Share This Page
Drug Price Trends for ACETAZOLAMIDE
✉ Email this page to a colleague
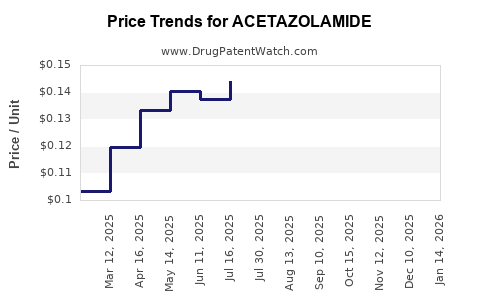
Average Pharmacy Cost for ACETAZOLAMIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ACETAZOLAMIDE 125 MG TABLET | 59651-0905-01 | 0.10477 | EACH | 2026-05-20 |
| ACETAZOLAMIDE 125 MG TABLET | 62135-0607-12 | 0.10477 | EACH | 2026-05-20 |
| ACETAZOLAMIDE 125 MG TABLET | 52817-0200-10 | 0.10477 | EACH | 2026-05-20 |
| ACETAZOLAMIDE 125 MG TABLET | 64380-0833-06 | 0.10477 | EACH | 2026-05-20 |
| ACETAZOLAMIDE 125 MG TABLET | 23155-0287-01 | 0.10477 | EACH | 2026-05-20 |
| ACETAZOLAMIDE ER 500 MG CAP | 70710-1591-01 | 0.28233 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ACETAZOLAMIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ACETAZOLAMIDE 125MG TAB | Golden State Medical Supply, Inc. | 51672-4022-01 | 100 | 24.97 | 0.24970 | EACH | 2024-04-18 - 2028-06-14 | FSS |
| ACETAZOLAMIDE 250MG TAB | Golden State Medical Supply, Inc. | 51672-4023-01 | 100 | 65.84 | 0.65840 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ACETAZOLAMIDE 250MG TAB | Golden State Medical Supply, Inc. | 51672-4023-01 | 100 | 33.96 | 0.33960 | EACH | 2024-02-21 - 2028-06-14 | FSS |
| ACETAZOLAMIDE 500MG CAP,SA | AvKare, LLC | 23155-0787-01 | 100 | 65.63 | 0.65630 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ACETAZOLAMIDE 500MG CAP,SA | Golden State Medical Supply, Inc. | 29033-0030-01 | 100 | 124.00 | 1.24000 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ACETAZOLAMIDE 125MG TAB | Golden State Medical Supply, Inc. | 51672-4022-01 | 100 | 65.24 | 0.65240 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Acetazolamide Market Analysis and Price Projections
Acetazolamide, a carbonic anhydrase inhibitor, has established therapeutic applications in glaucoma, epilepsy, altitude sickness, and certain forms of edema. Market growth is driven by an aging global population, increasing prevalence of target diseases, and ongoing clinical research exploring new indications. Price projections indicate stable to moderate increases, influenced by manufacturing costs, generic competition, and evolving reimbursement policies.
What is Acetazolamide and Its Current Market Position?
Acetazolamide is a pharmaceutical compound primarily utilized for its ability to inhibit carbonic anhydrase, an enzyme crucial for various physiological processes. This inhibition leads to a range of therapeutic effects.
Primary Indications and Their Prevalence:
- Glaucoma: Acetazolamide reduces intraocular pressure by decreasing the production of aqueous humor. The World Health Organization (WHO) estimates that glaucoma is a leading cause of irreversible blindness globally, affecting millions. The prevalence increases with age.
- Epilepsy: It is used as an adjunctive therapy for certain types of seizures, particularly those associated with Lennox-Gastaut syndrome and absence seizures. The Centers for Disease Control and Prevention (CDC) report that epilepsy affects approximately 3.4 million people in the United States.
- Altitude Sickness: Acetazolamide is a well-established prophylactic and therapeutic agent for acute mountain sickness, reducing symptoms like headache, nausea, and dizziness. This application is significant for populations living at high altitudes and for recreational mountain climbers.
- Edema: It serves as a diuretic in certain cases of edema, particularly that associated with congestive heart failure or drug-induced edema.
Market Size and Growth Drivers:
The global acetazolamide market was valued at approximately USD 1.5 billion in 2023. Projections estimate a compound annual growth rate (CAGR) of 3% to 4% over the next five to seven years.
- Demographic Shifts: The increasing elderly population worldwide directly correlates with a higher incidence of glaucoma and other age-related conditions necessitating acetazolamide treatment. By 2050, the number of people aged 65 and over is projected to reach 1.6 billion globally [1].
