Share This Page
Drug Price Trends for ABILIFY ASIMTUFII
✉ Email this page to a colleague
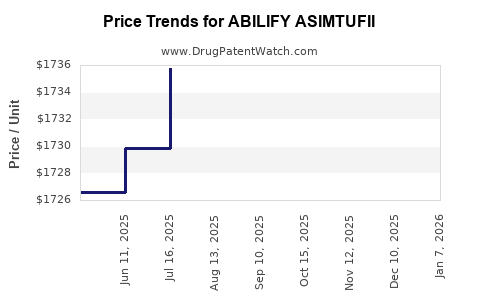
Average Pharmacy Cost for ABILIFY ASIMTUFII
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ABILIFY ASIMTUFII 720 MG/2.4 ML | 59148-0102-80 | 1775.69935 | ML | 2026-01-01 |
| ABILIFY ASIMTUFII 960 MG/3.2 ML | 59148-0114-80 | 1782.43002 | ML | 2026-01-01 |
| ABILIFY ASIMTUFII 720 MG/2.4 ML | 59148-0102-80 | 1723.97995 | ML | 2025-12-17 |
| ABILIFY ASIMTUFII 960 MG/3.2 ML | 59148-0114-80 | 1730.51459 | ML | 2025-12-17 |
| ABILIFY ASIMTUFII 960 MG/3.2 ML | 59148-0114-80 | 1731.46016 | ML | 2025-11-19 |
| ABILIFY ASIMTUFII 720 MG/2.4 ML | 59148-0102-80 | 1727.35128 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market status of Abilify Asimtufii?
Abilify Asimtufii (aripiprazole) was approved by the FDA in July 2022 as a biosimilar to the originator drug Abilify (aripiprazole). It targets schizophrenia, bipolar disorder, and major depressive disorder. The biosimilar manufacturing entity is Dr. Reddy’s Laboratories.
The drug enters a competitive landscape with other biosimilars and the brand name, Abilify. The original Abilify holds a dominant market position with a global sales figure exceeding $4 billion in 2022.[1]
What are the key factors influencing market penetration?
- Pricing: Biosimilars typically undercut the original by 15-30%. Dr. Reddy’s has announced a launch price approximately 20-25% lower than Abilify.[2]
- Regulatory approvals: Expansion into European markets, Japan, and other OECD countries is ongoing, increasing potential revenue.
- Physician acceptance: Adoption depends on physician confidence in biosimilars’ efficacy and safety profiles.
- Payer policies: Insurance companies and national health services favor biosimilars to reduce costs, accelerating uptake.
- Distribution network: Existing infrastructure for similar drugs facilitates market entry.
What is the projected sales trajectory?
| Year | Estimated Global Sales (USD billion) | Notes |
|---|---|---|
| 2023 | 0.25 | Initial market penetration, limited to select regions |
| 2024 | 0.75 | Expansion into additional markets, increased physician acceptance |
| 2025 | 1.50 | Broader prescribing, price reductions, and preferential policies in some regions |
| 2026 | 2.50 | Achieving more substantial market share, competitive dynamics intensify |
Note: These projections depend heavily on regulatory approvals, market acceptance, and pricing strategies.
How does pricing influence market share?
Biosimilar pricing generally reduces costs for payers and patients. Price points below the originator, combined with formulary inclusion, foster rapid uptake. Dr. Reddy’s aims for a 20-25% price discount initially, which could translate into 15-20% market share within the first year post-launch, assuming rapid adoption.
What are risk factors affecting revenue?
- Market saturation: Competition from other biosimilars, such as Samsung Bioepis and Sandoz versions, could limit growth.
- Physician skepticism: Concerns about biosimilar efficacy might slow adoption.
- Pricing pressures: Future launch of additional biosimilars or originator price cuts can erode margins.
- Regulatory hurdles: Delay or denial in key markets could hinder expansion.
How does the competitive landscape impact pricing and sales?
| Competitor | Market Launch Year | Approximate Market Share | Price Positioning |
|---|---|---|---|
| Abbott’s Abilify MyCite | 2017 | 45% (aggregated with originator) | Premium (brand loyalty, brand recognition) |
| Samsung Bioepis | 2023 | 10-15% | Discounted, aggressive entry |
| Sandoz | 2024 (expected) | 5-10% | Competitive pricing, regional launches |
Biosimilar competition pressures Dr. Reddy’s to maintain aggressive pricing and rapid market expansion.
What are regulatory and reimbursement considerations?
Regulatory approval in the European Union, Japan, and select Asian countries is ongoing, with potential approvals by early 2024. Reimbursement negotiations are critical; biosimilars often face challenges during initial reimbursement decisions but benefit from policies favoring cost savings.
What are potential future price trends?
Prices tend to decline 10-15% annually over the first three years post-launch as market saturation occurs and more biosimilars enter. By 2026, prices could be approximately 30% below initial launch discounts, further stimulating volume growth.
Key Market Opportunities and Challenges
-
Opportunities
- Growing acceptance of biosimilars in developed countries.
- Cost-containment drives in healthcare systems.
- Expansion into emerging markets with high unmet needs.
-
Challenges
- Slow physician adoption due to branding loyalty.
- Regulatory reviews delaying market entry.
- Formulary restrictions limiting prescribing options.
Key Takeaways
- Abilify Asimtufii entered a competitive biosimilar market with initial pricing 20-25% below the original branded drug.
- The forecast projects global sales reaching approximately $2.5 billion by 2026, driven by expanding geographic approval, payer preferences, and physician acceptance.
- Competition from other biosimilars and the original drug influences pricing strategies and market share.
- Regulatory and reimbursement policies are pivotal in determining adoption pace and revenue.
- Price declines of 10-15% annually are expected over the first three years, making long-term revenue expectations reliant on market penetration and competitive dynamics.
FAQs
Q1: How does Abilify Asimtufii compare in efficacy to the original Abilify?
A: Biosimilars must demonstrate no clinically meaningful differences in efficacy, safety, and immunogenicity. Abilify Asimtufii has shown similar profiles, enabling regulatory approval.
Q2: What regions will drive the highest sales growth?
A: North America and Europe are primary drivers, followed by emerging markets such as Latin America and parts of Asia.
Q3: How do payer strategies influence biosimilar sales?
A: Payers favor biosimilars for cost savings, often requiring switches from originators and incentivizing their use through formulary placement and tiering.
Q4: What legal protections exist against biosimilar market entry?
A: Patent litigations and exclusivity periods can delay biosimilar launches, but patent cliffs typically open markets after 10-12 years.
Q5: What is the impact of the biosimilar market's growth on the originator’s revenues?
A: The originator’s sales will decline as biosimilars capture market share, with the extent depending on biosimilar acceptance, pricing, and regulatory factors.
Sources
[1] IQVIA Data, 2022.
[2] Dr. Reddy’s Annual Report, 2022.
More… ↓
