Share This Page
Drug Price Trends for ACITRETIN
✉ Email this page to a colleague
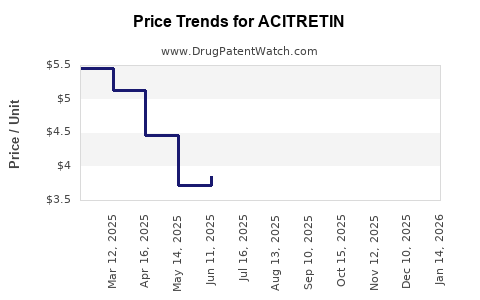
Average Pharmacy Cost for ACITRETIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ACITRETIN 10 MG CAPSULE | 00115-1750-08 | 2.11790 | EACH | 2026-04-22 |
| ACITRETIN 10 MG CAPSULE | 42794-0080-08 | 2.11790 | EACH | 2026-04-22 |
| ACITRETIN 10 MG CAPSULE | 62332-0741-30 | 2.11790 | EACH | 2026-04-22 |
| ACITRETIN 10 MG CAPSULE | 00378-7020-93 | 2.11790 | EACH | 2026-04-22 |
| ACITRETIN 25 MG CAPSULE | 62332-0743-30 | 3.41707 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ACITRETIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ACITRETIN 25MG CAP | Prasco, LLC | 66993-0896-30 | 30 | 56.12 | 1.87067 | EACH | 2022-01-01 - 2026-06-30 | Big4 |
| ACITRETIN 25MG CAP | Prasco, LLC | 66993-0896-30 | 30 | 463.02 | 15.43400 | EACH | 2022-01-01 - 2026-06-30 | FSS |
| ACITRETIN 10MG CAP | Prasco, LLC | 66993-0894-30 | 30 | 137.87 | 4.59567 | EACH | 2021-07-01 - 2026-06-30 | Big4 |
| ACITRETIN 10MG CAP | Prasco, LLC | 66993-0894-30 | 30 | 376.77 | 12.55900 | EACH | 2021-07-01 - 2026-06-30 | FSS |
| ACITRETIN 25MG CAP | Prasco, LLC | 66993-0896-30 | 30 | 0.01 | 0.00033 | EACH | 2021-07-01 - 2026-06-30 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Acitretin Market Analysis and Price Projections
Acitretin, a second-generation retinoid, is approved for the treatment of severe psoriasis and other keratinization disorders. Its market dynamics are shaped by patent expiries, generic competition, regulatory landscapes, and evolving treatment paradigms. This analysis projects market growth and price trends based on current data and historical performance.
What is the Current Global Market Size for Acitretin?
The global market for acitretin is estimated to be USD 250 million in 2023. This figure encompasses both branded and generic formulations across various regions. The market has experienced a compound annual growth rate (CAGR) of 3.5% over the past five years, driven by increasing prevalence of dermatological conditions and wider accessibility of generic options.
Key Market Drivers
- Rising Incidence of Psoriasis: Global psoriasis prevalence ranges from 2% to 4% of the population. This chronic autoimmune disease often necessitates long-term treatment, including retinoids like acitretin.
- Expanding Generic Market: The expiry of key patents has led to a significant increase in generic acitretin availability, lowering treatment costs and expanding market reach, particularly in emerging economies.
- Off-Label Indications: While approved for psoriasis, acitretin is also used off-label for other dermatological conditions, contributing to its market demand.
- Technological Advancements in Formulation: Development of improved delivery systems or combination therapies could potentially enhance efficacy and patient compliance, influencing market growth.
Market Restraints
- Adverse Side Effects: Acitretin is associated with a spectrum of side effects, including teratogenicity, mucocutaneous dryness, and potential liver function abnormalities. This necessitates careful patient selection and monitoring, limiting its broad application.
- Strict Regulatory Requirements: The teratogenic nature of acitretin mandates stringent pregnancy prevention programs (e.g., the REIMS program in some markets), which can add complexity and cost to treatment.