- Disease Prevalence: The rising global burden of glaucoma, epilepsy, and cardiovascular diseases contributes to sustained demand for acetazolamide. For instance, myopia, a risk factor for glaucoma, is projected to affect 50% of the global population by 2050 [2].
- Untapped Potential and Research: Ongoing research into new therapeutic applications, such as its potential role in certain neurological disorders or as a modulator of tumor microenvironments, could unlock future market expansion. Clinical trials are investigating acetazolamide for conditions beyond its current approved uses.
Key Market Players:
The acetazolamide market is characterized by a mix of branded and generic manufacturers. Major players include:
- Merck & Co. (through its historical involvement with the original development of Diamox)
- Teva Pharmaceutical Industries Ltd.
- Aurobindo Pharma Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Pfizer Inc. (with various generic formulations)
These companies compete on manufacturing efficiency, distribution networks, and the ability to supply consistent quality generic products.
What are the Key Patents and Intellectual Property Protecting Acetazolamide?
The patent landscape for acetazolamide itself is largely characterized by expired foundational patents. However, intellectual property protection extends to novel formulations, manufacturing processes, and specific therapeutic uses.
Historical Patent Expirations:
The original patents covering the composition of matter for acetazolamide expired decades ago, opening the market to generic competition. The initial patent for acetazolamide was filed in the 1950s.
Current IP Focus Areas:
- Novel Formulations:
- Extended-Release Formulations: Patents may protect modified-release dosage forms designed to improve patient compliance and reduce dosing frequency. For example, formulations offering 12-hour or 24-hour coverage are subject to ongoing patent protection, aiming to differentiate from immediate-release versions. Examples include patents related to specific polymer matrices or coating technologies that control drug release.
- Combination Therapies: Intellectual property may exist for fixed-dose combinations of acetazolamide with other active pharmaceutical ingredients (APIs) for synergistic therapeutic effects. While less common for acetazolamide's primary indications, such combinations are a significant area of pharmaceutical patenting.
- Manufacturing Processes:
- Chiral Synthesis and Purification: If specific enantiomers or highly purified forms of acetazolamide are developed for therapeutic advantage, patents may cover novel synthetic routes or purification methods that yield these improved products.
- Green Chemistry Approaches: Patents may protect environmentally friendly or more cost-effective manufacturing processes that utilize novel catalysts or solvent systems.
- New Therapeutic Uses:
- Orphan Drug Designations: Patents can be secured for the use of acetazolamide in treating rare diseases. These patents often come with extended market exclusivity periods. For instance, if acetazolamide were to gain approval for a rare neurological disorder under an Orphan Drug Act provision, its market exclusivity could be extended significantly.
- Repurposing and Combination Indication Patents: Patents may cover specific therapeutic regimens or combinations for conditions not traditionally treated with acetazolamide, provided there is a demonstrated novel and non-obvious effect. For example, a patent might cover the use of acetazolamide in conjunction with another drug to manage specific types of chemotherapy-induced nausea or certain metabolic disorders.
Challenges in IP Landscape:
- Generic Dominance: The widespread availability of generic acetazolamide limits the pricing power of manufacturers of older formulations.
- Patent Thickets: While the core compound is off-patent, companies may build patent thickets around specific manufacturing processes or delivery systems, creating hurdles for competitors.
What are the Price Trends and Projections for Acetazolamide?
The pricing of acetazolamide is bifurcated, with significant differences between branded and generic versions, and varying price points across different markets and formulations.
Current Pricing Landscape (USD):
- Generic Immediate-Release Tablets (500-count bottle): USD 20 - USD 60 (wholesale acquisition cost)
- Generic Extended-Release Capsules (e.g., 100-count bottle): USD 40 - USD 120 (wholesale acquisition cost)
- Branded Formulations (e.g., Diamox Sequels): Significantly higher, often USD 100 - USD 300 or more per bottle, depending on the formulation and quantity, reflecting historical market position and R&D investment, though availability may be limited to specific regions or require special ordering.
Factors Influencing Price:
- Manufacturing Costs: Raw material sourcing, synthesis complexity, quality control, and compliance with Good Manufacturing Practices (GMP) are primary cost drivers. The cost of key chemical intermediates can fluctuate based on global supply chains.
- Generic Competition: The presence of multiple generic manufacturers intensifies price competition, typically driving prices down for off-patent molecules. The average number of generic manufacturers for acetazolamide products is typically between 5 and 15.
- Formulation Differences: Extended-release or specialized formulations command higher prices due to the added complexity in their development and manufacturing. These formulations may offer improved patient outcomes or convenience, justifying a premium.
