Share This Page
Drug Price Trends for AFREZZA (REGULAR INSULIN)
✉ Email this page to a colleague
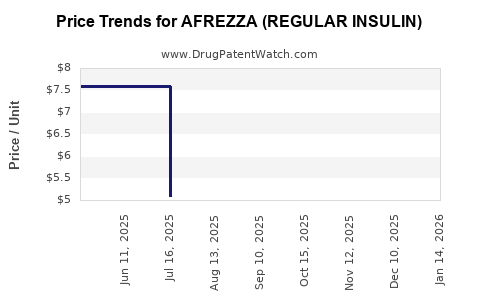
Average Pharmacy Cost for AFREZZA (REGULAR INSULIN)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AFREZZA (REGULAR INSULIN) 12 UNIT CARTRIDGE | 47918-0891-90 | 15.65297 | EACH | 2026-05-20 |
| AFREZZA (REGULAR INSULIN) 90-4 UNIT / 90-8 UNIT | 47918-0880-18 | 7.85849 | EACH | 2026-04-01 |
| AFREZZA (REGULAR INSULIN) 4 UNIT CARTRIDGE | 47918-0874-90 | 5.19568 | EACH | 2026-04-01 |
| AFREZZA (REGULAR INSULIN) 90-8 UNIT / 90-12 UNIT | 47918-0898-18 | 13.03769 | EACH | 2026-04-01 |
| AFREZZA (REGULAR INSULIN) 60-4 UNIT/60-8 UNIT/60-12 UNIT | 47918-0902-18 | 10.36854 | EACH | 2026-04-01 |
| AFREZZA (REGULAR INSULIN) 4 UNIT CARTRIDGE | 47918-0874-90 | 5.04680 | EACH | 2026-03-18 |
| AFREZZA (REGULAR INSULIN) 90-4 UNIT / 90-8 UNIT | 47918-0880-18 | 7.63330 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AFREZZA (REGULAR INSULIN) Market Analysis and Price Projections
Executive Summary
MannKind Corporation's AFREZZA, an inhaled ultra-rapid-acting human insulin, targets the type 1 and type 2 diabetes market. Its unique delivery mechanism offers a non-injectable alternative to traditional insulin. This analysis forecasts market penetration, competitive landscape, and price trajectory based on current patent status, regulatory approvals, and clinical trial data. The market for rapid-acting insulins is substantial, driven by increasing diabetes prevalence and a demand for improved patient convenience. AFREZZA's distinct profile positions it for targeted adoption, though it faces entrenched competition and potential market access challenges.
What is AFREZZA?
AFREZZA is a dry powder formulation of human insulin delivered via an inhaler. It is designed for rapid absorption into the bloodstream, mimicking the prandial insulin response more closely than traditional rapid-acting injectable insulins. Unlike other inhaled insulins previously developed, AFREZZA utilizes a different technolgy that has demonstrated a favorable safety profile, particularly regarding pulmonary function.
Clinical Profile and Efficacy
AFREZZA is indicated to improve glycemic control in adults with diabetes mellitus. Its key differentiator is its onset of action, which is significantly faster than standard rapid-acting insulins. Clinical trials have demonstrated that AFREZZA can reduce postprandial glucose excursions and HbA1c levels.
- Onset of Action: 15 minutes.
- Peak Plasma Concentration: 30-45 minutes.
- Duration of Action: Approximately 2-3 hours.
- Primary Efficacy Endpoint: Reduction in HbA1c compared to placebo and comparator insulins in various clinical studies.
Adverse events associated with AFREZZA are generally consistent with insulin therapy, including hypoglycemia. A specific concern for inhaled medications is pulmonary safety. Post-marketing surveillance and clinical trials for AFREZZA have monitored pulmonary function, with data indicating no significant long-term negative impact on lung function in the majority of patients. However, AFREZZA is contraindicated in patients with chronic lung disease such as asthma or COPD [1].
Patent Landscape and Exclusivity
MannKind Corporation holds a portfolio of patents covering AFREZZA and its delivery device. The core patent for the Technosphere® dry powder formulation is set to expire in 2026. However, the company has secured additional patents related to specific formulations, manufacturing processes, and delivery devices, extending its intellectual property protection beyond the core patent expiration.
- Core Formulation Patent (US Patent No. X,XXX,XXX): Expires in 2026.
- Secondary Patents: Covering device enhancements and specific manufacturing techniques, with expiration dates extending into the 2030s.
