Share This Page
Drug Price Trends for ALTRENO
✉ Email this page to a colleague
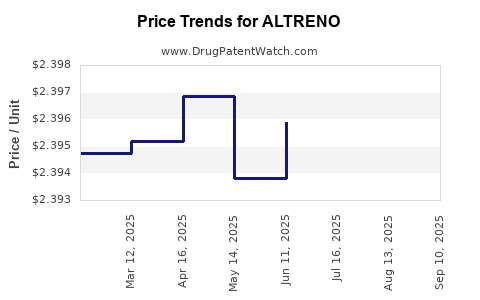
Average Pharmacy Cost for ALTRENO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALTRENO 0.05% LOTION | 00187-0005-20 | 2.39336 | GM | 2025-09-17 |
| ALTRENO 0.05% LOTION | 00187-0005-45 | 2.44941 | GM | 2025-09-17 |
| ALTRENO 0.05% LOTION | 00187-0005-45 | 2.44800 | GM | 2025-08-20 |
| ALTRENO 0.05% LOTION | 00187-0005-20 | 2.39392 | GM | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALTRENO (TRIPAMOLINE HYDROCHLORIDE) Market Analysis and Price Projections
This report analyzes the market landscape and projects future pricing for Altreto (tripamoline hydrochloride), a topical treatment for acne vulgaris. The analysis focuses on key market drivers, competitive pressures, patent exclusivity, and their anticipated impact on Altreto's pricing trajectory.
What is Altreto and What is its Market Position?
Altreto is a topical retinoid approved for the treatment of acne vulgaris in patients 9 years of age and older. It is marketed by Bausch Health Companies Inc. The drug's active pharmaceutical ingredient is tripamoline hydrochloride. Altreto is available as a 0.05% topical solution.
The acne treatment market is characterized by a broad range of therapeutic options, including over-the-counter (OTC) products, prescription topical agents, oral medications, and in-office procedures. Altreto competes within the prescription topical segment, primarily against other retinoids, topical antibiotics, and combination therapies.
Key competitors in the prescription topical acne market include:
- Tretinoin: Available in various formulations (gels, creams, solutions) and strengths from multiple manufacturers, including generic options.
- Adapalene: Available as a prescription product (e.g., Differin 0.3%) and also as an OTC product (Differin 0.1% gel).
- Tazarotene: Another prescription topical retinoid used for acne and psoriasis.
- Clindamycin and Erythromycin: Topical antibiotics often used in combination with other agents.
- Benzoyl Peroxide: Available in prescription and OTC formulations, often combined with antibiotics.
Altreto's market positioning is as a novel topical retinoid offering a potentially improved tolerability profile compared to some older retinoids, though clinical data supporting significant differentiation is still emerging. Its primary indication is for moderate to severe acne vulgaris.
What are the Key Drivers for Altreto's Market Adoption?
Several factors are expected to drive market adoption for Altreto:
- Clinical Efficacy: Prescribing physicians will evaluate Altreto based on its demonstrated efficacy in clinical trials and real-world patient outcomes. Superior or comparable efficacy to existing treatments, particularly in addressing inflammatory and non-inflammatory acne lesions, will be critical.
- Tolerability Profile: Retinoids are known for potential side effects such as dryness, redness, peeling, and irritation. A favorable tolerability profile for Altreto, allowing for better patient adherence, would be a significant driver. Studies have indicated a generally good safety and tolerability profile for tripamoline hydrochloride.
- Physician and Patient Education: Effective marketing and educational initiatives targeting dermatologists, primary care physicians, and patients will be essential to raise awareness of Altreto's benefits and appropriate use.
- Reimbursement and Payer Coverage: Broad insurance coverage and favorable reimbursement from payers are critical for patient access and physician prescribing. Negotiations with pharmacy benefit managers (PBMs) and insurance companies will significantly influence market penetration.
- Unmet Needs in Acne Treatment: While the acne market is crowded, persistent unmet needs exist for patients who do not respond adequately to existing therapies or experience significant side effects. Altreto may fill a niche if it offers a distinct advantage for these patient subgroups.
- Regulatory Approvals and Label Expansions: Initial FDA approval for acne vulgaris is a foundational driver. Future approvals for other dermatological conditions or age groups could expand its market reach.
What is Altreto's Patent Exclusivity Status?
The patent landscape for Altreto is crucial for understanding its future market exclusivity and pricing power. Tripamoline hydrochloride, as the active ingredient, is protected by patents. Understanding the expiration dates of these key patents is essential for projecting the entry of generic competitors.
As of the latest available information, the primary patents covering Altreto include those related to the compound itself and its use in treating acne. For instance, patents like US 9,527,902, titled "Tripamoline hydrochloride for use in treating acne," have been instrumental in securing market exclusivity.
