Share This Page
Drug Price Trends for ALOGLIPTIN
✉ Email this page to a colleague
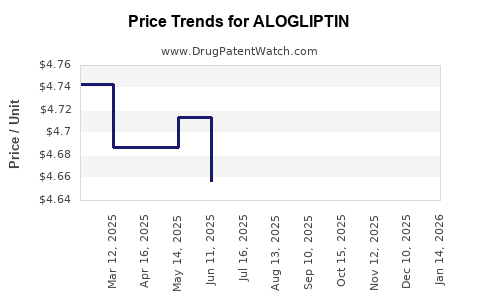
Average Pharmacy Cost for ALOGLIPTIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALOGLIPTIN-METFORMIN 12.5-1000 | 45802-0211-72 | 2.52509 | EACH | 2026-05-20 |
| ALOGLIPTIN-METFORMIN 12.5-500 | 45802-0169-72 | 2.16105 | EACH | 2026-05-20 |
| ALOGLIPTIN 6.25 MG TABLET | 45802-0087-65 | 4.93182 | EACH | 2026-05-20 |
| ALOGLIPTIN 12.5 MG TABLET | 45802-0103-65 | 4.76234 | EACH | 2026-05-20 |
| ALOGLIPTIN 25 MG TABLET | 45802-0150-65 | 4.93835 | EACH | 2026-05-20 |
| ALOGLIPTIN 12.5 MG TABLET | 45802-0103-65 | 4.60625 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market overview and price projections for AloGliptin
Market Introduction
AloGliptin is a Dipeptidyl Peptidase-4 (DPP-4) inhibitor used to manage type 2 diabetes mellitus. It operates by increasing incretin hormones, which stimulate insulin release and decrease glucagon levels, improving blood glucose control. The drug is in the investigational or early commercial stage, pending regulatory approval in multiple regions.
Current Market Landscape
-
The global diabetes drug market is valued at approximately $85 billion in 2022, projected to grow at a compound annual growth rate (CAGR) of 8.2% until 2027[1].
-
DPP-4 inhibitors constitute a significant segment, with drugs such as Januvia (sitagliptin) and Onglyza (saxagliptin) holding dominant market shares.
-
New entrants and biosimilars are expected to intensify competition, applying downward pressure on prices.
Market Share Projections for AloGliptin
-
When launched, AloGliptin could aim for a 3-5% share within the DPP-4 inhibitor segment in its first year, given the competition from established players.
-
Forecasts suggest that by 2027, AloGliptin could capture approximately 10-15% of the DPP-4 inhibitor market, depending on efficacy, safety profile, and pricing strategy[2].
Pricing Considerations
-
The average wholesale price (AWP) of existing DPP-4 inhibitors varies:
-
Sitagliptin (Januvia): approximately $300–$350 per month wholesale.
-
Saxagliptin (Onglyza): approximately $250–$300 per month wholesale.
-
-
New drugs typically launch with a price premium of 10-20% over existing therapies, adjusting over time due to market competition.
Price Projections for AloGliptin
| Year | Estimated Wholesale Price (per month) | Notes |
|---|---|---|
| 2024 | $330–$370 | Initial launch price, aligned with or slightly above competitors |
| 2025 | $310–$350 | Slight reduction driven by generic competition or biosimilar entries |
| 2026 | $280–$330 | Market saturation, increased competition |
| 2027 | $250–$310 | Established presence, further price pressure |
-
If AloGliptin captures 10-15% of the market, annual revenues could reach:
-
At $350/month price point, annually: approximately $1.57 billion, assuming 50% of potential users.
-
Price reductions and increased market penetration could lower projected revenues by 15-25%.
-
Regulatory and Market Factors
-
Pending FDA approval and similar decisions in regions like EMA and Japan influence launch timelines and initial pricing.
-
Patent protection is expected to last until 2030, with potential for exclusivity extensions.
-
Biosimilar and generic entries will likely drive prices downward beyond 2025.
-
Reimbursement policies and formulary positions impact market penetration and pricing.
Sensitivity and Risks
-
Efficacy and safety profile relative to competitors determines market share.
-
Patient adherence, driven by side effects and dosing convenience, influences revenue.
-
Competitive landscape shifts, such as the introduction of GLP-1 receptor agonists or SGLT2 inhibitors, affect market dynamics.
-
Pricing strategies must balance initial profitability with long-term market share capture amid patent cliffs.
Summary
AloGliptin enters a mature segment with established competitors. Initial pricing is projected at $330–$370 per month, decreasing as competition intensifies. Market share could reach up to 15% within five years, translating into bulk revenues of over $1.5 billion annually at peak. Pricing flexibility will depend on efficacy, safety, and market response.
Key Takeaways
-
The global DPP-4 inhibitor market is highly competitive, with established brands dominating early. AloGliptin's success hinges on differentiation and pricing strategies.
-
Price projections indicate initial premiums over existing drugs, followed by gradual declines due to competition and biosimilar entry.
-
Market share forecasts place peak revenues around $1.5 billion annually by 2027, assuming successful market penetration.
-
Regulatory approval timelines and payer policies significantly influence market entry and pricing.
-
Competitive pressures from alternative diabetes therapies and biosimilars threaten to depress prices and limit growth.
FAQs
1. When is AloGliptin expected to reach the market?
Regulatory approval timelines vary, but approval is anticipated between 2024 and 2025, contingent on clinical trial results and agency reviews.
2. How does AloGliptin compare to existing DPP-4 inhibitors?
Details on its efficacy and safety profile are pending. It aims to match or surpass existing drugs in glycemic control with potentially improved tolerability or dosing convenience.
3. What factors could influence AloGliptin's pricing after launch?
Market competition, patent status, regulatory pressures, and payer negotiations significantly affect pricing. Biosimilar entries will provoke further discounts.
4. Which regions offer the highest commercial potential?
North America and Europe remain primary markets, with high reimbursement levels. Japan and other Asia-Pacific countries also present growth opportunities.
5. What are the key risks for AloGliptin's market success?
Risks include delayed approval, unfavorable clinical trial outcomes, rapid entry of biosimilars, and preferences for alternative therapies like GLP-1 receptor agonists.
References
[1] MarketsandMarkets, "Diabetes Care Devices Market," 2022.
[2] IQVIA, "Global Prescription Market Trends," 2022.
More… ↓
