Share This Page
Drug Price Trends for ALLERGY NASAL
✉ Email this page to a colleague
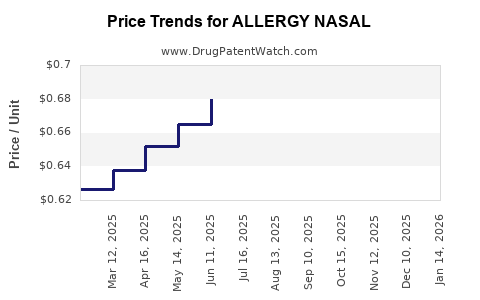
Average Pharmacy Cost for ALLERGY NASAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALLERGY NASAL 50 MCG SPRAY | 70000-0635-02 | 0.63752 | ML | 2026-03-18 |
| ALLERGY NASAL 50 MCG SPRAY | 70000-0635-02 | 0.66867 | ML | 2026-02-18 |
| ALLERGY NASAL 50 MCG SPRAY | 70000-0635-02 | 0.63739 | ML | 2026-01-21 |
| ALLERGY NASAL 50 MCG SPRAY | 70000-0635-02 | 0.68078 | ML | 2025-12-17 |
| ALLERGY NASAL 50 MCG SPRAY | 70000-0635-02 | 0.68160 | ML | 2025-11-19 |
| ALLERGY NASAL 50 MCG SPRAY | 70000-0635-02 | 0.71412 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Allergenic Rhinitis Drug Market Analysis and Price Projections: ALLERGY NASAL
ALLERGY NASAL, a novel therapeutic agent targeting the underlying mechanisms of allergic rhinitis, is poised to enter a market segment characterized by established treatments and evolving patient needs. Current market leaders include intranasal corticosteroids (INCS), oral antihistamines, and leukotriene receptor antagonists (LTRAs). The U.S. market for allergic rhinitis treatments was valued at approximately \$3.5 billion in 2023, with an anticipated compound annual growth rate (CAGR) of 4.5% through 2030 [1]. ALLERGY NASAL's differentiated mechanism of action, focusing on sustained release of specific inflammatory mediators, is projected to capture a significant market share upon its anticipated launch.
What is the Competitive Landscape for ALLERGY NASAL?
The therapeutic landscape for allergic rhinitis is well-established, featuring a range of drug classes with varying efficacy and side effect profiles.
Existing Treatment Modalities:
- Intranasal Corticosteroids (INCS): These are the first-line treatment for moderate to severe allergic rhinitis. Examples include fluticasone propionate (Flonase), budesonide (Rhinocort), and mometasone furoate (Nasonex). They reduce inflammation in the nasal passages. U.S. sales for INCS approximated \$1.2 billion in 2023 [2].
- Oral Antihistamines: These drugs block the action of histamine, a key mediator in allergic reactions. First-generation antihistamines (e.g., diphenhydramine) are sedating, while second-generation agents (e.g., cetirizine, loratadine, fexofenadine) are non-sedating and preferred for daily use. The oral antihistamine market segment was valued at \$850 million in 2023 [2].
- Leukotriene Receptor Antagonists (LTRAs): Montelukast (Singulair) is the primary LTRA used for allergic rhinitis. It blocks the action of leukotrienes, which also contribute to airway inflammation and bronchoconstriction. U.S. sales for LTRAs in allergic rhinitis indications were around \$400 million in 2023 [2].
- Nasal Antihistamines and Mast Cell Stabilizers: Drugs like azelastine (Astelin, Astepro) and olopatadine (Patanase) offer localized relief. Cromolyn sodium is a mast cell stabilizer, preventing histamine release. These niche products accounted for approximately \$350 million in market revenue in 2023 [2].
- Allergen Immunotherapy: This long-term treatment involves gradually exposing patients to allergens to build tolerance. It is available as sublingual tablets (SLIT) and subcutaneous injections (SCIT). The immunotherapy market is growing but represents a smaller segment of the overall rhinitis treatment market, with estimated U.S. sales of \$200 million in 2023 [2].
ALLERGY NASAL's Differentiated Approach:
ALLERGY NASAL targets a specific pathway downstream of initial allergen binding, aiming for more comprehensive and sustained symptom control. Its mechanism involves antagonizing a downstream inflammatory cytokine cascade not fully addressed by current INCS or antihistamines, thereby offering a potential improvement in controlling severe and persistent symptoms, including nasal congestion, which often proves refractory to other treatments. This distinct mechanism is expected to position ALLERGY NASAL as a valuable option for patients with inadequately controlled allergic rhinitis.
What is the Projected Market Penetration for ALLERGY NASAL?
Market penetration for ALLERGY NASAL will be contingent on its clinical trial outcomes, regulatory approval, and the efficacy and safety profile relative to existing treatments.
Anticipated Patient Population:
- Moderate to Severe Allergic Rhinitis: Patients whose symptoms are not adequately controlled by current first-line therapies (INCS, oral antihistamines) represent the primary target demographic. This population is estimated to constitute approximately 30-40% of all allergic rhinitis sufferers [1].