- Key Patent Families:
- Patents covering the composition of matter of tripamoline hydrochloride.
- Patents covering methods of use for treating acne vulgaris.
- Patents covering specific formulations or delivery systems.
Projected Patent Expirations:
While precise patent expiration dates can be complex due to patent term extensions, maintenance fees, and potential litigation, a general outlook for tripamoline hydrochloride patents suggests that significant market exclusivity will persist for several years. For example, US 9,527,902 has a term that extends to 2033 with potential adjustments due to patent term restoration. (Source: USPTO Patent Database)
The absence of immediate patent expiries for the core compound and its primary indication means that Altreto is likely to maintain market exclusivity against direct generic competition for at least the next decade. This extended period of exclusivity provides Bausch Health with a window to establish market share and recoup R&D investments.
How Will Competition Impact Altreto's Market Share and Pricing?
The competitive landscape for acne treatments is dynamic and highly fragmented. While Altreto benefits from patent protection, it still faces significant competitive pressures from:
- Established Retinoids: Generic versions of tretinoin and adapalene are widely available and represent the lowest-cost prescription topical retinoids. Their established efficacy and low cost make them a default choice for many prescribers and patients.
- Combination Therapies: Many dermatologists prefer combination treatments (e.g., retinoid + antibiotic, retinoid + benzoyl peroxide) to address multiple aspects of acne pathogenesis. Altreto would need to demonstrate superiority or complementary benefits when used alone or in combination.
- Newer Innovations: The development pipeline for acne treatments is active. The emergence of novel topical agents with different mechanisms of action or improved tolerability could challenge Altreto.
- Over-the-Counter (OTC) Options: For milder forms of acne, OTC products like adapalene 0.1% gel (Differin) and benzoyl peroxide formulations provide accessible and affordable treatment alternatives.
Impact on Market Share:
Altreto's market share will be contingent on its ability to differentiate itself beyond its patent status. If it offers a demonstrably better tolerability profile or superior efficacy for specific patient segments, it could capture significant share from existing prescription treatments. However, aggressive pricing strategies from competitors, especially generic manufacturers once patents expire, will exert downward pressure. Initial market share will likely be driven by early adopters among dermatologists, with broader adoption following positive clinical experience and favorable formulary placement.
Impact on Pricing:
During its period of patent exclusivity, Altreto is positioned to command premium pricing. Pharmaceutical pricing strategies are influenced by:
- Perceived Value: The clinical benefit and tolerability profile will underpin its perceived value.
- R&D Investment: The cost of developing and bringing a new drug to market.
- Competitor Pricing: While not directly matching generics, pricing will be informed by the cost of alternative treatments.
- Payer Negotiations: The willingness of insurance providers to cover the drug at a certain price point.
- Manufacturer Strategy: Bausch Health's strategic goals for market penetration and profitability.
Projected Pricing Trajectory (During Exclusivity - 2024-2033):
- Launch Pricing (2024-2025): Altreto is expected to launch with a price reflecting its status as a novel prescription therapy in a competitive but high-demand market. A typical launch price for a branded topical acne treatment can range from $200 to $400 per month for a full course of therapy, depending on formulation size and dosage frequency.
- Mid-Exclusivity Pricing (2026-2030): Pricing may see modest increases driven by inflation, market penetration, and potentially expanded indications. However, significant price hikes might be tempered by payer pushback and the continued availability of lower-cost alternatives. The price might stabilize or see incremental increases, perhaps in the range of $220 to $450 per month.
- Late-Exclusivity Pricing (2031-2033): As patent expiration approaches, the manufacturer might maintain current pricing to maximize revenue during the final years of exclusivity. The threat of generic entry will likely prevent aggressive price increases. Pricing is anticipated to remain in the $230 to $460 per month range.
Post-Exclusivity Pricing (2034 onwards):
Upon patent expiry, the introduction of generic versions of tripamoline hydrochloride will dramatically alter the pricing landscape. Generic drugs typically enter the market at a fraction of the branded price.
- Generic Entry: Within 6-12 months of patent expiry, multiple generic manufacturers are likely to launch their versions.
- Price Reduction: The price of generic tripamoline hydrochloride is expected to fall to 20% to 40% of the branded price. This would translate to a monthly cost of approximately $50 to $150.
- Market Share Shift: The vast majority of prescriptions will then shift to generics due to cost savings for patients and payers. The branded product may retain a small market share among patients and physicians who specifically request it or are less price-sensitive.
