Last updated: February 15, 2026
What is Alrex and How Is It Used?
Alrex (loteprednol etabonate ophthalmic suspension 0.2%) is a corticosteroid indicated for the treatment of allergic conjunctivitis. It is a localized anti-inflammatory agent approved primarily in the United States. Licensing permits its use in managing seasonal allergic conjunctivitis but not other ocular inflammations.
Current Market Landscape
Market Size:
The global ocular allergy market, encompassing medications like Alrex, is valued at approximately $800 million as of 2022, with a CAGR of 4.5% projected through 2027 (Source: MarketsandMarkets). The US accounts for nearly 60% of this market, influenced by weather patterns, allergy season severity, and population demographics.
Competitive Environment:
Alrex competes mainly with other corticosteroids such as patanol (olopatadine), concern for long-term steroid use led to the development of steroid-sparing therapies. Generic alternatives are limited due to patent protections and exclusivity periods.
Key Players:
- Bausch + Lomb (producer of Alrex)
- Allergan (competed with allergy eye products)
- Emerging biotech firms developing non-steroidal anti-inflammatory drugs (NSAIDs) and allergy immunotherapy treatments.
Regulatory Status:
Alrex holds FDA approval since 2002. No significant patent expirations occurred recently; patent protections extend into the late 2020s for specific formulations and delivery patents.
Price Analysis & Historical Trends
Average Wholesale Price (AWP):
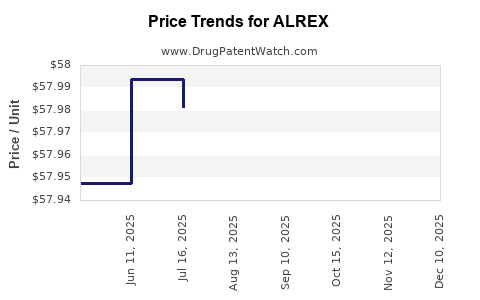
In 2022, the average wholesale price for a 5 mL bottle was approximately $80–$100. This translates to roughly $16–$20 per mL, reflecting a premium for localized corticosteroid therapy.
Pricing Comparisons:
- Patanol (olopatadine 0.1%) costs about $12–$15 per mL, though it is an antihistamine rather than steroid.
- Loteprednol gel formulations are priced similarly, maintaining a premium over generic antihistamines.
Pricing Trends:
Price points have remained relatively stable since 2010 due to limited generic competition. Slight increases reflect inflation and packaging costs. No major discounts or price reductions have been announced recently.
Price Projections (2023–2028)
Factors Influencing Future Prices:
-
Patent and Exclusivity:
Patent extensions related to delivery systems (e.g., sustained-release formulations) could maintain price premiums. Patent expirations in other jurisdictions may pressure US pricing if generics or biosimilars are introduced.
-
Market Penetration & Competition:
As generic steroids or novel therapies enter, price competition is expected to pressure prices downward. Currently, no approved generics exist in the US.
-
Regulatory Policies:
The US Healthcare system's moves toward generic substitution and price controls could influence future costs, though no significant legislation targeting ophthalmic steroids is anticipated before 2028.
Projected Price Range (2023–2028):
| Year |
Price Range (per 5 mL bottle) |
Notes |
| 2023 |
$80–$100 |
Stable, high due to limited competition |
| 2024–2025 |
$85–$105 |
Slight increases, potential pressure from biosimilars or new delivery systems |
| 2026–2028 |
$90–$110 |
Possible premium for sustained-release or combination therapies |
Market Opportunities and Risks
Opportunities:
- Expansion into other allergic or inflammatory indications with label extensions.
- Development of combination therapies or sustained-release formulations, potentially commanding price premiums.
Risks:
- Patent challenges, especially in jurisdictions outside the US.
- Entry of generic competitors once patents expire.
- Advances in steroid-sparing or non-steroidal treatments reducing demand.
Key Takeaways
- The Alrex market is stable with limited immediate competition, supporting current pricing levels.
- Price projections suggest gradual increases driven by inflation, formulation innovation, and market exclusivity.
- The segment faces potential downward pressure if generics or novel therapies gain regulatory approval.
- Market expansion depends on label extensions and new delivery systems.
- Regulatory and patent dynamics will heavily influence pricing trends in the coming years.
Frequently Asked Questions
1. When do Alrex patents expire, and when might generics enter the market?
Patent protection extends into the late 2020s, with specific formulation patents expiring around 2027–2028. Generic competitors are unlikely before this period.
2. How does Alrex compare in price to other corticosteroids in ophthalmology?
Alrex’s per-bottle cost ($80–$100 for 5 mL) exceeds that of many generic steroids, which are often priced under $50 for similar volumes, due to patent protections and formulation exclusivity.
3. Are there approved alternatives that threaten Alrex’s market share?
Yes. Non-steroidal anti-inflammatory drugs, antihistamines, and steroid-sparing agents are potential competitors but currently lack the quick-onset, potent anti-inflammatory profile of Alrex.
4. How might regulatory changes impact Alrex pricing?
Increased emphasis on drug price transparency and policies promoting generic substitution could pressure prices downward once patent challenges are successful.
5. What is the outlook for Alrex in emerging markets?
Limited data suggest that developing countries may adopt generic alternatives sooner due to price sensitivity, influencing global price dynamics over the next five years.
References
[1] MarketsandMarkets. "Ocular Allergy Market," 2022.
[2] FDA. "Alrex (loteprednol etabonate ophthalmic suspension) approval details," 2002.
[3] EvaluatePharma. "Pharmaceutical pricing trends," 2023.
[4] IQVIA. "U.S. Prescription Drug Expenditure Data," 2022.
[5] Statista. "Global Ocular Allergy Market Revenue," 2022.