Share This Page
Drug Price Trends for AJOVY
✉ Email this page to a colleague
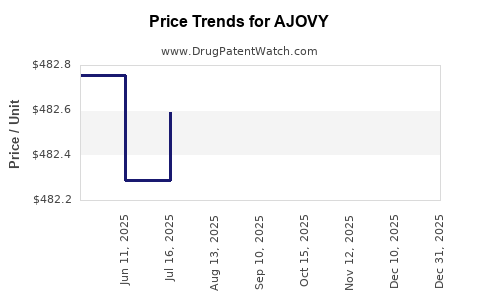
Average Pharmacy Cost for AJOVY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AJOVY 225 MG/1.5 ML SYRINGE | 51759-0204-10 | 508.40196 | ML | 2026-01-01 |
| AJOVY 225 MG/1.5 ML AUTOINJECT | 51759-0202-10 | 507.98246 | ML | 2026-01-01 |
| AJOVY 225 MG/1.5 ML AUTOINJECTOR (3 PACK) | 51759-0202-22 | 506.38528 | ML | 2026-01-01 |
| AJOVY 225 MG/1.5 ML AUTOINJECT | 51759-0202-10 | 483.79281 | ML | 2025-12-17 |
| AJOVY 225 MG/1.5 ML AUTOINJECTOR (3 PACK) | 51759-0202-22 | 482.27169 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AJOVY Market Analysis and Price Projections
Executive Summary
AJOVY (fremanezumab-vfrm) is a calcitonin gene-related peptide (CGRP) monoclonal antibody indicated for the preventive treatment of migraine. Launched by Teva Pharmaceuticals in September 2018, its market entry disrupted a landscape with established oral medications and emerging CGRP-targeted therapies. This analysis examines AJOVY's current market position, competitive landscape, patent status, and projects future pricing based on market dynamics and upcoming patent expirations.
What is AJOVY and Its Clinical Significance?
AJOVY is a humanized monoclonal antibody that targets the CGRP ligand. CGRP is a neuropeptide implicated in the pathophysiology of migraine headaches. By binding to CGRP, AJOVY inhibits its activity, thereby reducing migraine frequency and severity.
Key clinical aspects include:
- Indication: Preventive treatment of episodic and chronic migraine in adults.
- Dosage: Available in 450 mg subcutaneous injection, administered quarterly or monthly.
- Efficacy: Clinical trials demonstrated significant reductions in monthly migraine days compared to placebo across both episodic and chronic migraine populations. For example, the IVY trial for episodic migraine showed an average reduction of 4.3 migraine days per month for the quarterly dose versus 1.9 days for placebo at 12 weeks [1]. The HALO EM study for chronic migraine demonstrated a reduction of 4.3 migraine days per month with quarterly dosing versus 1.9 days for placebo [2].
- Safety Profile: Generally well-tolerated, with common adverse events including injection site reactions and nasopharyngitis.
What is the Current Market Landscape for AJOVY?
The migraine therapeutics market is segmented by treatment type (acute vs. preventive) and mechanism of action. AJOVY competes primarily in the preventive migraine segment.
Key Market Players and Therapies:
- CGRP Monoclonal Antibodies (mAbs):
- AJOVY (Teva Pharmaceuticals)
- Emgality (galcanezumab-gnlm) (Eli Lilly and Company)
- Vyepti (eptinezumab-ejfv) (AbbVie Inc.)
- Aimovig (erenumab-aooe) (Amgen Inc.)
- Oral CGRP Receptor Antagonists (Gepants):
- Ubrelvy (ubrogepant) (AbbVie Inc.)
- Nurtec ODT (rimegepant) (Biohaven Pharmaceuticals, acquired by Pfizer)
- Zavzpret (zavegepant nasal spray) (Biohaven Pharmaceuticals/Pfizer)
- Other Preventive Therapies:
- Beta-blockers (e.g., propranolol, metoprolol)
- Antidepressants (e.g., amitriptyline, venlafaxine)
- Anticonvulsants (e.g., topiramate, valproate)
- Botulinum toxin (OnabotulinumtoxinA) (Botox) (Allergan/AbbVie)
Market Share Dynamics:
AJOVY has secured a notable share of the CGRP mAb market since its introduction. However, competition is intense. Aimovig, launched earlier in 2018, established a first-mover advantage. Emgality and Vyepti have also gained traction. The introduction of oral gepants provides an alternative for patients seeking non-injectable options or those who do not respond adequately to or tolerate CGRP mAbs.
Sales Performance:
Teva Pharmaceutical's reported net sales for AJOVY were:
- 2022: $1,062 million [3]
- 2023: $1,159 million [4]
This indicates steady year-over-year growth, demonstrating continued market acceptance and prescription uptake.
