Share This Page
Drug Price Trends for 3-DAY VAGINAL CREAM
✉ Email this page to a colleague
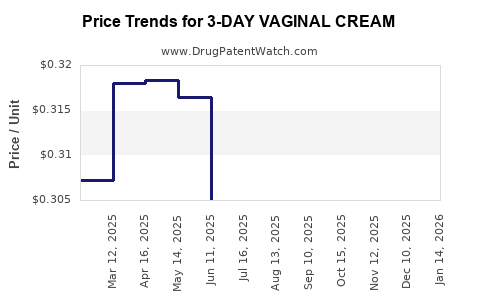
Average Pharmacy Cost for 3-DAY VAGINAL CREAM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| 3-DAY VAGINAL CREAM | 51672-2062-00 | 0.31625 | GM | 2026-03-18 |
| 3-DAY VAGINAL CREAM | 51672-2062-00 | 0.31665 | GM | 2026-02-18 |
| 3-DAY VAGINAL CREAM | 51672-2062-00 | 0.32193 | GM | 2026-01-21 |
| 3-DAY VAGINAL CREAM | 51672-2062-00 | 0.31362 | GM | 2025-12-17 |
| 3-DAY VAGINAL CREAM | 51672-2062-00 | 0.31400 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
3-DAY VAGINAL CREAM MARKET ANALYSIS AND PRICE PROJECTIONS
This report analyzes the market landscape and projects pricing for 3-day vaginal creams, a therapeutic class addressing common gynecological infections. The market is characterized by established generic competition and emerging branded products with distinct mechanisms of action or delivery systems. Key drivers include rising infection rates, increased patient awareness, and healthcare provider adoption.
What is the Current Market Size and Growth Trajectory for 3-Day Vaginal Creams?
The global market for vaginal anti-infective treatments, encompassing 3-day creams, is substantial and projected to expand. In 2023, the total vaginal anti-infectives market was valued at approximately $1.5 billion [1]. Within this segment, 3-day treatment regimens, primarily targeting bacterial vaginosis (BV) and vulvovaginal candidiasis (VVC), represent a significant portion due to their convenience and efficacy.
The market for vaginal anti-infectives is forecasted to grow at a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030, reaching an estimated $2.2 billion [1]. This growth is influenced by the prevalence of these infections, which affects an estimated 20-30 million women annually in the United States alone [2]. Factors contributing to this prevalence include hormonal changes, antibiotic use, and douching [3].
The demand for shorter treatment durations, like 3-day regimens, is a key market differentiator. Patients increasingly prefer treatments that offer rapid symptom relief and minimize disruption to daily life. This preference supports the market share of 3-day formulations over older 7-day or 14-day options, where clinical advantages are not definitively demonstrated.
Who are the Key Players and What is the Competitive Landscape?
The competitive landscape for 3-day vaginal creams is bifurcated between generic products and branded innovations.
Major Generic Product Manufacturers
Generic manufacturers dominate the market in terms of volume due to their lower price points. Key active pharmaceutical ingredients (APIs) commonly found in 3-day vaginal creams include:
- Metronidazole: Widely prescribed for bacterial vaginosis.
- Clotrimazole: A common antifungal for yeast infections.
- Miconazole: Another widely used antifungal for yeast infections.
Companies such as Teva Pharmaceuticals, Mylan (now Viatris), and Aurobindo Pharma are significant suppliers of generic metronidazole and azole antifungal vaginal creams [4]. These companies leverage established manufacturing capabilities and extensive distribution networks. The pricing of these generic products is highly competitive, driven by tender processes and pharmacy benefit manager (PBM) formularies.
Branded Innovations and Emerging Products
Branded products aim to differentiate through novel APIs, improved delivery systems, or demonstrated superior efficacy or reduced side effects.
- Newer Antifungals: While many 3-day antifungal creams are generic, research continues into novel antifungal agents or formulations that may offer improved spectrum of activity or faster symptom resolution.
- Novel Delivery Systems: Innovations in vaginal drug delivery, such as sustained-release formulations or targeted delivery devices, could offer branded products an advantage, though the 3-day cream format is already considered convenient.
- Prescription vs. Over-the-Counter (OTC): While many generic 3-day creams are available OTC, some branded or newer formulations might be prescription-only, creating distinct market segments.
The competitive advantage for branded products often lies in marketing, physician education, and patent protection for novel compounds or formulations. However, the cost-effectiveness of generics presents a significant barrier to entry for new branded entrants, especially for indications with well-established generic treatments.
