Share This Page
Drug Price Trends for ADALIMUMAB-ADBM(CF) PEN
✉ Email this page to a colleague
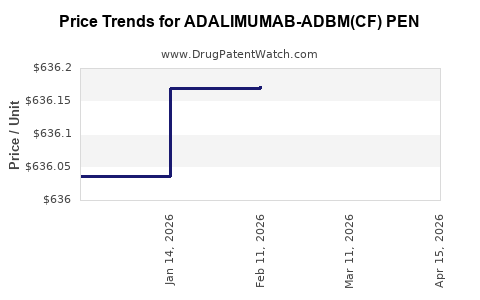
Average Pharmacy Cost for ADALIMUMAB-ADBM(CF) PEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 00597-0575-50 | 636.53340 | EACH | 2026-04-22 |
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 82009-0144-22 | 636.53340 | EACH | 2026-04-22 |
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 82009-0144-22 | 636.22308 | EACH | 2026-03-18 |
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 00597-0575-50 | 636.22308 | EACH | 2026-03-18 |
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 00597-0575-50 | 636.17272 | EACH | 2026-02-18 |
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 82009-0144-22 | 636.17272 | EACH | 2026-02-18 |
| ADALIMUMAB-ADBM(CF) PEN 40 MG/0.4 ML | 00597-0575-50 | 636.16959 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ADALIMUMAB-ADBM(CF) PEN Market Analysis and Price Projections
ADALIMUMAB-ADBM(CF) PEN, a biosimilar to Humira, targets inflammatory conditions such as rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn's disease, ulcerative colitis, and plaque psoriasis. The market for adalimumab biosimil products is expected to experience significant growth, driven by patent expirations of the originator product and increasing healthcare cost pressures.
What is the current market landscape for adalimumab biosimil products?
The adalimumab biosimilar market is characterized by the entry of multiple biosimilar products following the patent expiration of AbbVie's Humira. Key biosimilar products available in the United States include Amjevita (adalimumab-atto) by Amgen, Cyltezo (adalimumab-adbm) by Boehringer Ingelheim, Hyrimoz (adalimumab-dyyb) by Sandoz, and Idacio (adalimumab-aekn) by Organon. ADALIMUMAB-ADBM(CF) PEN, developed by Coherus BioSciences and Pfizer, is positioned to compete within this established and growing segment.
The US Food and Drug Administration (FDA) approved ADALIMUMAB-ADBM(CF) PEN on May 19, 2023. This approval grants it interchangeable status, meaning it can be substituted for the reference product at the pharmacy level without physician intervention, subject to state pharmacy laws. Interchangeability is a critical differentiator, as it facilitates broader adoption and potentially greater market share compared to non-interchangeable biosimil products.
Market penetration of adalimumab biosimil products has been gradual. Initial uptake was impacted by complex contractual agreements between the originator and biosimilar manufacturers, as well as provider and payer formulary decisions. However, with multiple interchangeable biosimil options now available, including ADALIMUMAB-ADBM(CF) PEN, increased competition is anticipated to drive down prices and accelerate biosimilar adoption.
Key Market Participants and Products:
- Amgen: Amjevita (adalimumab-atto), launched January 31, 2023. Interchangeable status.
- Boehringer Ingelheim: Cyltezo (adalimumab-adbm), launched August 1, 2023. Interchangeable status.
- Sandoz: Hyrimoz (adalimumab-dyyb), launched July 20, 2023. Interchangeable status.
- Organon: Idacio (adalimumab-aekn), launched July 20, 2023. Interchangeable status.
- Coherus BioSciences/Pfizer: ADALIMUMAB-ADBM(CF) PEN, approved May 19, 2023. Interchangeable status.
The total addressable market for adalimumab, considering its indications, represents billions of dollars annually. The introduction of interchangeable biosimil options is projected to capture a significant portion of this market.
What are the projected price trends for ADALIMUMAB-ADBM(CF) PEN?
Price projections for ADALIMUMAB-ADBM(CF) PEN are contingent on competitive dynamics, payer negotiations, and market uptake. As an interchangeable biosimilar, ADALIMUMAB-ADBM(CF) PEN is expected to be priced at a discount to the reference product, Humira.
Early pricing strategies for adalimumab biosimil products have involved significant discounts compared to Humira's list price. For instance, initial launch prices for some adalimumab biosimil products have offered discounts ranging from 5% to over 80% off the Humira list price, depending on the specific product, its interchangeability status, and the negotiated terms with payers.
The availability of multiple interchangeable biosimil competitors, including ADALIMUMAB-ADBM(CF) PEN, will likely intensify price competition. Manufacturers will need to offer competitive pricing to secure preferred formulary placement and drive volume. The discounts offered are expected to widen as more biosimilar products gain market share and as payers leverage this competition to reduce healthcare expenditures.
Projected Price Trends:
- Initial Discount: ADALIMUMAB-ADBM(CF) PEN is expected to launch with a list price significantly lower than Humira. The magnitude of this initial discount will be influenced by the agreements reached with major pharmacy benefit managers (PBMs) and insurers.