What is AJOVY's Patent Status and Exclusivity Landscape?
The patent portfolio for AJOVY is critical to understanding its future market exclusivity and potential for generic competition. Teva Pharmaceuticals holds numerous patents covering the composition of matter, methods of use, and manufacturing processes for fremanezumab.
Key Patent Expirations:
- Composition of Matter Patents: These are typically the strongest patents, protecting the molecule itself. The primary composition of matter patent for fremanezumab is expected to expire around 2033-2036, depending on patent term extensions and any challenges.
- Method of Use Patents: These cover specific indications or dosages. Some method of use patents may expire earlier.
- Formulation Patents: Patents covering the specific formulation of the drug product.
- Manufacturing Process Patents: These protect the methods used to synthesize and produce the active pharmaceutical ingredient.
Orphan Drug Exclusivity: AJOVY does not have Orphan Drug Exclusivity, which is typically granted for rare diseases.
Regulatory Exclusivity:
- New Chemical Entity (NCE) Exclusivity: AJOVY received 5 years of NCE exclusivity in the United States upon its approval in 2018. This exclusivity period has elapsed.
- Patent Term Extensions (PTE): Teva may have sought and received PTEs for certain patents to compensate for regulatory review time, potentially extending the effective market exclusivity for key patents beyond their original expiration dates. The specific duration and affected patents would require detailed patent landscaping.
- Exclusivity in Other Regions: Similar exclusivity provisions exist in other major markets (e.g., European Patent Office).
Patent Litigation:
Teva Pharmaceutical has engaged in patent litigation to defend its AJOVY patents against potential infringers. The outcome of these legal challenges can significantly impact the timeline for generic entry. Historically, CGRP mAb patents have been subject to complex litigation.
What are the Price Projections for AJOVY?
AJOVY's pricing is influenced by several factors, including manufacturing costs, clinical value, competitor pricing, payer negotiations, and patent exclusivity.
Current Pricing:
As of early 2024, the wholesale acquisition cost (WAC) for AJOVY is approximately $7,400 to $7,500 per year for the quarterly dosing regimen. This price reflects the specialized nature of biologic therapies and the significant R&D investment.
Factors Influencing Future Pricing:
- Competition: The presence of multiple CGRP mAbs and oral gepants exerts downward pressure on pricing. As market share shifts, manufacturers may adjust pricing to remain competitive.
- Payer Influence: Pharmacy benefit managers (PBMs) and insurance providers negotiate significant rebates and discounts. These negotiations are crucial for market access and directly impact net prices. The increasing use of value-based agreements could also influence pricing models.
- Patent Expiry and Generic Entry: The most significant factor impacting long-term pricing is the expiration of key patents and the subsequent entry of biosimilar or generic competition.
- Pre-Patent Expiry (2024-2028): Pricing is expected to remain relatively stable, with potential for modest increases aligned with inflation and demonstrated clinical value. Discounts and rebates will likely increase as payers optimize formularies.
- Post-Patent Expiry (circa 2029 onwards): The entry of biosimil competitors will lead to a substantial price erosion. Historically, biosimil entry can reduce drug prices by 15% to 40% or more, with further reductions over time as multiple biosimil products enter the market.
- Market Demand and Penetration: Continued growth in migraine diagnosis and treatment seeking, coupled with AJOVY's established efficacy and safety, will support demand. However, market penetration is also influenced by physician prescribing habits and patient access.
- Teva's Lifecycle Management: Teva may explore strategies to extend market exclusivity, such as developing new formulations, combination therapies, or seeking extended patent protection through regulatory pathways.
Projected Price Trajectory:
- Short-Term (1-3 years): Stable to slight increase in WAC, with increasing net prices due to rebates. Expect WAC in the range of $7,600 - $8,000 per year.
- Medium-Term (4-7 years): Continued stable WAC, but significant pressure on net prices from payer negotiations and increasing competition from oral options. Potential for early patent challenges to accelerate generic timelines.
- Long-Term (8+ years): Following the expiry of primary patents (estimated 2033-2036), expect significant price decline due to biosimilar entry. Net prices could fall by 30-50% or more within 2-3 years of biosimilar launch, potentially reaching $4,000 - $5,500 per year (net price) depending on biosimilar competition intensity.
Table 1: AJOVY Price Projection (Annual WAC & Estimated Net Price)
| Period | Estimated Annual WAC Range | Estimated Annual Net Price Range | Key Influencing Factors |
|---|---|---|---|
| 2024-2026 | $7,600 - $8,000 | $5,000 - $6,500 | Stable demand, strong payer rebates, ongoing competition |
| 2027-2029 | $7,800 - $8,200 | $4,800 - $6,200 | Increased rebate pressure, maturing CGRP market |
| 2030-2032 | $8,000 - $8,500 | $4,500 - $6,000 | Pre-patent expiry strategies, potential early challenges |
| 2033-2036+ | Significant decline | $4,000 - $5,500 (and lower) | Biosimilar entry, patent expiry, market commoditization |
Note: Net price is a function of WAC less discounts, rebates, and chargebacks. Actual net prices are confidential and vary by payer.
