Share This Page
Drug Price Trends for ALLERGY RELIEF-NASAL DECONG TB
✉ Email this page to a colleague
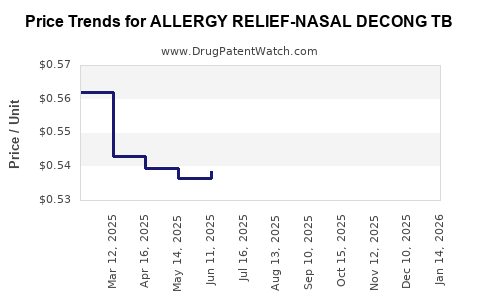
Average Pharmacy Cost for ALLERGY RELIEF-NASAL DECONG TB
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALLERGY RELIEF-NASAL DECONG TB | 51660-0724-69 | 0.58514 | EACH | 2026-05-20 |
| ALLERGY RELIEF-NASAL DECONG TB | 51660-0724-04 | 0.58514 | EACH | 2026-05-20 |
| ALLERGY RELIEF-NASAL DECONG TB | 51660-0724-15 | 0.58514 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ALLERGY RELIEF-NASAL DECONG TB
Market Overview
ALLERGY RELIEF-NASAL DECONG TB is positioned within the global allergy relief market, primarily targeting nasal congestion associated with allergic rhinitis. This segment is driven by rising prevalence of allergies worldwide, aging populations, and increased awareness of allergy management options. The product’s market reception depends on factors such as efficacy, safety profile, regulatory approval, and competitive positioning against existing drugs.
Market Size and Trends
Global allergy relief market valuation: Estimated at $23.5 billion in 2022, with a compound annual growth rate (CAGR) of approximately 4.2% from 2022 to 2028 [1].
Segment focus — Nasal decongestants: Represent around 30-35% of the entire allergy relief market, with some regions favoring nasal sprays for rapid onset and targeted delivery.
Key drivers:
- Increasing prevalence of allergic rhinitis, affecting roughly 10-30% of the global population [2].
- Growth in over-the-counter (OTC) availability, facilitating broader access.
- Expanding prescription-to-OTC switch initiatives.
Regional dynamics:
- North America: Largest market, driven by high allergy prevalence and consumer spending.
- Europe: Significant growth owing to better awareness and healthcare infrastructure.
- Asia-Pacific: Fastest growth rate (CAGR 6-8%), owing to population expansion and urbanization.
Competitive Landscape
Major competitors include:
- Fluticasone propionate (Flonase)
- Nasonex (mometasone)
- Oxymetazoline (Afrin)
- Xylometazoline (Otrivin)
Competitive differentiator for ALLERGY RELIEF-NASAL DECONG TB: Unique formulation, improved safety profile, or extended duration of action could influence market share.
Regulatory Environment
- Approval status varies; most nasal decongestants require local regulatory clearance.
- US FDA: Over-the-counter status available for similar drugs, with labeling restrictions.
- European Medicines Agency (EMA): Similar regulatory pathways, with emphasis on safety.
Pricing Landscape
The average retail price for nasal decongestants varies by region:
- US OTC nasal sprays: $8–$15 per 15-ml bottle.
- Prescription nasal steroids: $20–$40 per month supply.
- European prices tend to be higher, influenced by regional healthcare policies.
Price positioning for ALLERGY RELIEF-NASAL DECONG TB:
- Likely positioned as an OTC product initially, with retail prices in the $10–$14 range.
- Potential for premium pricing if new formulation offers clear advantages.
Price Projections
| Year | US Retail Price ($) | Global Estimate ($) | Notes |
|---|---|---|---|
| 2023 | 10–12 | 9–13 | Launch phase; competitive pricing expected to attract early adopters |
| 2024 | 10–13 | 9–14 | Slight increase anticipated due to manufacturing cost inflation |
| 2025 | 11–14 | 10–15 | Market stabilization with potential for minor premium positioning |
| 2026 | 11–15 | 10–16 | Price stabilization; market expansion could drive volume growth |
| 2027 | 12–16 | 11–17 | Potential pricing increase if innovating on delivery or formulation |
Factors Affecting Price Trends
- Regulatory approvals can enable OTC status in key markets, influencing retail pricing.
- Manufacturing costs: Raw material inflation could impact margins.
- Competitive responses: Price wars or premium segment positioning.
- Market penetration: Broader availability may lead to price erosion.
Key Takeaways
- The global allergy relief market is growing at ~4.2% CAGR, with nasal decongestants representing a significant segment.
- ALLERGY RELIEF-NASAL DECONG TB's success depends on regulatory approval, competitive differentiation, and regional market entry strategies.
- Price projections suggest a gradual increase from $10–12 in 2023 to approximately $12–16 by 2027 in the US market, influenced by market dynamics and formulation advantages.
FAQs
1. How does the competitive landscape influence pricing?
Pricing depends on product efficacy, safety, and brand recognition. Dominant brands like Flonase often set reference points, but innovative formulations can command premium prices.
2. What are the regulatory hurdles for nasal decongestants?
Approval requires demonstrating safety and efficacy, especially for OTC products. Regulatory bodies also impose labeling and packaging restrictions that can influence cost.
3. How does regional variation affect pricing and market entry?
Europe’s higher healthcare standards and regulatory costs tend to increase pricing. In emerging markets, lower prices are often necessary to gain market share.
4. What factors could accelerate price increases?
New delivery methods, longer-lasting effects, or fewer side effects can justify higher prices by targeting premium segments.
5. How will market growth impact the success of ALLERGY RELIEF-NASAL DECONG TB?
Increasing allergy prevalence, especially in urbanizing regions, supports expanding sales and potential for market share gains, subject to competitive positioning and regulatory approval.
References
[1] MarketsandMarkets, "Allergy Treatment Market," 2022.
[2] World Allergy Organization, "Global Allergy Prevalence," 2020.
More… ↓