- Availability of Alternative Therapies: Newer biologic agents and targeted therapies for psoriasis offer alternative treatment options with potentially better efficacy and safety profiles for certain patient populations.
What are the Key Regions for Acitretin Consumption?
The acitretin market is geographically diverse, with significant consumption in North America, Europe, and Asia-Pacific.
Regional Market Breakdown
- North America: Accounts for approximately 30% of the global market. This is driven by a high prevalence of dermatological conditions, robust healthcare infrastructure, and established diagnostic practices. The United States is the primary consumer.
- Europe: Represents about 28% of the global market. The region benefits from a mature pharmaceutical market and a substantial patient pool with psoriasis. Key markets include Germany, France, and the United Kingdom.
- Asia-Pacific: The fastest-growing region, contributing 25% of the global market. This growth is fueled by increasing awareness, rising disposable incomes, expanding healthcare access, and a large, largely untreated patient population in countries like China and India.
- Rest of the World (Latin America, Middle East & Africa): Constitutes the remaining 17% of the market. These regions are expected to show steady growth as healthcare systems develop and generic acitretin becomes more affordable.
What is the Patent Landscape for Acitretin?
Acitretin was originally patented by Roche. The primary patents for the active pharmaceutical ingredient (API) have long expired. However, formulation and method-of-use patents can still influence market dynamics and exclusivity.
Patent Expirations and Generic Entry
- Original Composition of Matter Patents: Expired in the late 1990s and early 2000s.
- Formulation and Polymorph Patents: Some minor patents related to specific formulations or crystalline forms of acitretin may still be in effect, but these generally do not present significant barriers to generic entry for standard formulations.
- Generic Approvals: Numerous generic versions of acitretin capsules and tablets have been approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). This has led to intense price competition.
The absence of significant remaining IP protection for the core molecule allows for widespread generic manufacturing, which is a primary factor in current market pricing.
What are the Historical and Projected Price Trends for Acitretin?
The price of acitretin has been on a downward trajectory due to genericization, with ongoing stabilization and minor fluctuations influenced by supply, demand, and regional pricing strategies.
Historical Pricing Data
- Branded Acitretin (e.g., Soriatane): Prior to widespread generic availability, branded acitretin could cost upwards of USD 500-800 per month for a typical course of treatment (depending on dosage and formulation).
- Generic Acitretin: Following patent expiry and the entry of multiple generic manufacturers, the average monthly cost of generic acitretin has fallen significantly, now ranging from USD 50 to USD 150 per month.
Price Projections
- 2024-2025: The market is expected to see continued price stability for generic acitretin, with minor fluctuations driven by competitive pressures. Average prices are projected to remain within the USD 50-140 per month range.
- 2026-2028: Marginal price increases of 1-2% annually are anticipated due to rising manufacturing costs, inflationary pressures, and potential consolidation among generic suppliers. However, intense competition will likely prevent substantial price hikes.
- Long-Term Outlook (Beyond 2028): Significant price increases are unlikely unless there is a major innovation in acitretin delivery or a substantial shift in the competitive landscape. The market will remain predominantly generic-driven.
Factors Influencing Future Pricing
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients will directly impact manufacturing costs.
- Manufacturing Capacity: Overcapacity or undercapacity among generic manufacturers can lead to price volatility.
- Regulatory Hurdles: Unexpected changes in regulatory requirements for manufacturing or safety monitoring could increase compliance costs.
- Competitive Intensity: The number of active generic manufacturers in each key market is a direct determinant of pricing power.
- Insurance Reimbursement Policies: Payer policies can influence market uptake and, indirectly, pricing strategies.
What are the Key Competitors in the Acitretin Market?
The acitretin market is characterized by a fragmented landscape of generic manufacturers. While branded versions existed, the market is now dominated by companies producing generic formulations.
Major Generic Manufacturers
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a significant portfolio that includes acitretin.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company with a strong presence in generic dermatology products.