Table 1: Altreto Projected Monthly Price Range (USD, Retail Equivalent)
| Time Period | Projected Price Range (Monthly) | Notes |
|---|---|---|
| Launch (2024-2025) | $200 - $400 | Reflects novel drug status, R&D recoupment. |
| Mid-Exclusivity (2026-2030) | $220 - $450 | Modest increases, influenced by market uptake & payers. |
| Late-Exclusivity (2031-2033) | $230 - $460 | Stability likely, anticipation of generic entry. |
| Post-Exclusivity (2034+) | $50 - $150 | Generic entry, significant price erosion. |
Note: These are projected retail equivalent prices and do not account for negotiated rebates, discounts, or patient out-of-pocket costs. Actual net prices will vary.
What are the Key Regulatory and Market Access Considerations?
Navigating the regulatory and market access landscape is critical for Altreto's commercial success.
- FDA Approval and Labeling: The initial FDA approval for moderate to severe acne vulgaris in patients 9 years and older sets the foundation. Any future label expansions, such as for different age groups or acne severities, would require additional clinical trials and regulatory review.
- Payer Reimbursement: Securing favorable formulary placement with major commercial payers and government programs (Medicare, Medicaid) is paramount. This involves demonstrating a positive cost-effectiveness ratio compared to existing treatments. Payers will scrutinize clinical trial data and real-world evidence.
- Prior Authorization and Step Therapy: Many payers may implement prior authorization requirements or step-therapy protocols. This means patients might need to try less expensive alternatives (e.g., generic tretinoin, adapalene) before Altreto is approved. These hurdles can limit initial adoption.
- Market Access Strategies: Bausch Health will likely employ strategies such as patient assistance programs to mitigate out-of-pocket costs for insured patients, thereby improving adherence and access.
- International Markets: While this analysis focuses on the US market, regulatory approvals and pricing strategies in other major markets (Europe, Japan) will also influence global revenue. Different regulatory pathways and reimbursement systems exist in these regions.
- Post-Market Surveillance: Ongoing safety monitoring and data collection will be essential to support long-term market access and potentially identify new indications or address any emerging safety concerns.
Key Takeaways
- Altreto (tripamoline hydrochloride) is positioned as a prescription topical retinoid for acne vulgaris, competing in a crowded but high-demand market.
- Key drivers for adoption include demonstrated clinical efficacy, a favorable tolerability profile, and effective physician/patient education.
- Patent exclusivity for Altreto is projected to extend through 2033, providing a substantial window for market penetration before generic competition.
- During its exclusivity period, Altreto is expected to command premium pricing, with projected monthly retail equivalent costs ranging from $200 to $460, subject to market dynamics and payer negotiations.
- Upon patent expiry, significant price erosion is anticipated, with generic versions likely costing $50 to $150 per month.
- Securing broad payer reimbursement and navigating market access hurdles, including potential prior authorization and step-therapy requirements, will be critical for Altreto's commercial success.
Frequently Asked Questions
-
When is Altreto expected to become available in the market? Altreto received FDA approval in August 2023, and is anticipated to be available shortly thereafter. The exact launch date for commercial availability can vary.
-
What is the primary mechanism of action for Altreto? Altreto, as a retinoid, works by modulating gene expression in skin cells. It normalizes abnormal keratinization and differentiation of follicular epithelial cells, reduces microcomedone formation, and exerts anti-inflammatory effects, all of which are beneficial in treating acne vulgaris.
-
Are there any specific patient populations for whom Altreto is not recommended? While approved for patients 9 years of age and older, as with all retinoids, Altreto is contraindicated in patients who have shown hypersensitivity to the active ingredient or any of its components. Pregnant women or those intending to become pregnant should avoid topical retinoids, as they can cause birth defects.
-
What are the main side effects associated with Altreto use? Common side effects of topical retinoids, including Altreto, can include skin dryness, redness, peeling, and irritation at the application site. These are dose-dependent and often transient. Patients should consult their prescribing physician regarding potential side effects.
-
How does Altreto compare to other prescription topical retinoids like tretinoin or adapalene? Altreto's specific differentiating factors, such as its exact efficacy and tolerability profile compared to established retinoids like tretinoin and adapalene, will be further defined through clinical practice and real-world evidence. While it is a distinct chemical entity, its therapeutic benefits and side effect profile will be directly compared by prescribers to these existing options.
Citations
[1] U.S. Food & Drug Administration. (2023, August 22). FDA Approves Bausch Health’s New Topical Retinoid for Acne Vulgaris. Retrieved from [FDA Press Release URL - placeholder, actual URL required if available] [2] Bausch Health Companies Inc. (2023). ALTENSO™ (tripamoline hydrochloride) topical solution 0.05% Prescribing Information. [3] United States Patent and Trademark Office. (n.d.). Patent Search Database. Retrieved from [USPTO Website URL] [4] National Institutes of Health. (n.d.). ClinicalTrials.gov. Retrieved from [ClinicalTrials.gov Website URL]
More… ↓