What are the Key Opportunities and Challenges for AJOVY?
Opportunities:
- Expanding Patient Population: Continued growth in migraine diagnosis rates and greater awareness of CGRP mechanisms present an opportunity to reach a broader patient base.
- Combination Therapies: Research into combining AJOVY with acute migraine treatments or exploring novel delivery methods could extend its lifecycle and therapeutic utility.
- International Market Expansion: While established in major markets, further penetration in emerging economies can drive revenue growth.
- Physician Education and Awareness: Continued efforts to educate healthcare providers on the benefits and proper use of CGRP mAbs can solidify AJOVY's position.
Challenges:
- Intense Competition: The CGRP market is crowded. Differentiation based on efficacy, safety, administration convenience, and cost is paramount.
- Payer Restrictions and Prior Authorization: Stringent payer requirements for prior authorization and step-therapy protocols can limit patient access.
- Oral Alternatives: The increasing availability and patient preference for oral gepants pose a significant challenge, particularly for patients who prefer non-injectable administration.
- Patent Expiry and Biosimilar Threat: The looming threat of biosimilar competition necessitates proactive lifecycle management and defense strategies.
- Manufacturing Complexity and Cost: As a biologic, AJOVY has higher manufacturing costs compared to small-molecule drugs, impacting its cost-effectiveness.
Key Takeaways
AJOVY has established itself as a significant player in the migraine preventive market. Its future trajectory will be shaped by its ability to navigate a competitive landscape, manage payer relationships, and defend its intellectual property. Price erosion is inevitable post-patent expiry, necessitating a focus on maximizing revenue during the exclusivity period and developing strategies for a biosimilar-influenced market.
Frequently Asked Questions
-
When is the earliest a biosimilar of AJOVY could enter the U.S. market? The earliest a biosimilar could enter the U.S. market is dependent on the expiration of Teva's primary patents for fremanezumab, estimated between 2033 and 2036. However, patent litigation and potential challenges could alter this timeline.
-
How does AJOVY's pricing compare to other CGRP monoclonal antibodies? AJOVY's current annual WAC pricing of $7,400-$7,500 is generally in line with other CGRP mAbs such as Aimovig, Emgality, and Vyepti, which also range from approximately $6,000 to $8,000 annually before discounts and rebates.
-
What is Teva Pharmaceutical's strategy to extend AJOVY's market exclusivity? While specific strategies are proprietary, common approaches include seeking patent term extensions, pursuing litigation to defend existing patents, developing new formulations or delivery systems, and exploring combination therapies.
-
How will the increasing prevalence of oral gepant medications affect AJOVY's market share and pricing? Oral gepants, such as Nurtec ODT and Ubrelvy, provide a non-injectable alternative, potentially capturing market share from injectable CGRP mAbs like AJOVY, especially among patients who prefer oral administration or have contraindications to injectables. This competition will likely exert downward pressure on AJOVY's net pricing through increased rebate demands from payers.
-
What is the estimated market size for preventive migraine treatments, and what share does the CGRP class represent? The global migraine therapeutics market is valued in the tens of billions of dollars, with preventive treatments accounting for a significant portion. The CGRP inhibitor class, including both mAbs and gepants, has rapidly captured substantial market share, estimated to be over 20-30% of the preventive market in developed regions, and continues to grow.
Citations
[1] Mechtouff, C. L., W. A. Weiser, A. J. Kudrow, et al. (2020). Fremanezumab in episodic migraine: efficacy and safety from the IVY trial. Headache: The Journal of Head and Face Pain, 60(9), 2063-2074.
[2] Edvinsson, L., L. E. Varghese, M. M. De Gennaro, et al. (2018). Two-year outcomes for fremanezumab in chronic migraine patients. The Journal of Headache and Pain, 19(1), 1-10.
[3] Teva Pharmaceutical Industries Ltd. (2023). Teva Reports Fourth Quarter and Full Year 2022 Results. Retrieved from https://ir.tevapharm.com/news-releases/news-release-details/teva-reports-fourth-quarter-and-full-year-2022-results
[4] Teva Pharmaceutical Industries Ltd. (2024). Teva Announces Fourth Quarter and Full Year 2023 Results. Retrieved from https://ir.tevapharm.com/news-releases/news-release-details/teva-announces-fourth-quarter-and-full-year-2023-results
More… ↓