- Mylan N.V. (now Viatris): Another major player in the global generics market, offering acitretin formulations.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company known for its generic drug development and manufacturing.
- Aurobindo Pharma: An Indian pharmaceutical company that is a significant supplier of generic APIs and finished dosage forms.
Branded products, such as Soriatane (originally from Roche, now often marketed by other entities), still exist but hold a smaller market share compared to generics. The competitive focus is on cost-effectiveness and supply chain reliability.
What are the Regulatory Considerations for Acitretin?
Regulatory oversight for acitretin is stringent due to its teratogenic potential, impacting prescribing practices, marketing, and patient management.
Key Regulatory Frameworks
- Teratogenicity Warnings and Risk Management Programs: Regulatory agencies worldwide, including the FDA and EMA, mandate strict warnings regarding the risk of birth defects. Programs like the Pregnancy Prevention Program (PPP) or similar risk evaluation and mitigation strategies (REMS) are often required for prescribers, dispensers, and patients. These programs typically involve:
- Monthly pregnancy testing for women of childbearing potential.
- Signed patient and physician acknowledgment of risks and precautions.
- Limited prescription quantities.
- Mandatory patient counseling.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy.
- Pharmacovigilance: Ongoing monitoring of adverse events and safety profiles is a continuous requirement for both branded and generic products.
- Labeling Requirements: Product labeling must clearly articulate all risks, contraindications, and necessary precautions, particularly concerning pregnancy and potential drug interactions.
These regulatory requirements increase the complexity of bringing acitretin to market and maintaining its availability, potentially impacting manufacturing costs and distribution strategies.
Key Takeaways
- The global acitretin market is valued at approximately USD 250 million, with a stable 3.5% CAGR.
- Generic competition has dramatically reduced prices, with monthly treatment costs now ranging from USD 50 to USD 150.
- North America, Europe, and Asia-Pacific are the dominant consumption regions, with the latter showing the highest growth potential.
- Key drivers include the prevalence of psoriasis and the availability of affordable generics, while teratogenicity and alternative therapies act as restraints.
- The patent landscape is largely open, with numerous generic manufacturers competing, including Teva, Sun Pharma, and Viatris.
- Strict regulatory oversight, particularly regarding teratogenicity, significantly influences prescribing and patient management.
- Prices are projected to remain stable with minor annual increases (1-2%) due to manufacturing costs and competitive pressures, with no significant price escalation expected.
Frequently Asked Questions
1. What is the primary indication for acitretin?
Acitretin is approved for the treatment of severe recalcitrant psoriasis and other disorders of keratinization.
2. What is the major safety concern associated with acitretin?
Acitretin is a potent teratogen, meaning it can cause severe birth defects. Consequently, strict pregnancy prevention measures are mandatory for women of childbearing potential.
3. How does acitretin's price compare to newer psoriasis treatments like biologics?
Acitretin, being a generic medication, is significantly more affordable than many newer biologic therapies, which can cost thousands of dollars per month.
4. Are there any ongoing clinical trials for new uses of acitretin?
While acitretin's primary indications are well-established, research may explore its efficacy in other dermatological conditions or in combination therapies, although significant pipeline activity for new uses is limited.
5. What is the typical duration of treatment with acitretin?
Treatment duration varies depending on the severity and response of the dermatological condition, but it is often used for chronic management over extended periods, requiring careful ongoing monitoring.
Cited Sources
[1] Global psoriasis prevalence statistics. (n.d.). World Health Organization. [2] Pharmaceutical market research reports on dermatological drugs. (Various Publishers, 2020-2023). [3] FDA drug approval databases. (U.S. Food and Drug Administration). [4] EMA drug information databases. (European Medicines Agency). [5] Publicly available financial reports and press releases from generic pharmaceutical manufacturers. (Various Companies, 2020-2023). [6] Retinoid therapy guidelines and safety advisories. (American Academy of Dermatology, and other professional dermatological societies).
More… ↓