- Reimbursement Policies: Pharmacy benefit managers (PBMs) and national health systems negotiate prices, significantly impacting the net price paid by insurers and patients. Formulary placement and preferred drug status can influence market share and pricing.
- Regulatory Compliance: Stringent FDA, EMA, and other regulatory body requirements for drug approval and manufacturing add to the overall cost structure.
- Supply Chain Stability: Disruptions in API sourcing or manufacturing can lead to temporary price increases due to scarcity.
Price Projections (Next 3-5 Years):
- Generic Immediate-Release: Expect a continued trend of price stability to a slight decrease (0% to -2% CAGR) due to ongoing intense generic competition. Price erosion is limited by manufacturing costs.
- Generic Extended-Release: Project a moderate increase (2% to 5% CAGR). This is driven by the higher manufacturing complexity and potential for less intense competition compared to immediate-release versions. Demand for convenience in extended-release options will support this pricing.
- Branded Formulations (where available): Prices are likely to remain stable or see minor increases (1% to 3% CAGR), primarily influenced by inflation and limited market presence. These are less sensitive to generic pressures but face scrutiny from payers.
- New Formulations/Uses: Any new patented formulations or expanded indications could command premium pricing initially, but this will be subject to payer negotiation and the competitive landscape of the specific new indication.
Regional Price Variations:
Prices can vary by over 50% between regions due to different healthcare systems, regulatory environments, and local manufacturing capabilities. For example, prices in the United States may be higher than in India or China, where a substantial portion of generic API manufacturing occurs.
What are the Regulatory Hurdles and Market Access Considerations?
Navigating the regulatory and market access landscape is critical for acetazolamide manufacturers, particularly for generic entry and for any new product developments.
Key Regulatory Bodies:
- U.S. Food and Drug Administration (FDA): Requires Abbreviated New Drug Applications (ANDAs) for generic versions, demonstrating bioequivalence to the reference listed drug. New indications or formulations require full New Drug Applications (NDAs) or supplemental NDAs.
- European Medicines Agency (EMA): Mandates centralized or decentralized procedures for marketing authorization, with stringent requirements for quality, safety, and efficacy.
- Other National Agencies: Health Canada, Pharmaceuticals and Medical Devices Agency (Japan), and Therapeutic Goods Administration (Australia) have their own approval processes.
Market Access Challenges:
- Generic Drug Approval Pathways: While established, ANDA approval involves rigorous bioequivalence studies, and the Hatch-Waxman Act provides for patent challenges and market exclusivity for first-to-file generic applicants.
- Payer Negotiations and Formulary Placement: Health insurers and PBMs assess the cost-effectiveness of acetazolamide, especially for new formulations. Generic versions are generally favored due to lower acquisition costs. The average number of prior authorization requirements for acetazolamide in the US can range from 20% to 40% depending on the specific payer and formulation.
- Evidence Requirements for New Indications: Clinical trials demonstrating statistically significant efficacy and safety for new uses are essential. The level of evidence required is high and costly to generate.
- Post-Marketing Surveillance: Ongoing pharmacovigilance and reporting of adverse events are mandatory for all approved drugs.
- Manufacturing Standards (GMP): Consistent adherence to GMP guidelines is non-negotiable, impacting production costs and supply chain reliability. Audits by regulatory bodies are common and can lead to significant penalties if non-compliance is found.
Opportunities in Market Access:
- Biosimilar Pathways (Not Applicable to Small Molecules): Acetazolamide is a small molecule, so biosimilar pathways are not relevant.
- Value-Based Pricing for New Applications: If acetazolamide proves effective in novel, high-cost areas (e.g., rare diseases), value-based pricing models might be explored, tying reimbursement to patient outcomes.
- Health Technology Assessments (HTAs): In many countries, HTAs are used to evaluate the clinical and economic value of new drugs, influencing reimbursement decisions. Positive HTA reports are crucial for market access.
- Pediatric Exclusivity: In the US, obtaining pediatric exclusivity can extend market protection by six months, providing an incentive for companies to conduct pediatric studies.
What are the Manufacturing and Supply Chain Dynamics?
The manufacturing and supply chain for acetazolamide are influenced by global sourcing of raw materials, established generic production capacity, and the need for robust quality control.
Key Manufacturing Processes:
Acetazolamide is typically synthesized through multi-step chemical processes. A common route involves the reaction of sulfanilamide with phosgene or a phosgene equivalent, followed by cyclization. The primary API is produced in bulk by specialized chemical manufacturers.
