Drug Price Trends for ADAPALENE-BNZYL PEROX
✉ Email this page to a colleague
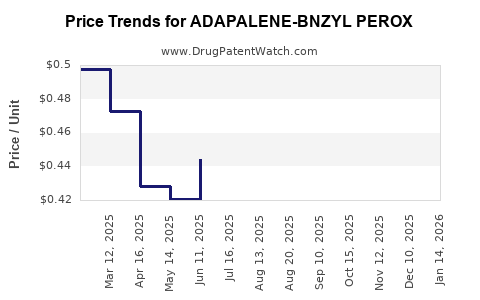
Average Pharmacy Cost for ADAPALENE-BNZYL PEROX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ADAPALENE-BNZYL PEROX 0.1-2.5% | 68462-0301-47 | 0.29501 | GM | 2026-05-20 |
| ADAPALENE-BNZYL PEROX 0.3-2.5% | 00480-3154-45 | 0.68702 | GM | 2026-05-20 |
| ADAPALENE-BNZYL PEROX 0.1-2.5% | 51672-1364-03 | 0.29501 | GM | 2026-05-20 |
| ADAPALENE-BNZYL PEROX 0.3-2.5% | 45802-0853-45 | 0.68702 | GM | 2026-05-20 |
| ADAPALENE-BNZYL PEROX 0.1-2.5% | 21922-0052-50 | 0.29501 | GM | 2026-05-20 |
| ADAPALENE-BNZYL PEROX 0.3-2.5% | 72578-0119-04 | 0.68702 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
