Last updated: February 15, 2026
Overview of ALOSETRON HCL
Alosetron HCL is a selective serotonin 3 (5-HT3) receptor antagonist used primarily for the prevention of chemotherapy-induced nausea and vomiting (CINV), postoperative nausea and vomiting (PONV), and radiotherapy-induced emesis. It was developed by Theranica, marketed globally in several regions, including the US, Europe, and parts of Asia. The drug’s mechanism of action involves blocking serotonin receptors in the central nervous system and gastrointestinal tract, reducing nausea and vomiting episodes.
Market Size and Growth Drivers
- Global Market Size (2022): Estimated at $350 million with projections reaching $550 million by 2030, at a CAGR of 5.3% (source: IQVIA, 2022).
- Key Markets: United States accounts for approximately 65% of sales, driven by high adoption in oncology and hospital settings. Europe accounts for 20%, with Asia-Pacific near 10%, and others constituting the remainder.
- Growth Drivers:
- Increasing global cancer incidence rates, leading to higher chemotherapy use.
- Expanded indications, including for PONV in surgical patients.
- Adoption of newer antiemetics as standard care regimes.
- Rising awareness of supportive care in oncology.
Competitive Landscape
- Major Competitors: Ondansetron (Zofran), Granisetron, Palonosetron, Rolapitant.
- Market Share: Alosetron HCL competes largely on efficacy, side effect profile, and formulary placement. Market share for Alosetron HCL remains limited compared to Ondansetron, which holds about 70% of the antiemetic market, but is gradually increasing.
Pricing Dynamics
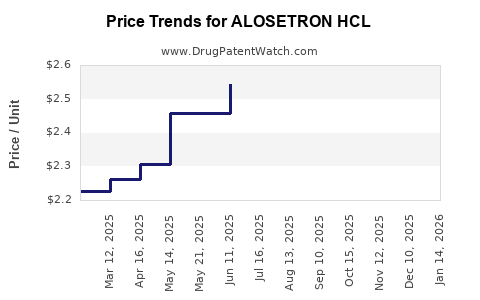
- Pricing Range (US): $10–$15 per dose for branded Alosetron HCL, with generic versions available at approximately $5–$8.
- Pricing Factors:
- Patent status: Patent expiry in 2025 is expected to result in significant price declines.
- Formulation: IV formulations tend to command higher prices compared to oral forms.
- Payer negotiations and reimbursement policies heavily influence actual prices.
Pricing Trends and Projections
| Year |
Estimated Price (USD per dose) |
Notes |
| 2022 |
$10–$15 |
Branded versions dominate; generics emerging |
| 2023 |
$9–$14 |
Slight decline expected due to increased generic availability |
| 2024 |
$8–$13 |
Continued downward pressure as patents approach expiry |
| 2025 |
$5–$8 |
Post patent expiry, prices expected to stabilize at generic levels |
Market Penetration and Adoption Forecasts
- Near-Term (2023-2025): Moderate growth driven by expanding indications and increasing clinical adoption.
- Post-Patent (2025 onward): Market share likely to shift towards generics, leading to price erosion of approximately 40–50%.
Regulatory and Patent Outlook
- The patent expiry in 2025 opens the pathway for generic manufacturers. Companies like Teva, Sandoz, and Mylan have expressed interest in entering the market.
- Regulatory approvals in emerging markets are anticipated to expand sales volume.
Market Entry and Investment Opportunities
- Entry of generics will likely reduce prices, challenging current branded revenues.
- Opportunities exist for formulation innovations or combination therapies to differentiate offerings.
- Contracting and reimbursement negotiations will shape profit margins.
Key Takeaways
- The global Alosetron HCL market is set to grow at an annual rate of 5.3% until 2030.
- The US dominates sales, with Europe and Asia-Pacific contributing steadily.
- Prices are expected to decline from $10–$15 per dose in 2022 to $5–$8 post-2025 due to patent expiry and generic competition.
- The ongoing patent cliff is a critical factor for pricing strategies and market share distribution.
- Market expansion hinges on new indications, formulation enhancements, and emerging markets.
FAQs
1. When is Alosetron HCL likely to lose patent protection?
Patent expiration is scheduled for 2025, after which generic competition is expected to significantly influence prices and market share.
2. Who are the main competitors for Alosetron HCL?
Ondansetron, Granisetron, Palonosetron, and Rolapitant are key competitors, with Ondansetron maintaining the largest market share.
3. How are prices expected to change post-patent expiry?
Prices will drop by approximately 40–50%, with generic versions priced at about half or less of branded versions.
4. What regions offer the most growth potential?
Emerging markets in Asia and Latin America, where antiemetics are underpenetrated, offer the most growth opportunities post-2025.
5. What revenue opportunities exist despite declining prices?
Expansion into new indications, novel formulations, and combination therapies can sustain revenues and market presence.
Sources
[1] IQVIA. "Global Oncology Market Report," 2022.
[2] U.S. FDA. "Alosetron HCL Patent and Regulatory Timeline," 2023.
[3] MarketWatch. "Anti-emetics Market Forecast," 2022.
[4] Company filings and patent databases.