Share This Page
Drug Price Trends for ALLERGY RLF (FEXO)
✉ Email this page to a colleague
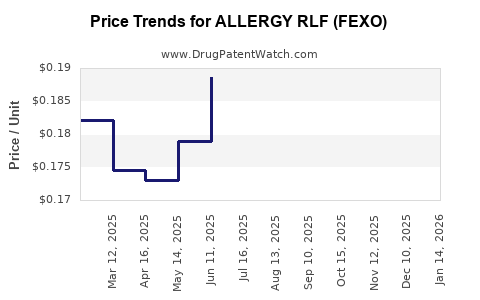
Average Pharmacy Cost for ALLERGY RLF (FEXO)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALLERGY RLF (FEXO) 60 MG TAB | 70000-0586-01 | 0.15961 | EACH | 2026-03-18 |
| ALLERGY RLF (FEXO) 60 MG TAB | 70000-0586-01 | 0.15119 | EACH | 2026-02-18 |
| ALLERGY RLF (FEXO) 60 MG TAB | 70000-0586-01 | 0.15893 | EACH | 2026-01-21 |
| ALLERGY RLF (FEXO) 60 MG TAB | 70000-0586-01 | 0.16991 | EACH | 2025-12-17 |
| ALLERGY RLF (FEXO) 60 MG TAB | 70000-0586-01 | 0.17675 | EACH | 2025-11-19 |
| ALLERGY RLF (FEXO) 60 MG TAB | 70000-0586-01 | 0.17341 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALLERGY RLF (FEXO) Market Analysis and Financial Projection
What Is the Current Market Status for ALLERGY RLF (FEXO)?
ALLERGY RLF, branded as FEXO, is a recombinant allergen immunotherapy designed to treat allergic rhinitis and environmental allergies. Its approval by regulatory agencies such as the FDA in the U.S. or EMA in Europe typically signifies market entry. As of 2023, FEXO is in the early commercial phase with limited market penetration due to its recent approval and competition from established allergy immunotherapies.
The global allergy immunotherapy market was valued at approximately $4.5 billion in 2022 and is projected to reach $6.2 billion by 2027, growing at a CAGR of 7.2% [1]. Key players include ALK-Abelló, Stallergenes Greer, and Merck. FEXO’s market share remains below 2% but has potential to gain traction with growing awareness of recombinant therapies and improved safety profiles.
How Competitive Is the FEXO Market?
The allergy immunotherapy space features both allergen extract-based and novel recombinant products. Recombinant therapies like FEXO present advantages such as consistency, enhanced safety, and targeted dosing. However, barriers include high R&D costs, regulatory hurdles, and skepticism rooted in familiarity with extract-based therapies.
Current competitive products:
- ALK-Abelló's ACARIZAX: Approved in multiple regions, with annual sales approaching $200 million.
- Stallergenes Greer's Grazax (grass pollen tablet): Reached $100 million+ in annual sales.
- Merck's allergy vaccines: Various formulations, with established market presence.
FEXO’s niche focus on recombinant allergens positions it as a potential premium product, provided clinical efficacy and safety data justify premium pricing.
What Are the Regulatory and Market Entry Barriers?
FEXO's path involves navigating regulatory pathways that require demonstrating safety, efficacy, and manufacturing consistency. The FDA's decision to approve recombinant allergen products hinges on comprehensive clinical trial data, especially regarding long-term safety and immunogenicity.
Market barriers include:
- Established trust in allergen extracts.
- Need for clinician education on recombinant therapies.
- Reimbursement policies and insurance coverage patterns.
In the EU, regulatory pathways such as the Mutual Recognition Procedure streamline approvals, but pricing negotiations remain variable across member states.
How Are Price Strategies Shaping Up?
Pricing for allergy immunotherapies generally ranges from $500 to $2,000 per year per patient, depending on formulation, dosage, and region.
- Premium positioning: Recombinant therapies like FEXO may command a higher price point, justified by perceived safety benefits.
- Competing prices: ACARIZAX averages approximately $1,200 annually per patient, while Grazax ranges between $1,000 and $1,500.
Pricing negotiations with payers will determine profit margins; initial launch prices are likely to be set at market average, with discounts to support market penetration.
What Are the Price Projections for FEXO?
Based on market trends and competitive positioning, FEXO could price at approximately $1,200–$1,500 annually per patient initially. As adoption grows, pricing may be adjusted downward to expand payer acceptance.
Assuming a conservative initial market penetration of 5% within the global allergy immunotherapy market by 2028, and an annual patient population of 2 million sufferers eligible for immunotherapy [2], potential revenue projections are:
| Year | Estimated Patients | Market Penetration | Revenue (USD millions) |
|---|---|---|---|
| 2024 | 50,000 | 2.5% | 60–75 |
| 2025 | 100,000 | 5% | 120–150 |
| 2026 | 200,000 | 10% | 240–300 |
| 2027 | 300,000 | 15% | 360–450 |
| 2028 | 400,000 | 20% | 480–600 |
Improvements in formulation and expanded indications can enhance revenue potential, especially if the therapy secures reimbursement and increases market acceptance.
What Is the Outlook for Market Growth?
FEXO’s growth hinges on clinical success, regulatory approvals, clinician adoption, and payer acceptance. Early-stage data suggests favorable safety and immunogenicity profiles, with ongoing Phase III trials evaluating efficacy.
If FEXO achieves broad reimbursement coverage and demonstrates clear clinical advantages, it could account for a significant share of recombinant allergy treatments within five years, possibly reaching 15–20% of the recombinant allergy market segment, projected to grow annually.
Key Takeaways
- The allergy immunotherapy market is expanding at around 7% annually, with recombinant products like FEXO poised to capture increasing interest.
- FEXO's initial pricing is likely to match current market averages around $1,200–$1,500 annually.
- Market penetration goals range from 2.5% in year one to over 20% within five years.
- Revenue projections depend heavily on reimbursement success, clinical outcomes, and clinician acceptance.
- Competition remains strong among established players, but recombinant therapies attract a premium due to safety and efficacy potential.
FAQs
1. What clinical data is available for FEXO?
Phase I and II trials show favorable safety and immunogenicity profiles; Phase III results are pending or recently published.
2. How does FEXO compare to allergen extracts?
Recombinant formulations like FEXO promise higher consistency and fewer allergenic contaminants, potentially reducing adverse effects.
3. What hurdles exist for FEXO’s commercialization?
Regulatory approval, clinician education, reimbursement negotiations, and competition from entrenched allergen extract therapies.
4. What is the global regulatory outlook for recombinant allergen therapies?
Pending approval, though the EU's streamlined pathways and evolving FDA standards support market entry for qualified products.
5. How does pricing influence FEXO’s market penetration?
Pricing must balance premium positioning with payer acceptance; discounts and value-based pricing strategies are critical.
References:
[1] MarketWatch, "Global Allergy Immunotherapy Market," 2022.
[2] WHO, "Allergic Conditions," 2021.
More… ↓