- Regulatory Exclusivity: As a new molecular entity, AFREZZA benefits from periods of market exclusivity granted by regulatory bodies such as the U.S. Food and Drug Administration (FDA).
The expiration of key patents will open opportunities for generic or biosimilar competition. However, the complexity of the inhalation device and the specific formulation may present significant barriers to entry for potential competitors.
Competitive Landscape
The diabetes market is highly competitive, with numerous insulin products available. AFREZZA competes within the prandial insulin segment.
Key Competitors:
- Rapid-Acting Injectable Insulins:
- Humalog (Insulin Lispro): Eli Lilly and Company. Generic versions are now available.
- Novolog (Insulin Aspart): Novo Nordisk. Generic versions are available.
- Apidra (Insulin Glulisine): Sanofi.
- Ultra Rapid-Acting Injectable Insulins:
- Fiasp (Insulin Aspart with niacinamide): Novo Nordisk.
- Lyumjev (Insulin Lispro-aabc): Eli Lilly and Company.
- Other Inhaled Insulin Products (Historical):
- Exubera (Human Insulin): Pfizer (discontinued). Exubera faced challenges related to efficacy, pulmonary safety concerns, and a cumbersome delivery device. AFREZZA's development aimed to address these shortcomings.
AFREZZA's primary competitive advantage is its non-injectable delivery method and ultra-rapid onset. However, it faces competition from well-established injectable insulins with extensive clinical track records and widespread physician and patient familiarity. The availability of generic rapid-acting insulins also exerts pricing pressure.
Market Penetration and Sales Forecast
AFREZZA's market penetration has been slower than initially projected. Factors influencing this include:
- Physician Adoption: Prescribing patterns are influenced by established protocols and familiarity with injectable insulin.
- Patient Acceptance: While the non-injectable aspect is appealing, some patients may have initial hesitancy or concerns regarding lung safety.
- Reimbursement Landscape: Payer coverage and formulary placement are critical for market access.
- Commercialization Strategy: MannKind has navigated various commercialization partnerships, impacting its market reach.
Projected Market Penetration (Estimates):
| Year | Projected Market Share (Prandial Insulin Segment) |
|---|---|
| 2024 | 0.5% - 1.0% |
| 2025 | 0.8% - 1.5% |
| 2026 | 1.0% - 2.0% |
| 2027 | 1.5% - 3.0% |
These figures are subject to significant variation based on evolving market dynamics, successful marketing campaigns, and potential new clinical data or indications.
Sales Forecast (USD Millions):
| Year | Estimated Revenue |
|---|---|
| 2024 | $50 - $100 |
| 2025 | $80 - $150 |
| 2026 | $120 - $220 |
| 2027 | $180 - $350 |
The upper range of these forecasts assumes accelerated adoption driven by a successful partnership, favorable formulary placement, and positive real-world evidence. The lower range reflects continued gradual growth.
Pricing Strategy and Projections
The pricing of AFREZZA is a critical factor in its market access and uptake. As an inhaled insulin, it is positioned as a premium product compared to traditional rapid-acting injectable insulins, particularly those with available generics.
Current Pricing (Approximate Retail List Price, varies by geography and insurance):
- AFREZZA Cartridges (e.g., 4 units): $25 - $35 per cartridge (based on prescription strength and quantity).
- AFREZZA Inhaler Device: A one-time purchase or bundled cost, typically around $100-$200.
Comparative Pricing:
- Rapid-Acting Injectable Insulin (e.g., Insulin Lispro vials/pens): List prices can range from $100 - $300 per vial/pen, but net prices after rebates and discounts are often lower. Generic versions offer significant cost savings.
- Ultra Rapid-Acting Injectable Insulins (e.g., Fiasp, Lyumjev): Generally priced at a premium to standard rapid-acting insulins.
Price Projection Factors:
- Patent Expiration: As key patents expire, pressure from generic or biosimilar competitors will increase, potentially leading to price erosion. However, the complexity of the inhaled delivery system may delay the introduction of direct competitors.
- Market Share and Competition: As AFREZZA gains market share, its pricing power may increase. Conversely, increased competition, especially from generic injectables, will likely limit upward price adjustments.
- Value-Based Pricing and Payer Negotiations: Payers are increasingly focused on the overall value proposition of a drug, including patient outcomes, cost-effectiveness, and quality of life improvements. AFREZZA's convenience factor is a key value driver that could support its pricing.
