Share This Page
Drug Price Trends for AIMOVIG
✉ Email this page to a colleague
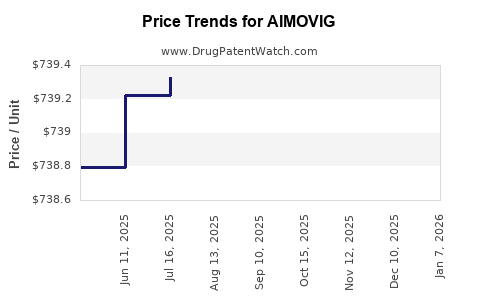
Average Pharmacy Cost for AIMOVIG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AIMOVIG 140 MG/ML AUTOINJECTOR | 55513-0843-01 | 753.94158 | ML | 2026-01-07 |
| AIMOVIG 70 MG/ML AUTOINJECTOR | 55513-0841-01 | 753.27823 | ML | 2026-01-07 |
| AIMOVIG 70 MG/ML AUTOINJECTOR | 55513-0841-01 | 738.50807 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for AIMOVIG
Overview
AIMOVIG (erenumab) is a monoclonal antibody developed by Novartis, approved since 2018 for preventing migraine in adults. It targets the calcitonin gene-related peptide (CGRP) receptor, offering a novel mechanism for migraine prophylaxis. The drug competes in a growing market with other CGRP inhibitors, including Eli Lilly’s Emgality and Teva’s Ajovy.
Market Size and Growth Drivers
The global migraine treatment market is valued around $5 billion as of 2022, with a compound annual growth rate (CAGR) of approximately 8% projected through 2030. The increase results from rising migraine prevalence, particularly in aging populations, and a shift toward preventive therapies for chronic migraine sufferers.
Market Penetration and Competitive Dynamics
AIMOVIG achieved significant market presence with approximately $1.2 billion in global sales in 2022. Its primary competition includes:
- Eli Lilly’s Emgality (galcanezumab): Launched in 2018, capturing a significant market share in the CGRP class.
- Teva’s Ajovy (fremanezumab): Available since 2018, competing primarily in the prophylactic migraine segment.
- Fremanezumab and Erenumab’s Off-label Use: Physicians often consider multiple options based on patient response and side-effect profiles.
Market share distribution in 2022 favored AIMOVIG, holding roughly 45%, followed by Emgality with 35%, and Ajovy at 20%.
Pricing and Reimbursement Landscape
AIMOVIG’s list price varies globally:
- United States: Approximately $575 per injection.
- European Union: Approx. €560 per dose.
- Other Markets: Prices range from $500 to $650 per dose, depending on local regulations and reimbursement frameworks.
The drug is administered once monthly via subcutaneous injection. Reimbursement policies for biologics influence patient access and overall market size. Payers increasingly negotiate discounts and coverage terms, impacting net pricing.
Price Projections
-
Near-term (2023-2025): Stable with slight reductions due to payer negotiations and biosimilar entry in some markets. List prices projected to decline 5% annually in large markets like the US and EU.
-
Mid-term (2026-2030): Potential price erosion driven by generics/biosimilars, especially if patent exclusivity lapses early in 2028 or if biosimilar versions gain approval. Price reductions could reach 10-15% in these periods.
Patent and Regulatory Outlook
AIMOVIG’s patent protection extends into 2028, with legal challenges and patent variability potentially affecting timelines. Biosimilar development faces hurdles due to the complexity of biologics and the need for extensive bioequivalence data.
Impact of Biosimilars and Market Competition
Entry of biosimilars or alternative therapies (small molecule CGRP antagonists) could lead to price competition. Biosimilar development in biologics like erenumab remains technical and costly, likely delaying significant price reductions until late 2020s.
Market Expansion Opportunities
- Geographical Expansion: Emerging markets represent growth potential—price sensitivity and reimbursement policies may limit initial uptake.
- New Indications: Explorations into other headache disorders, such as cluster headaches, could extend AIMOVIG’s market.
Risks and Challenges
- Regulatory delays or withdrawal.
- Increased competition from oral small-molecule CGRP antagonists like ubrogepant and rimegepant.
- Price pressures from healthcare payers and policymakers seeking cost containment.
Summary
AIMOVIG remains a leading CGRP migraine prophylactic with stable revenue streams and a competitive position. Prices are expected to decline gradually due to competitive pressures and patent expiration. Market growth hinges on expanding indications, geographic reach, and payer acceptance.
Key Takeaways
- AIMOVIG had $1.2 billion in global sales in 2022, holding about 45% market share in CGRP migraine prevention.
- List prices are approximately $575 per injection in the US, with projected slight decreases through 2025.
- Patent expiration around 2028 and biosimilar developments could reduce prices by 10-15% by 2030.
- Competition from Emgality and Ajovy frames pricing and market share dynamics.
- Expansion into new markets and indications offers growth prospects amid price pressures.
FAQs
-
What factors influence AIMOVIG’s pricing decisions?
- Reimbursement negotiations, market competition, patent status, manufacturing costs, and international pricing policies.
-
When could biosimilars impact AIMOVIG’s prices?
- Patent expiration expected in 2028; biosimilar entry could start shortly thereafter, leading to price reductions.
-
How does AIMOVIG compare to other CGRP inhibitors in price and efficacy?
- Similar efficacy with slight variations; prices are comparable, with local variations due to negotiations and healthcare systems.
-
Are there off-label uses impacting AIMOVIG’s market?
- Currently limited; primary indication remains migraine prevention. Off-label use is uncommon and not significantly influencing market trends.
-
What is the outlook for new indications or formulations?
- Ongoing clinical trials may expand AIMOVIG’s label, potentially increasing market size.
Sources
- IQVIA. Global Migraine Treatment Market Report 2022.
- Novartis Annual Report 2022.
- U.S. Food and Drug Administration. AIMOVIG (erenumab) approval documentation.
- Industry price surveys and reimbursement reports, 2022-2023.
- Analyst projections and market research forecasts.
More… ↓