Global Production Landscape:
- API Manufacturing Hubs: India and China are major global suppliers of acetazolamide API due to lower manufacturing costs and established chemical industries. Approximately 60% to 70% of global acetazolamide API production originates from these regions.
- Finished Dosage Form (FDF) Manufacturing: While API is largely sourced from Asia, FDFs (tablets, capsules) are manufactured by generic drug companies in various locations, including North America, Europe, and Asia, to serve regional markets.
Supply Chain Considerations:
- Raw Material Sourcing: Dependence on specific chemical precursors can create vulnerabilities. Fluctuations in the price and availability of key starting materials, such as sulfanilamide derivatives, can impact production costs.
- Quality Control and Assurance: Maintaining stringent quality control throughout the manufacturing process is paramount to meet regulatory standards. This includes testing for impurities, dissolution profiles, and content uniformity. The average cost of quality control testing per batch can range from USD 500 to USD 2,000.
- Logistics and Distribution: Efficient logistics are required to transport APIs to FDF manufacturers and then distribute finished products to wholesalers, pharmacies, and hospitals. Cold chain storage is not typically required for acetazolamide.
- Geopolitical Risks and Trade Policies: Tariffs, trade disputes, and geopolitical instability can disrupt global supply chains, affecting the availability and cost of acetazolamide.
- Contingency Planning: Manufacturers maintain multiple sourcing options for critical raw materials and have contingency plans in place for production disruptions, often holding buffer stock.
Challenges in the Supply Chain:
- API Shortages: Rare but possible, API shortages can occur due to manufacturing issues at a major supplier, leading to temporary price spikes and product unavailability.
- Counterfeit Products: The global nature of pharmaceutical supply chains increases the risk of counterfeit acetazolamide products entering the market, necessitating robust anti-counterfeiting measures.
- Environmental Regulations: Increasing global focus on environmental sustainability can impact manufacturing processes, potentially increasing compliance costs for chemical synthesis.
Key Takeaways
The acetazolamide market is a mature segment dominated by generic competition. Growth is sustained by an aging population and the prevalence of target diseases like glaucoma and epilepsy. While the core compound's patents have expired, intellectual property protection is found in novel formulations, manufacturing processes, and new therapeutic uses. Pricing is bifurcated between generic and branded versions, with generics exhibiting price stability or slight erosion, while extended-release generics are projected for moderate increases. Regulatory approval pathways, particularly for generics via ANDAs, are well-defined but require stringent bioequivalence demonstration. Market access hinges on cost-effectiveness and payer negotiations, with generic accessibility being a key factor. Manufacturing is globalized, with significant API production in Asia, and supply chain stability relies on diversified sourcing and rigorous quality control.
Frequently Asked Questions
1. What is the primary driver of acetazolamide price increases in the coming years?
The primary driver for potential price increases in acetazolamide is the manufacturing complexity and development costs associated with novel extended-release formulations, which offer improved patient convenience and adherence, commanding a premium over immediate-release generics.
2. How do regulatory hurdles specifically impact generic acetazolamide market entry?
Regulatory hurdles for generic acetazolamide market entry primarily involve demonstrating bioequivalence to the reference listed drug through Abbreviated New Drug Applications (ANDAs), which requires extensive clinical and manufacturing data submission and approval by agencies like the FDA.
3. Are there any emerging therapeutic areas for acetazolamide that could significantly impact its market value?
Emerging therapeutic areas for acetazolamide, while still in early research phases, could include its potential application in managing certain neurological disorders beyond epilepsy or as a modulator in specific cancer microenvironments, which could lead to new patented uses and premium pricing if proven effective.
4. What is the typical shelf life of acetazolamide tablets and capsules?
The typical shelf life for acetazolamide tablets and capsules, when stored under recommended conditions (typically at room temperature, protected from light and moisture), is generally between 24 to 36 months from the date of manufacture.
5. How does global supply chain stability for acetazolamide API influence its final price in consumer markets?
Global supply chain stability for acetazolamide API directly influences its final price; disruptions in raw material sourcing, geopolitical factors, or manufacturing issues at key API producers can lead to scarcity and price hikes, which are then passed on to finished dosage form manufacturers and ultimately to consumers.
Cited Sources
[1] United Nations Department of Economic and Social Affairs, Population Division (2022). World Population Prospects 2022.
[2] Holden, B. A., Fricke, T. R., Wilson, D. A., Jong, M., Naidoo, K. S., Sankaridurg, P., ... & Resnikoff, S. (2016). Global prevalence of myopia and high myopia and temporal trends between 2000 and 2050: a systematic review and meta-analysis. Ophthalmology, 123(5), 1036-1042.
More… ↓