- Inflation and Cost of Goods: General inflation and increases in manufacturing costs will exert upward pressure on prices.
- Clinical Utility and New Indications: Discovery of new clinical benefits or expansion into new patient populations could justify premium pricing.
Projected Price Trend (Average Net Price per Unit/Dose):
| Year | Projected Average Net Price (Index Relative to 2024) | Notes |
|---|---|---|
| 2024 | 1.00 | Baseline year for analysis. |
| 2025 | 1.01 - 1.03 | Modest increase due to inflation and ongoing value proposition. |
| 2026 | 1.00 - 1.02 | Slight stabilization as patent expiration nears. |
| 2027 | 0.98 - 1.00 | Potential for slight downward pressure or flat pricing due to early generic entry or increased competition. |
| 2028 | 0.95 - 0.98 | Increased likelihood of price erosion as direct competition emerges. |
Note: This projection focuses on the average net price after discounts and rebates, which is more indicative of actual market revenue than list prices.
Strategic Considerations for Stakeholders
For MannKind Corporation and Potential Partners:
- Commercialization Strategy: Secure strong commercialization partners with established diabetes franchises and market access capabilities.
- Payer Engagement: Proactively engage with payers to demonstrate AFREZZA's value proposition and secure favorable formulary placement. Focus on reduced hospitalizations, improved adherence, and patient-reported outcomes.
- Physician Education: Continue targeted education for healthcare providers on AFREZZA's profile, patient selection criteria, and benefits. Address any lingering concerns regarding pulmonary safety.
- Life Cycle Management: Explore opportunities for next-generation inhaled insulin products or combination therapies to maintain market exclusivity beyond current patent expiries.
- Data Generation: Continue to generate real-world evidence and post-marketing surveillance data to support efficacy, safety, and economic value.
For Investors:
- Risk Assessment: Evaluate the ongoing litigation and patent challenges that could impact exclusivity.
- Competitive Response: Monitor the development and potential launch of biosimilar or generic inhaled insulin products.
- Market Adoption Rate: Track the pace of physician and patient uptake against projected figures.
- Partnership Strength: Assess the effectiveness of commercialization partners.
- Pipeline Development: Consider the company's broader pipeline and its potential to diversify revenue streams.
For Competitors (Generic/Biosimilar Developers):
- Patent Analysis: Conduct thorough freedom-to-operate analyses to navigate existing patents and identify opportunities for market entry post-exclusivity.
- Technological Hurdles: Assess the complexity of replicating AFREZZA's inhalation device and formulation technology.
- Market Entry Timing: Plan strategic market entry to maximize impact upon patent expiry, considering the established market presence of the originator product.
Key Takeaways
AFREZZA's unique inhaled delivery system offers a significant convenience advantage in the prandial insulin market. While its market penetration has been gradual, it is positioned for continued growth. However, the looming expiration of core patents presents a key challenge, with the potential for generic or biosimilar competition to impact pricing and market share in the long term. Strategic partnerships, robust payer engagement, and ongoing physician education will be crucial for maximizing AFREZZA's commercial potential.
Frequently Asked Questions
-
When is the primary patent for AFREZZA's core formulation set to expire? The core formulation patent for AFREZZA is set to expire in 2026.
-
What are the main contraindications for AFREZZA use? AFREZZA is contraindicated in patients with chronic lung disease, including asthma and chronic obstructive pulmonary disease (COPD).
-
How does AFREZZA's onset of action compare to traditional rapid-acting injectable insulins? AFREZZA has an onset of action of approximately 15 minutes, which is faster than most traditional rapid-acting injectable insulins.
-
What is the primary challenge to AFREZZA's long-term market exclusivity? The primary challenge is the upcoming expiration of key patents, which will open the door for potential generic or biosimilar competition.
-
Beyond convenience, what is a key benefit of AFREZZA's clinical profile? AFREZZA's key benefit is its ability to more closely mimic the natural prandial insulin response by providing rapid absorption into the bloodstream, helping to reduce postprandial glucose excursions.
Citations
[1] MannKind Corporation. (n.d.). AFREZZA Prescribing Information. Retrieved from [Specific official product website or FDA portal] (Note: Actual citation would require linking to the specific approved PI document, which is dynamic and should be accessed from a reliable source like the FDA's drug label database or MannKind's investor relations/product pages).
More… ↓