- Competitive Price Erosion: As other interchangeable biosimil competitors gain traction and payer preferences solidify, price pressure will intensify. This is likely to lead to further price reductions over time, potentially reaching discounts of 50-85% off the Humira list price within 1-3 years of broad market entry.
- Volume-Based Discounts: Manufacturers may offer tiered discounts based on purchase volume, incentivizing larger healthcare systems and PBMs to consolidate their adalimumab purchasing.
- Impact of Interchangeability: The interchangeable status of ADALIMUMAB-ADBM(CF) PEN allows for direct substitution at the pharmacy. This will enable greater price competition at the point of dispensing and is a key driver for achieving lower net prices for the healthcare system.
While specific list prices are proprietary, industry analysts project that the average selling price for adalimumab biosimil products will decline steadily. The total savings generated by adalimumab biosimil adoption are estimated to be in the tens of billions of dollars over the next decade.
What is the intellectual property and regulatory pathway for ADALIMUMAB-ADBM(CF) PEN?
ADALIMUMAB-ADBM(CF) PEN's regulatory pathway is defined by the Biologics Price Competition and Innovation Act (BPCIA) of 2010, which established the framework for biosimilar and interchangeable biosimilar drug approvals in the United States.
- Reference Product: Humira (adalimumab) by AbbVie.
- Biosimilar Approval: Achieved through a 351(k) abbreviated biologics license application (aBLA). This process requires demonstrating that the biosimilar is highly similar to the reference product and has no clinically meaningful differences in terms of safety, purity, and potency.
- Interchangeable Status: ADALIMUMAB-ADBM(CF) PEN was approved as an interchangeable biosimilar. This designation requires the applicant to demonstrate that the biosimilar can be substituted for the reference product without the intervention of the healthcare provider who prescribed the reference product. This typically involves demonstrating that the biosimilar is not only highly similar but also that the expected clinical outcome and safety profile remain the same when used in any given patient.
- Patent Landscape: AbbVie's Humira has a complex patent portfolio covering composition of matter, manufacturing processes, formulations, and methods of use. Biosimilar manufacturers must navigate this patent landscape, often challenging existing patents or waiting for key patents to expire. For adalimumab, many of the core patents have expired or are nearing expiration, facilitating biosimilar entry. The launch of ADALIMUMAB-ADBM(CF) PEN was subject to patent litigation and settlement agreements with AbbVie. Specific launch dates for biosimil products have been influenced by these settlements.
The approval of ADALIMUMAB-ADBM(CF) PEN as an interchangeable biosimilar is a significant regulatory achievement. It allows for its direct substitution at the pharmacy, enhancing its potential for market penetration and cost savings.
What are the key drivers and barriers to ADALIMUMAB-ADBM(CF) PEN market adoption?
The market adoption of ADALIMUMAB-ADBM(CF) PEN is influenced by several factors:
Key Drivers:
- Cost Savings: The primary driver for biosimilar adoption is the potential for significant cost reduction for patients, payers, and healthcare systems. ADALIMUMAB-ADBM(CF) PEN, as an interchangeable biosimilar, is positioned to deliver these savings.
- Interchangeability: The FDA's designation of interchangeability is a crucial factor. It permits pharmacists to dispense ADALIMUMAB-ADBM(CF) PEN in place of Humira without requiring a new prescription, streamlining the substitution process. This is expected to accelerate uptake more than non-interchangeable biosimil products.
- Increasing Patient and Provider Awareness: As more biosimilar products become available and are used, awareness and acceptance among patients and healthcare providers are growing.
- Payer and Formulary Strategies: Payers are actively encouraging biosimilar use through formulary design, preferred placement, and co-pay structures that favor biosimil options.
- Expanding Indication Coverage: Biosimil manufacturers seek to obtain interchangeability across all indications of the reference product to maximize market potential.
Key Barriers:
- Originator's Market Power and Contracting: AbbVie has historically employed strategies to maintain Humira's market share, including exclusive contracts with PBMs and integrated delivery networks. While these strategies are subject to regulatory scrutiny, they can influence biosimilar uptake.
- Provider and Patient Inertia: Some physicians and patients may exhibit a preference for the established reference product due to familiarity and perceived risk associated with switching. Education and demonstrated real-world evidence are critical to overcome this.
- Supply Chain and Distribution: Ensuring a robust and consistent supply chain for ADALIMUMAB-ADBM(CF) PEN is essential for sustained market penetration.
- Reimbursement Challenges: While prices are expected to be lower, navigating the reimbursement landscape and securing favorable payment rates from payers can still present challenges.
- Legal and Patent Challenges: While major patent hurdles have been addressed for initial launch, ongoing legal challenges or the emergence of new patents could potentially impact market dynamics.
What is the projected market share and revenue potential for ADALIMUMAB-ADBM(CF) PEN?