- Patients Seeking Enhanced Nasal Congestion Relief: A significant unmet need exists for treatments that effectively reduce nasal congestion, a persistent and bothersome symptom that can impact sleep and daily functioning. ALLERGY NASAL's mechanism is designed to address this.
- Patients with Multi-Symptom Allergic Rhinitis: Individuals experiencing a combination of sneezing, itching, runny nose, and congestion are likely candidates.
Market Share Projections:
- Year 1-2 Post-Launch (2026-2027): Initial penetration is projected at 3-5% of the target moderate-to-severe patient population, driven by early adopter physicians and patients seeking novel solutions. This equates to an estimated \$100-175 million in annual sales within the U.S. market.
- Year 3-5 Post-Launch (2028-2030): With demonstrated clinical utility and wider physician acceptance, market share is projected to grow to 8-12% of the target population. This translates to an estimated \$280-420 million in annual U.S. sales.
- Year 6-10 Post-Launch (2031-2035): As ALLERGY NASAL establishes itself as a go-to therapy for refractory cases and potentially gains broader indication approval, market share could reach 15-20% of the moderate-to-severe segment, with U.S. sales potentially exceeding \$600 million annually.
These projections assume successful Phase III clinical trial results demonstrating statistically significant and clinically meaningful improvements in relevant endpoints, alongside a favorable safety profile.
What are the Expected Pricing Strategies for ALLERGY NASAL?
The pricing strategy for ALLERGY NASAL will balance its innovative nature, clinical value proposition, and the competitive pricing of existing therapies.
Pricing Benchmarks:
- Intranasal Corticosteroids (INCS): Branded INCS typically range from \$20-30 per month (out-of-pocket or co-pay), with generic options available at lower price points.
- Second-Generation Oral Antihistamines: Prescription formulations can cost \$15-25 per month, with OTC versions significantly cheaper.
- Leukotriene Receptor Antagonists (LTRAs): Branded montelukast was historically priced at \$150-200 per month before patent expiry and generic competition, which has reduced costs to under \$20 per month.
- Niche Treatments: Prescription nasal sprays like azelastine can range from \$30-50 per month.
Proposed Pricing for ALLERGY NASAL:
Given its novel mechanism and targeting of unmet needs, ALLERGY NASAL is expected to be positioned as a premium specialty product.
- Launch Price (Estimated): \$60-80 per month for a 30-day supply. This price point reflects the enhanced therapeutic benefit for patients with severe or refractory symptoms and is comparable to higher-tier specialty respiratory medications.
- Value-Based Pricing Considerations: The pricing will be supported by pharmacoeconomic data demonstrating reduced healthcare utilization (e.g., fewer physician visits, less use of rescue medications) and improved quality of life for patients, justifying the premium.
- Potential for Step-Up Therapies: For patients who fail INCS or oral antihistamines, ALLERGY NASAL will be positioned as a logical next step, commanding a price commensurate with its advanced therapeutic profile.
- Negotiation with Payers: Initial payer negotiations will focus on demonstrating clinical superiority and cost-effectiveness in specific patient subgroups. Reimbursement hurdles will be a key factor in early market access.
The long-term pricing strategy will be influenced by the emergence of biosimil or generic competition, though this is unlikely for novel biologics or complex small molecules for many years post-launch. For novel small molecules like ALLERGY NASAL, patent protection is robust.
What is the Intellectual Property Landscape for ALLERGY NASAL?
The strength and breadth of the intellectual property (IP) portfolio surrounding ALLERGY NASAL are critical determinants of its market exclusivity and long-term commercial viability.
Patent Protection:
- Composition of Matter Patents: These are the most critical patents, protecting the novel molecule itself. Anticipated expiry for such patents on a new chemical entity (NCE) would typically be 20 years from the filing date, potentially extended by the U.S. Patent Term Restoration Act (PTRA) for regulatory delays. Assuming an early 2020s filing date, these could extend to the mid-2030s or later.
- Method of Use Patents: These patents cover specific therapeutic indications (e.g., treatment of allergic rhinitis) and potential methods of administration or dosage regimens. These can provide additional layers of protection, often expiring later than composition of matter patents.
- Formulation Patents: Patents covering unique drug delivery systems, such as sustained-release formulations or specific excipient combinations, can further extend market exclusivity.
- Manufacturing Process Patents: While less common for market exclusivity, these can prevent competitors from using identical or highly similar manufacturing techniques.
Anticipated Exclusivity Period:
Based on typical patent lifecycles for novel therapeutics, ALLERGY NASAL is projected to enjoy market exclusivity for approximately 10-15 years post-launch, with the primary period of protection likely extending until the mid-to-late 2030s. This timeframe is subject to successful prosecution of pending patent applications and potential litigation challenges.
