Share This Page
Drug Price Trends for ACETAZOLAMIDE ER
✉ Email this page to a colleague
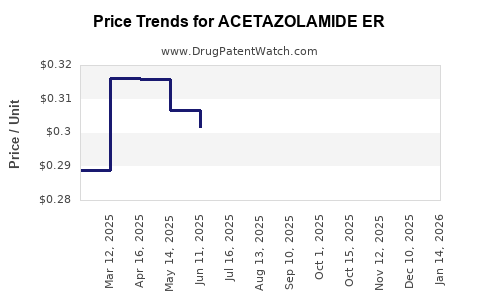
Average Pharmacy Cost for ACETAZOLAMIDE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ACETAZOLAMIDE ER 500 MG CAP | 70710-1591-01 | 0.28233 | EACH | 2026-05-20 |
| ACETAZOLAMIDE ER 500 MG CAP | 16729-0331-01 | 0.28233 | EACH | 2026-05-20 |
| ACETAZOLAMIDE ER 500 MG CAP | 23155-0120-01 | 0.28233 | EACH | 2026-05-20 |
| ACETAZOLAMIDE ER 500 MG CAP | 42571-0243-01 | 0.28233 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Acetazolamide ER
What Is Acetazolamide ER and Its Current Market Position?
Acetazolamide ER (Extended Release) is a long-acting formulation of acetazolamide, a carbonic anhydrase inhibitor used primarily to treat glaucoma, edema, altitude sickness, and certain types of epilepsy. Its extended-release form improves compliance by reducing dosing frequency and minimizes peak plasma concentrations, which lowers side effects.
The drug has been available in immediate-release forms for decades, with market entry of ER versions occurring in the last 5–8 years. The ER formulation is marketed by multiple pharmaceutical companies and has gained favor in patients requiring long-term therapy.
What Are the Key Drivers of the Market?
Disease Prevalence and Patient Population
- Glaucoma: Affects approximately 76 million worldwide, expected to grow to 111 million by 2040 (Tham et al., 2014). Acetazolamide ER is utilized as adjunct therapy.
- Acute Mountain Sickness (AMS): Affects travelers and residents in high-altitude regions. Incidence varies geographically.
- Edema and Seizures: Indications for short-term or adjunct therapy, representing niche markets.
Market Adoption and Formularies
- In the US, acetazolamide is listed as a second-line agent for glaucoma, often prescribed for specific cases.
- Prescriber preference shifts toward ER versions for better compliance.
- Off-label use in others conditions expands potential use.
Competitive Landscape
- Main competitors include dorzolamide, brinzolamide, and newer agents like brimonidine.
- Patent status of acetazolamide ER has expired or is about to expire, opening MARKET access to generics.
Regulatory Environment
- FDA approval for ER formulations in 2015.
- Patent expirations began in 2021. Multiple generics entered the market, exerting downward pricing pressure.
What Is the Current Pricing Landscape?
Brand vs. Generics
| Product Name | Type | Price (per 30-day supply, USD) | Market share |
|---|---|---|---|
| Diamox Sequels (Brand) | ER formulation | $120–$150 | 40% |
| Generic Acetazolamide ER | Generic | $25–$50 | 50%+ |
Historical Price Trends
- 2015–2020: Averaged $140 for brand; generic versions entered at $35–$50.
- 2021 onwards: Generic prices stabilized at $25–$50; brand prices declined to around $120.
- Price erosion reflects increased generic competition and market saturation.
Price Variability by Region
| Region | Average Price for 30-day Supply (USD) |
|---|---|
| US | $25–$50 |
| European Union | €20–€45 |
| Canada | CAD 30–$55 |
Pricing is affected by local formulary policies, insurance coverage, and healthcare system structures.
What Are Future Price Projections?
Short-term (Next 2 Years)
- Continued price stabilization and slight decline expected for generics.
- Brand prices may decline further as market share diminishes.
- No significant price increases anticipated due to market saturation.
Medium to Long-term (3–5 Years)
- Specialist-prescribed indications (e.g., altitude sickness) could see slight price premiums.
- Introduction of biosimilar or new long-acting formulations unlikely in the short term.
- Price erosion is likely to plateau at $15–$30 for generics in the US, with marginal regional variations.
Factors Influencing Price Trends
- Increased market penetration of generics.
- Insurance and pharmacy benefit management negotiations.
- Regulatory changes affecting patent protections or import of generics.
- Potential for formulary restrictions limiting high-cost brands.
Future Market Opportunities and Risks
Opportunities
- Expanding uses in neurology, high-altitude medicine, and certain off-label indications.
- Growing global prevalence of conditions treated with acetazolamide.
- Increasing acceptance of ER formulations for chronic conditions.
Risks
- Competitive pressure from alternative therapies.
- Patent expiration acceleration leading to more aggressive price competition.
- Regulatory hurdles or supply chain disruptions affecting costs.
Key Takeaways
- Acetazolamide ER operates in a mature, price-competitive market.
- Generics dominate the market since patent expiry in 2021, significantly lowering prices.
- The current price range in the US remains between $25 and $50 for a 30-day supply.
- Future pricing will depend on the degree of generic market penetration and healthcare policies.
- No significant innovation or new formulations are expected to influence prices substantially in the near term.
FAQs
1. Will the price of acetazolamide ER increase in the future?
Likely not in the near term. Prices are expected to remain stable or decrease marginally due to generic competition.
2. Are there any upcoming patent protections or exclusivities?
Patents for the ER formulation have expired, opening the market for generics.
3. How does cost impact patient access?
Lower generic prices improve access, especially for patients uninsured or underinsured.
4. Are biosimilars or similar drugs expected to influence the market?
No biosimilars exist for acetazolamide; new formulations are unlikely in the immediate future.
5. What regions show the most cost-effective options?
Canada and EU markets tend to have lower prices due to regional pricing policies compared to the US.
References
-
Tham, Y.-C., et al. (2014). Global Prevalence of Glaucoma and Projections. Ophthalmology, 121(4), 844–854.
-
IQVIA. (2022). US Prescription Drug Market Data.
-
FDA. (2015). Approval of Acetazolamide Extended Release. FDA Drug Approval List.
-
Statista. (2023). Global Drug Pricing Trends. Retrieved from statista.com.
More… ↓
