placing it in a mature generic market.
- The drug remains a cost-effective treatment for major depressive disorder and neuropathic pain, sustaining consistent demand.
- Pricing is highly competitive, characterized by low single-digit dollar values per prescription, with projections indicating continued stability or marginal declines.
- Key manufacturers are generic pharmaceutical giants with established global supply chains.
- Regulatory focus is on GMP compliance, pharmacovigilance, and accurate labeling, rather than on granting new market exclusivity.
- Future opportunities are primarily in emerging markets and its role in cost-containment strategies, rather than through innovation or new patentable claims.
Frequently Asked Questions
What is the difference in efficacy between amitriptyline HCl and newer antidepressants like SSRIs?
While SSRIs are often preferred as first-line agents due to a better side effect profile, amitriptyline HCl demonstrates comparable efficacy for many patients with moderate to severe depression. Its effectiveness in neuropathic pain is also well-established and can be superior to some SSRIs for certain pain types.
Are there any specific risks associated with long-term use of amitriptyline HCl?
Long-term use can be associated with anticholinergic side effects (dry mouth, constipation, blurred vision, urinary retention), weight gain, and sedation. Cardiovascular effects and potential for overdose are also significant risks, necessitating careful patient selection and monitoring.
What is the typical dosage range for amitriptyline HCl in treating depression versus neuropathic pain?
For depression, typical starting doses range from 25-50 mg per day, gradually increased to 75-150 mg, and sometimes up to 300 mg per day for severe cases. For neuropathic pain, lower doses are often effective, starting at 10-25 mg per day and titrating upwards based on response and tolerability, typically to a maximum of 50-100 mg per day.
How does amitriptyline HCl's cost-effectiveness compare to alternative pain management options?
Amitriptyline HCl is significantly more cost-effective than many newer pain management drugs, including gabapentinoids and certain specialized therapies. Its low manufacturing cost and widespread generic availability make it an attractive option for public health systems and uninsured patients.
What are the implications of increased global competition on API sourcing for amitriptyline HCl?
Increased competition among API manufacturers, particularly from regions like India and China, can drive down API costs. However, it also necessitates rigorous quality control and supply chain management to ensure consistent quality and avoid reliance on single, potentially vulnerable, sources.
Citations
[1] Grand View Research. (2023). Antidepressant Drugs Market Size, Share & Trends Analysis Report By Drug Class, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030.
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[3] U.S. Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. Retrieved from https://patft.uspto.gov/
More… ↓
