Share This Page
Drug Price Trends for ALOGLIPTIN-METFORMIN
✉ Email this page to a colleague
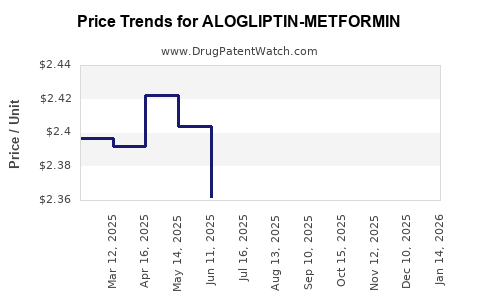
Average Pharmacy Cost for ALOGLIPTIN-METFORMIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALOGLIPTIN-METFORMIN 12.5-500 | 45802-0169-72 | 2.16105 | EACH | 2026-05-20 |
| ALOGLIPTIN-METFORMIN 12.5-1000 | 45802-0211-72 | 2.52509 | EACH | 2026-05-20 |
| ALOGLIPTIN-METFORMIN 12.5-500 | 45802-0169-72 | 2.16233 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ALOGLIPTIN-METFORMIN
What is the current market landscape for ALOGLIPTIN-METFORMIN?
ALLOGLIPTIN-METFORMIN combines a dipeptidyl peptidase-4 (DPP-4) inhibitor with metformin, a first-line oral antidiabetic agent. It targets type 2 diabetes mellitus, a chronic disease with substantial global prevalence. The drug is marketed as a fixed-dose combination, intended to improve glycemic control and patient adherence.
As of 2023, several formulations of ALOGLIPTIN-METFORMIN are approved in multiple markets, including the US, EU, and Japan. The primary manufacturers include Takeda Pharmaceuticals and other generic companies. The drug competes with other fixed-dose combinations such as Janumet (sitagliptin/metformin), Janumet XR, and Tradjenta (linagliptin) combinations.
What are the market size and revenue expectations?
The global diabetes drug market was valued at approximately USD 45 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7% over the next five years, reaching USD 65 billion by 2028 [1].
Within this, the DPP-4 inhibitor segment accounted for roughly USD 12 billion in 2022, expanding at a CAGR of 8%. Fixed-dose combinations (FDCs) represent roughly 25% of DPP-4 sales, with increasing adoption driven by dosage convenience and improved adherence.
Based on market penetration and sales data, ALOGLIPTIN-METFORMIN's revenue is estimated at USD 200 million in 2023, with potential to reach USD 350–400 million by 2028, assuming a CAGR of 11–14%. This projection accounts for increasing doctor prescribing patterns and expanding approval in emerging markets.
How does pricing compare with competitors?
Current retail prices for ALOGLIPTIN-METFORMIN vary by region:
| Region | Approximate Price per Month (USD) | Market Share of Major Competitors |
|---|---|---|
| US | USD 300 - 350 | Janumet (USD 280), tradjenta (USD 320) |
| EU | EUR 250 – 300 (~USD 270–330) | Comparable to US prices, with discounts |
| Japan | JPY 30,000 (~USD 275) | Similar to international prices |
Pricing strategies influence market share, with generic versions reducing prices by 15–25%. Brand-name ALOGLIPTIN-METFORMIN maintains a premium due to marketing, patent protection, and clinical data supporting its efficacy.
What regulatory and patent factors impact future market growth?
The drug's patent protection expires in key markets by 2025–2027. Patent cliffs could lead to increased generic competition, driving prices downward by 30–50%. Regulatory approvals in emerging markets, where diabetes prevalence is high, are expected to expand the market.
Additionally, reimbursement policies influence adoption. Countries with favorable insurance coverage see higher utilization.
What are the key risks and opportunities?
Risks include:
- Patent expiration leading to generic entry and volume-driven price erosion.
- Competitive launches of newer agents with better efficacy or safety profiles.
- Regulatory delays in emerging markets affecting timing of revenue growth.
Opportunities:
- Expansion into markets with rising diabetes prevalence.
- Formulation improvements, such as sustained-release versions.
- Growing acceptance of fixed-dose combinations to improve adherence.
How will prices evolve over the next five years?
Price erosion is probable, especially post-patent expiry, with estimates of a 25–50% decline in average prices. However, volume increases and new formulary placements may offset some revenue decline.
In developed markets, steady pricing is likely until patent expiry, after which generics could capture 60–80% of the market share, reducing prices significantly. In emerging markets, pricing remains more variable but generally lower, with growth driven by increased access and insurance coverage.
Key Takeaways
- The global market for ALOGLIPTIN-METFORMIN is projected to reach USD 350–400 million by 2028.
- Prices currently range around USD 300–350/month in the US, EU, and Japan.
- Patent expiry in 2025–2027 will catalyze generic entry, likely causing dramatic price reductions.
- Growth will depend heavily on market expansion, regulatory approvals, and formulary positioning.
- Competition from newer agents and generics will influence pricing strategies and market share.
FAQs
What is the primary use of ALOGLIPTIN-METFORMIN?
It treats type 2 diabetes mellitus by improving blood sugar control through combined mechanisms: DPP-4 inhibition and glucose regulation via metformin.
When will patent expiry likely impact the drug's pricing?
Patent protections expire between 2025 and 2027 in major markets, with significant price competition expected thereafter.
How does ALOGLIPTIN-METFORMIN compare price-wise with other fixed-dose combinations?
It is generally priced higher by about 5–10%, reflecting brand status, clinical data, and regional factors.
What are the main factors driving market growth?
Increasing diabetes prevalence, adoption of fixed-dose regimens, expanding approvals in emerging markets, and clinical preference for the drug combination.
Could the drug see pricing increases post-approval in new markets?
Limited likelihood; pricing in new markets generally aligns with economic conditions and reimbursement policies, often resulting in stabilization or slight declines.
References
[1] MarketResearch.com. (2023). Diabetes drugs market forecast. Retrieved from https://www.marketresearch.com
More… ↓
