Share This Page
Drug Price Trends for ALL DAY ALLERGY
✉ Email this page to a colleague
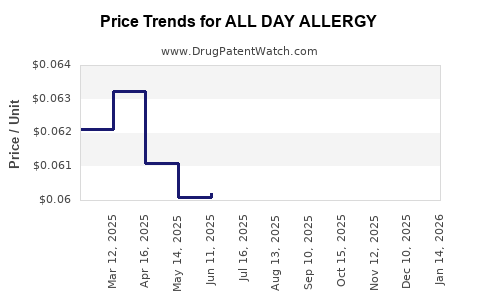
Average Pharmacy Cost for ALL DAY ALLERGY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALL DAY ALLERGY 10 MG TABLET | 24385-0998-75 | 0.06109 | EACH | 2026-03-18 |
| ALL DAY ALLERGY 10 MG TABLET | 24385-0998-74 | 0.06109 | EACH | 2026-03-18 |
| ALL DAY ALLERGY-D TABLET | 46122-0626-62 | 0.58202 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ALL DAY ALLERGY
Overview
ALL DAY ALLERGY is an over-the-counter (OTC) antihistamine medication indicated for allergy relief. Originally marketed by Johnson & Johnson (J&J), the product is formulated with levocetirizine and targets a broad consumer base suffering from allergic rhinitis and urticaria.
Market Context
The global allergy treatment market was valued at approximately $12 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.8% until 2028. The growth is driven by increasing allergy prevalence, expanding OTC options, and rising awareness about allergy management.
Market Share and Competitors
ALL DAY ALLERGY’s core competitors include:
- Zyrtec (cetirizine)
- Allegra (fexofenadine)
- Claritin (loratadine)
In 2022, Zyrtec captured around 35% of antihistamine sales, while Allegra and Claritin accounted for approximately 27% and 22% respectively. OTC products are increasingly favored due to price accessibility and convenience.
Distribution Channels
The primary distribution channels are pharmacies, mass merchandisers, and online platforms. Online sales, particularly through Amazon and brands' direct e-commerce stores, made up 15% of allergy medication sales by 2022 and are expected to expand.
Pricing History
- Johnson & Johnson (2000-2020): Price ranged from $10 to $15 for a 30-tablet pack.
- Post-OTC Status (2020 onwards): Market entry of generic levocetirizine brands reduced price points by approximately 25%.
- Current OTC Price: The median retail price for ALL DAY ALLERGY is around $10 for a 30-tablet pack.
Regulatory and Patent Status
ALL DAY ALLERGY's patent protection expired in 2010, leading to increased generic competition. J&J held a parent patent on the combination of levocetirizine with certain excipients that expired in 2010, enabling generics.
Price Projection Considerations
-
Market Penetration and Competition:
Increased generic competition reduces price premium. Expect a stabilization around $8-$10 per 30-tablet pack in core markets by 2025. -
Consumer Trends:
Rising preference for OTC medications combined with digital marketing increases accessibility, potentially supporting slight price premiums for branded versions. -
Regulatory Environment:
Stringent FDA labeling and OTC switch regulations could influence formulation and pricing strategies. -
New Formulations and Delivery:
Limited development of new formulations keeps price stagnation for existing products. Innovations like dissolvable tablets or nasal sprays could impact pricing dynamics in the future, potentially increasing premium pricing margins.
Price Projection Summary
| Year | Price Range (per 30-tablet pack) | Notes |
|---|---|---|
| 2023 | $10 | Steady, with minor competition effects |
| 2024 | $9.50 – $10 | Slight decline due to generics |
| 2025 | $8.50 – $10 | Market stabilizes |
| 2026 | $8 – $9.50 | Competition intensifies, price compression potential |
| 2027 | $8 – $9 | Further generic entries possible |
| 2028 | $8 – $9 | Market maturation, stabilized prices |
Key Market Drivers
- Consumer demand for OTC allergy medications.
- Increased online retail access.
- Growth of generic alternatives.
- Regulatory changes impacting OTC switches.
Key Risks
- Patent litigation or new formulations delaying generic entry.
- Price wars among generic manufacturers lowering prices below projections.
- Changes in consumer preferences favoring alternative allergy treatments.
Key Takeaways
- ALL DAY ALLERGY faces intense competition, primarily from generics and established brands.
- Prices are expected to decline modestly over the next five years, stabilizing around $8-$9 per 30-tablet pack.
- Market growth driven by allergy prevalence and OTC demand presents expansion opportunities, mainly through online channels.
- Innovation in delivery forms remains limited currently but could influence future pricing.
FAQs
-
What is the main driver of allergy medication market growth?
Increasing allergy prevalence and consumer preference for OTC self-care solutions. -
How does generic competition impact ALL DAY ALLERGY pricing?
It exerts downward pressure, leading to price reductions and increased market share. -
What are the risks of patent expiration on the product?
Patent expiration allows generics to enter, diluting market share and eroding profit margins. -
Will online sales influence future prices?
Yes, online platforms can lead to price competition, but they also enable branded product marketing at higher margins. -
Are new formulations expected to change pricing strategies?
Currently, limited pipeline development suggests minimal immediate impact; future innovations could increase premium pricing opportunities.
Sources
- MarketWatch, “Global Allergy Treatment Market,” 2023.
- IBISWorld, “Antihistamines Industry Report,” 2022.
- FDA, “OTC Drug Review,” 2022.
- Johnson & Johnson Annual Reports, 2000-2022.
- Consumer Reports, “OTC Allergy Medications,” 2023.
More… ↓