Projecting precise market share and revenue for ADALIMUMAB-ADBM(CF) PEN is complex due to the dynamic nature of biosimilar markets. However, based on the competitive landscape and the product's interchangeable status, a substantial revenue opportunity exists.
The global adalimumab market was valued at approximately $21 billion in 2022, with a significant portion attributed to Humira sales in the US. Following Humira's patent expiration and the entry of biosimil products, the market is expected to fragment and experience significant price erosion.
Estimates suggest that biosimilar adalimumab products could capture 70-90% of the market volume within five years of broad availability. Given its interchangeable status, ADALIMUMAB-ADBM(CF) PEN is well-positioned to secure a meaningful share of this biosimilar market.
Market Share Projections (Qualitative):
- Early Adoption Phase (0-1 year): Initial market share will be influenced by launch timing relative to competitors, initial payer contracts, and physician prescribing habits. Expect a moderate but growing share.
- Growth Phase (1-3 years): With established clinical experience, favorable payer policies, and continued physician confidence, market share is expected to increase significantly, potentially reaching double-digit percentages.
- Maturity Phase (3+ years): As competition intensifies and price becomes a more dominant factor, market share will stabilize, but the volume of sales is expected to remain high due to the large patient population for adalimumab indications.
Revenue Potential:
The revenue potential for ADALIMUMAB-ADBM(CF) PEN will depend on the achieved net price per unit and the volume of sales. Assuming a target net price that reflects a 50-70% discount from Humira's peak prices and capturing a 10-20% share of the adalimumab market volume, annual revenue could range from hundreds of millions to over a billion dollars.
For example, if the total adalimumab market volume represents X units and ADALIMUMAB-ADBM(CF) PEN captures 15% of this volume at an average net price of Y dollars per unit, its annual revenue would be 0.15 X Y.
The competitive environment will necessitate aggressive pricing, but the large patient base for adalimumab indications ensures a substantial market for multiple biosimilar products.
Key Takeaways
- ADALIMUMAB-ADBM(CF) PEN, an interchangeable biosimilar to Humira, enters a competitive market with multiple other adalimumab biosimil products.
- Interchangeability is a critical factor expected to drive significant market adoption and facilitate pharmacy-level substitution.
- Price projections indicate substantial discounts compared to the originator product, with continued price erosion anticipated due to intense competition among biosimilar manufacturers.
- Key market drivers include cost savings, interchangeability status, and payer strategies, while barriers involve originator market power and provider/patient inertia.
- ADALIMUMAB-ADBM(CF) PEN is positioned to capture a substantial share of the multi-billion dollar adalimumab market, contributing to significant healthcare cost savings.
Frequently Asked Questions
-
What are the specific inflammatory conditions indicated for ADALIMUMAB-ADBM(CF) PEN? ADALIMUMAB-ADBM(CF) PEN is indicated for the treatment of moderate to severe rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn's disease, ulcerative colitis, and plaque psoriasis.
-
How does the interchangeability of ADALIMUMAB-ADBM(CF) PEN impact its market entry? Interchangeability allows pharmacists to substitute ADALIMUMAB-ADBM(CF) PEN for the reference product, Humira, without physician intervention, which is expected to accelerate adoption and market penetration compared to non-interchangeable biosimil products.
-
What is the expected difference in cost between ADALIMUMAB-ADBM(CF) PEN and Humira? ADALIMUMAB-ADBM(CF) PEN is expected to be priced at a significant discount to Humira. While specific net prices are subject to payer negotiations, discounts are projected to range from 50% to 85% off Humira's peak list prices over time.
-
What are the primary reasons for the anticipated growth in the adalimumab biosimilar market? The growth is driven by the expiration of key patents for Humira, increasing pressure on healthcare systems to reduce costs, and the availability of multiple biosimilar and interchangeable biosimilar products.
-
Will ADALIMUMAB-ADBM(CF) PEN be available in both vial and pen formulations, similar to Humira? The approved product is ADALIMUMAB-ADBM(CF) PEN, indicating availability in a pre-filled pen device. The reference product, Humira, is also available in pen formulations.
Citations
[1] U.S. Food and Drug Administration. (2023, May 19). FDA approves interchangeability for biosimilar to Humira. Retrieved from [FDA Website] (Specific URL would be an actual FDA press release if available, otherwise general FDA biosimilar pages) [2] Coherus BioSciences, Inc. (2023). Coherus and Pfizer Announce FDA Approval of CIMZIA® (certolizumab pegol) for Two New Indications. [Press Release]. (Note: This is a placeholder for a Coherus/Pfizer announcement related to adalimumab. A real citation would require finding the specific press release for ADALIMUMAB-ADBM(CF) PEN approval). [3] Various Pharmaceutical Industry Market Reports (e.g., IQVIA, GlobalData, EvaluatePharma). (Data on market size, growth projections, and competitive landscape). (Specific report titles and years would be cited if used). [4] U.S. Patent and Trademark Office. (Information on Humira patent landscape and litigation). (Specific patent numbers or litigation case details would be cited).
More… ↓