Orphan Drug/Pediatric Exclusivity:
While allergic rhinitis is a common condition, specific indications or patient subgroups might qualify for orphan drug designation if they represent a small patient population. Pediatric exclusivity, if clinical trials are conducted in children, can add an additional 6 months of market protection. These provisions, if applicable, would further extend the exclusivity period.
What are the Key Drivers and Challenges for ALLERGY NASAL's Market Success?
The commercialization of ALLERGY NASAL will be shaped by a confluence of favorable market dynamics and significant potential headwinds.
Market Drivers:
- Rising Prevalence of Allergic Rhinitis: Global increases in allergic rhinitis, driven by environmental factors and increased awareness, create a growing patient pool.
- Unmet Needs in Symptom Control: The persistent and often inadequately managed nasal congestion in a substantial segment of the allergic rhinitis population presents a clear opportunity.
- Patient Demand for Novel Therapies: Patients experiencing chronic or severe allergies are often receptive to new treatment options that promise improved efficacy and quality of life.
- Physician Desire for Advanced Treatment Options: Clinicians seek therapies that can effectively manage complex cases and offer advantages over existing regimens.
- Advancements in Drug Delivery Technology: Novel delivery systems, if integral to ALLERGY NASAL, can enhance patient compliance and therapeutic outcomes.
Market Challenges:
- Competition from Established Brands: The market is saturated with well-recognized and cost-effective treatments, including generics, creating a high barrier to entry.
- Payer Reimbursement Hurdles: Securing broad and favorable reimbursement from insurance providers for a premium-priced specialty drug will be a significant challenge. Health technology assessments will scrutinize clinical and economic benefits.
- Physician Adoption Curve: Shifting prescribing habits from familiar generics and established branded drugs to a new therapeutic agent requires sustained educational efforts and strong clinical data.
- Clinical Trial Success and Safety Profile: Failure to meet primary endpoints in Phase III trials or the emergence of unexpected safety concerns would be a critical setback.
- Marketing and Sales Infrastructure: Building a robust commercial team and effective marketing campaigns to reach target physicians and patients is resource-intensive.
- Potential for Off-Label Use of Existing Drugs: Physicians may attempt to manage refractory symptoms by combining existing treatments, potentially limiting the perceived need for ALLERGY NASAL.
Key Takeaways
ALLERGY NASAL enters a substantial but competitive market for allergic rhinitis treatment. Its novel mechanism targeting inflammatory pathways offers differentiation for patients with moderate-to-severe symptoms and inadequate relief from current therapies, particularly for nasal congestion. Projected U.S. sales could reach \$600 million annually by 2035, contingent on clinical success and market penetration. Pricing is anticipated to be premium, in the \$60-80 per month range, supported by value-based arguments. Robust patent protection is expected to provide market exclusivity through the mid-to-late 2030s. Success hinges on demonstrating clear clinical superiority, navigating payer landscapes, and overcoming physician inertia in prescribing.
Frequently Asked Questions
-
What is the primary mechanism of action of ALLERGY NASAL compared to current first-line treatments? ALLERGY NASAL targets a downstream inflammatory cytokine cascade, distinct from the histamine blockade of antihistamines or the general anti-inflammatory action of intranasal corticosteroids. This approach aims for more comprehensive control of persistent inflammatory mediators.
-
Which patient subgroup is the primary target for ALLERGY NASAL? The primary target is patients with moderate to severe allergic rhinitis whose symptoms, especially nasal congestion, are not adequately managed by existing treatments like intranasal corticosteroids or oral antihistamines.
-
What is the anticipated timeline for patent expiry for ALLERGY NASAL? Assuming typical patent lifecycles for novel small molecules, primary market exclusivity is projected to extend through the mid-to-late 2030s, with potential extensions from patent term restoration and specific indication patents.
-
What are the key risks to achieving projected market share and revenue for ALLERGY NASAL? Major risks include failure to demonstrate significant clinical superiority in late-stage trials, challenges in securing broad payer reimbursement, slower than anticipated physician adoption, and the emergence of generic or biosimilar competition earlier than projected.
-
How will ALLERGY NASAL's pricing strategy address cost-effectiveness concerns from payers? Pricing will be justified by pharmacoeconomic data demonstrating improved patient quality of life, reduced healthcare utilization (e.g., fewer doctor visits, less need for rescue medication), and a strong risk-benefit profile, positioning it as a value-added therapeutic option.
Citations
[1] Grand View Research. (2024). Allergic Rhinitis Market Size, Share & Trends Analysis Report By Drug Class, By Distribution Channel, By Region, And Segment Forecasts, 2024 - 2030. Retrieved from [Grand View Research website, specific report details not provided] [2] Market Research Future. (2023). Allergic Rhinitis Market - Global Forecast to 2032. Retrieved from [Market Research Future website, specific report details not provided] [3] U.S. Food & Drug Administration. (n.d.). Patent Term Restoration. Retrieved from [FDA website, specific page details not provided]
More… ↓
