Last updated: February 12, 2026
What is Allopurinol and What is its Market?
Allopurinol is a xanthine oxidase inhibitor used primarily to treat gout, kidney stones, and hyperuricemia associated with chemotherapy. It was first approved by the FDA in 1966 and remains a standard medication in its class. The drug is available as a generic and under brand names such as Zyloprim.
The global market for allopurinol was valued at approximately $1.2 billion in 2022. Growth drivers include increasing prevalence of gout and hyperuricemia, expanding geriatric populations, and rising awareness of uric acid management. It is marketed in various regions, with the highest sales in North America and Europe, followed by Asia-Pacific.
How is the market structured?
The manufacturing landscape involves several generic producers and a few brand-name developers. Major players include Teva, Sandoz, and Mylan, with some originators like Sanofi holding patents during early market phases.
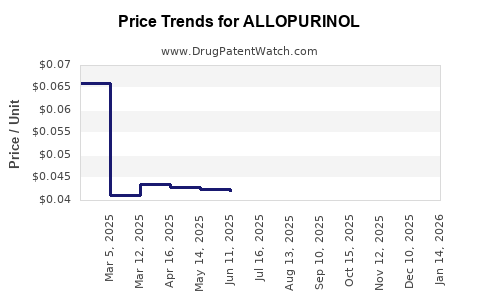
Pricing varies based on formulation, dosage, and region. In the U.S., the average retail price for a 100-count bottle of 300 mg tablets is approximately $70–$100. Generic versions generally cost 20%-40% less than branded formulations.
What are the key factors influencing pricing?
- Regulatory status: Generic availability drives prices downward.
- Manufacturing costs: Slight variations impact retail prices.
- Market competition: Increased generic entries reduce prices.
- Reimbursement policies: Insurance coverage impacts consumer out-of-pocket expenses.
- Regional economic factors: Pricing is often higher in developed countries, influenced by healthcare system structures.
What are the current market trends?
- Generic proliferation: Nearly all formulations are available as generics, intensifying price competition.
- Patent expirations: Many originator patents expired in the late 1990s and early 2000s, leading to increased generic penetration.
- Emerging markets: Growth in Chinese and Indian markets owing to rising gout prevalence and expanding healthcare infrastructure.
- Development of new formulations: Efforts to improve bioavailability or reduce side effects are ongoing, but few have reached market status.
Price Projections (2023–2028)
| Year |
Estimated Market Size |
Projected Average Price (per 300 mg tablet) |
Key Drivers |
| 2023 |
$1.2 billion |
$0.70–$1.00 |
Continued generic availability, stable demand |
| 2024 |
$1.3 billion |
$0.65–$0.95 |
Further generic entry, price competition |
| 2025 |
$1.4 billion |
$0.60–$0.90 |
Expansion in emerging markets |
| 2026 |
$1.5 billion |
$0.55–$0.85 |
Increased market saturation, potential biosimilar developments |
| 2027 |
$1.6 billion |
$0.50–$0.80 |
Price stabilization with market maturity |
| 2028 |
$1.7 billion |
$0.45–$0.75 |
Price erosion continues, new formulations increasing costs |
Projections assume continued patent expiration and generic competition. Price declines are primarily due to multiple generic entrants and regional price adjustments.
Future Market Opportunities and Challenges
Opportunities
- Biosimilars and advanced formulations: Potential for new delivery systems or reduced side effects.
- Expanding markets: Gout prevalence increasing globally, especially in urbanized regions.
- Patent challenges: For any remaining patented formulations, opportunities for generic entry.
Challenges
- Price erosion: Market saturation leads to declining prices.
- Regulatory hurdles: Variability across regions complicates market entry.
- Market consolidation: Fewer players may lead to pricing stability but also reduce innovation.
Regulatory Landscape
World Health Organization (WHO) includes allopurinol on the Essential Medicines List, facilitating access in developing countries. The U.S. FDA regulates formulations, with generic drugs approved via abbreviated new drug applications (ANDAs). European Medicines Agency (EMA) follows similar procedures, promoting market competition.
Investment Implications
Given the mature stage of the allopurinol market, investments should focus on potential biosimilar competitors and formulation innovations. The market's stability favors established generic producers with cost advantages. Patents covering ligand modifications or combination products remain potential opportunities.
Key Takeaways
- The allopurinol market exceeds $1.2 billion globally, with stable growth driven by aging populations and increasing gout prevalence.
- Price declines are driven by widespread generic availability, with prices expected to decrease from an average of $0.70–$1.00 per tablet in 2023 to as low as $0.45 per tablet by 2028.
- Market saturation and price erosion pose challenges for profit margins, favoring large-volume, low-cost producers.
- Opportunities exist in emerging markets and in developing new formulations, but regulatory variability remains a barrier.
- Competition from biosimilars and novel treatments may influence future dynamics.
FAQs
1. How does the patent landscape affect allopurinol pricing?
Most patents expired in the late 1990s and early 2000s, allowing generics to enter markets globally and driving prices downward.
2. Are there any recent developments in allopurinol formulations?
Limited, but research is ongoing into formulations with improved bioavailability and reduced side effects, though few have reached commercial markets.
3. How does regional pricing differ?
Prices are generally higher in North America and Europe due to healthcare system structures, with lower prices in Asia-Pacific and Latin America owing to different regulatory and economic factors.
4. What is the impact of biosimilars on the allopurinol market?
Biosimilars are less common for allopurinol due to its small-molecule nature, but any new formulations or combination products could alter market dynamics.
5. What are the prospects for market growth?
The market is mature, with slow but steady growth driven by emerging markets. Major growth drivers are demographic shifts and increased awareness.
References
[1] Market Research Future, 2022. "Global Allopurinol Market Overview."
[2] IQVIA, 2022. "Pharmaceutical Price Trends."
[3] FDA, 2022. "Approval and Patent Status of Allopurinol."