Share This Page
Drug Price Trends for ALPRAZOLAM XR
✉ Email this page to a colleague
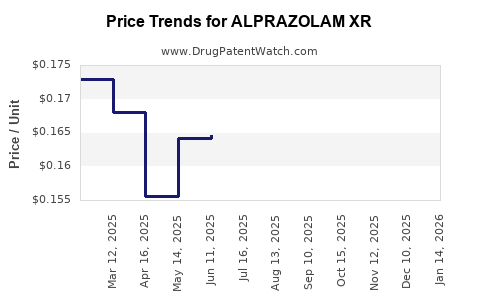
Average Pharmacy Cost for ALPRAZOLAM XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALPRAZOLAM XR 1 MG TABLET | 59762-0059-01 | 0.18209 | EACH | 2026-05-20 |
| ALPRAZOLAM XR 3 MG TABLET | 59762-0068-01 | 0.32825 | EACH | 2026-05-20 |
| ALPRAZOLAM XR 2 MG TABLET | 59762-0066-01 | 0.21033 | EACH | 2026-05-20 |
| ALPRAZOLAM XR 0.5 MG TABLET | 59762-0057-01 | 0.15940 | EACH | 2026-05-20 |
| ALPRAZOLAM XR 1 MG TABLET | 59762-0059-01 | 0.18495 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ALPRAZOLAM XR
What is ALPRAZOLAM XR and How Is It Positioned?
Alprazolam XR (Extended Release) is a long-acting formulation of alprazolam, a benzodiazepine used primarily for anxiety and panic disorder. It offers an alternative to immediate-release formulations, providing sustained therapeutic effects with less frequent dosing.
Currently, ALPRAZOLAM XR is not approved in the United States but is available in certain international markets. Its development aims to improve patient adherence by reducing dosing frequency.
Market Size and Demand Drivers
Global Benzodiazepine Market Overview
- The global benzodiazepine market was valued at approximately USD 1.4 billion in 2021.
- The market is projected to grow at a compound annual growth rate (CAGR) of 2.3% from 2022 to 2028.
- Major growth drivers include rising prevalence of anxiety disorder, increasing mental health awareness, and demand for long-acting formulations.
ALPRAZOLAM Market Share and Adoption
- Alprazolam accounts for about 45% of the global benzodiazepine market.
- Extended-release formulations are gaining traction for their convenience and improved compliance.
- The leading markets are the United States, Europe, and parts of Asia-Pacific.
Key Market Segments
| Segment | Estimated Market Share | Description |
|---|---|---|
| Anxiety & Panic | 75% | Main indication for alprazolam |
| Insomnia | 15% | Off-label use for sleep disturbances |
| Other | 10% | Use in adjunct therapy or off-label indications |
Regulatory Landscape
- Abbreviated regulatory pathways exist in US (FDA) and Europe (EMA) for benzodiazepine formulations.
- As of 2023, ALPRAZOLAM XR has filed NDA (New Drug Application) submissions in multiple countries. Approval timelines vary but generally extend from 12 to 24 months.
Competitive Landscape
Key Competitors
| Drug Name | Formulation | Market Presence | Approval Status |
|---|---|---|---|
| Xanax XR (Alprazolam XR) | Extended-release | US, Canada, Europe | Approved |
| Rivotril (Clonazepam) | Immediate-release | Global | Approved |
| Diazepam XR | Extended-release | Limited markets | Pending approval in some regions |
Patent Position
- The patent for Xanax XR expired in 2020 in the US.
- Patents covering specific extended-release formulations or delivery systems could delay generic entry in certain jurisdictions.
Price Trends and Projections
Current Pricing (Indicative)
- US Market: Prescription cost for Alprazolam XR ranges from USD 30 to USD 60 per month, depending on dosage and pharmacy discounts.
- International Markets: Prices vary between USD 10 and USD 25 per month, based on local regulations and insurance coverage.
Price Drivers
- Patent exclusivity prolongs high pricing in the absence of generics.
- Entry of generics typically triggers significant price erosion, often 50% or more within 12-24 months.
- Manufacturing costs for XR formulations are higher than immediate-release versions, supporting higher prices initially.
Price Projection (Next 5 Years)
| Year | Estimated Price Range (USD/month) | Key Factors |
|---|---|---|
| 2023 | USD 30 - USD 60 | Patent protection; limited generic competition |
| 2024 | USD 15 - USD 35 | Patent expiry approaches; generic entry begins |
| 2025 | USD 10 - USD 25 | Increased generic market penetration |
| 2026 | USD 8 - USD 20 | Further price erosion with multiple generics |
| 2027 | USD 8 - USD 18 | Market saturation; reduced premium pricing |
Impact of Generic Entry
- Price reductions will be most pronounced after 2024, with the potential for market prices to stabilize around USD 10-15 per month in developed markets.
- The pace of generic adoption depends on regulatory approvals and市場慣行.
Strategic Considerations
- Differentiation through formulations or delivery mechanisms could extend patent life or market exclusivity.
- Early market entry in regions with favorable regulatory environments offers revenue opportunities.
- Partnering with local manufacturers may reduce costs and improve competitiveness.
Key Risks
- Regulatory delays in approved markets.
- Market saturation with generics leading to price suppression.
- Stringent control measures for benzodiazepines may limit prescribing volume.
Conclusion
ALPRAZOLAM XR holds a niche in the long-acting benzodiazepine segment, with its market size expected to expand gradually. Initial high prices are likely to decrease significantly within two years post-generic entry. Manufacturers should focus on securing regulatory approvals, optimizing manufacturing, and strategic partnerships to maximize market share.
Key Takeaways
- ALPRAZOLAM XR aims at improving adherence in anxiety treatment but faces imminent generic competition.
- Market size is driven by annual growth in anxiety disorders and demand for extended-release formulations.
- Pricing is expected to decline sharply after patent expiry, with initial prices around USD 30-60 monthly.
- Competitive landscape includes several established formulations, with patent expiries opening opportunities.
- Success depends on regulatory approval timelines and market acceptability of extended-release benzodiazepines.
FAQs
-
When is ALPRAZOLAM XR expected to get FDA approval in the US?
Approval timelines are uncertain; filing was completed in 2022 with expected decision in 12-24 months, subject to review outcomes and additional data requirements.
-
How does ALPRAZOLAM XR differ from immediate-release formulations?
It provides sustained therapeutic levels over 24 hours, reducing dosing frequency from multiple times daily to once daily, improving patient compliance.
-
What are the main challenges for market penetration of ALPRAZOLAM XR?
Key challenges include regulatory delays, patent expiries leading to generic competition, and evolving prescribing preferences favoring non-benzodiazepine therapies.
-
What pricing strategies are effective post-patent expiry?
Competitive pricing aligned with generics, cost optimization, and value demonstration for clinicians can help maintain market share.
-
Are there any known safety concerns with ALPRAZOLAM XR?
Safety profile is aligned with other benzodiazepines; risks include dependence, sedation, and impairment, necessitating careful patient management and monitoring.
References
[1] MarketWatch. Benzodiazepines Market Size, Share & Trends Analysis Report 2021-2028. Retrieved from https://www.marketwatch.com
[2] IQVIA. Global Psychiatry & Mental Health Market Insights 2022. Report available upon subscription.
[3] FDA. Drug Approved Applications. 2022. Retrieved from https://www.fda.gov
[4] European Medicines Agency. Marketing Authorization Approvals 2022. Retrieved from https://www.ema.europa.eu
More… ↓