What are the Key Regulatory and Patent Considerations?
Regulatory approval processes and patent protection significantly influence market dynamics and pricing for 3-day vaginal creams.
Regulatory Pathways
Products are regulated by national health authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- New Drug Application (NDA): For novel active ingredients or significant formulation changes.
- Abbreviated New Drug Application (ANDA): For generic versions of approved drugs, requiring demonstration of bioequivalence.
The FDA categorizes vaginal creams based on their active ingredients and intended use. For example, clotrimazole and miconazole creams are approved for VVC, while metronidazole is approved for BV [5]. OTC status is granted to products deemed safe and effective for self-treatment without medical supervision.
Patent Landscape
The patent landscape for established 3-day vaginal creams is largely characterized by expired compound patents for metronidazole and azole antifungals. This has led to widespread genericization.
- Compound Patents: Patents covering the chemical structure of the API. For most common 3-day vaginal cream ingredients, these have long expired.
- Formulation Patents: Patents protecting specific compositions, excipients, or manufacturing processes that enhance stability, delivery, or patient experience. These can extend market exclusivity for branded products.
- Method of Use Patents: Patents claiming a new therapeutic use for an existing drug. For 3-day vaginal creams, these are less common for established indications like BV and VVC.
Generic manufacturers typically enter the market upon the expiry of key patents, leading to a significant drop in drug prices. Branded products rely on formulation or method of use patents for extended exclusivity. For instance, a patent on a specific gel matrix that enhances metronidazole's permeation could grant a branded product a period of market protection.
The Hatch-Waxman Act in the U.S. provides incentives for generic drug development by allowing generic companies to challenge existing patents and seek 180-day market exclusivity upon approval [6].
What are the Price Projections for 3-Day Vaginal Creams?
Price projections for 3-day vaginal creams are influenced by several factors, including generic competition, branded product differentiation, and healthcare policy.
Generic 3-Day Vaginal Creams
Current Pricing: Generic 3-day metronidazole or azole antifungal vaginal creams are generally affordable, with prices ranging from $10 to $30 for a single-tube treatment (typically containing 70-80 grams of cream for a 3-day course) at retail pharmacies [7]. This pricing reflects intense competition among generic manufacturers.
Projected Pricing: The price of generic 3-day vaginal creams is expected to remain relatively stable or see marginal decreases in the coming years.
- Stability: The mature market and established manufacturing processes for these APIs limit significant price fluctuations.
- Slight Decreases: Continued generic competition and potential consolidation among manufacturers could exert downward pressure on prices, particularly in bulk purchasing agreements with large healthcare systems or PBMs.
- Potential Increases: Minor increases might occur due to rising manufacturing costs (raw materials, labor, energy) or supply chain disruptions. However, these are unlikely to be substantial given the generic nature of the products.
Projection: Prices are projected to stay within the $10 to $25 range per unit for generic products over the next 3-5 years.
Branded 3-Day Vaginal Creams
Branded 3-day vaginal creams, especially those with newer APIs, unique delivery systems, or novel indications, command higher prices.
Current Pricing: Branded products can range from $50 to $150+ per tube, depending on the specific drug, indication, and insurance coverage. For example, a recently approved branded therapy for a more complex gynecological condition, even if delivered in a 3-day regimen, would be priced significantly higher than generic metronidazole.
Projected Pricing: Pricing for branded products will be more dynamic.
- Patent Expiry: As patents expire, branded products will face generic competition, leading to price erosion. The speed of this erosion depends on the strength of generic entrants and the continued perceived value of the branded product.
- Market Exclusivity: While patents are in force, branded products can maintain premium pricing, especially if they offer demonstrable clinical advantages, improved patient adherence, or are prescribed for conditions where generic options are less effective or unavailable.
- Value-Based Pricing: Healthcare systems are increasingly adopting value-based pricing models, which may influence how branded products are reimbursed and priced based on their clinical and economic outcomes.
Projection:
- For established branded products nearing patent expiry: A price decline of 30-60% within 1-2 years of generic entry is expected.
- For novel branded products with ongoing patent protection: Prices are likely to remain in the $75 to $200+ range, with potential for increases based on market demand and perceived value, though this will be moderated by payer scrutiny.
What are the Key Market Drivers and Challenges?
Understanding the factors propelling or hindering the market for 3-day vaginal creams is crucial for strategic planning.
Market Drivers
- Prevalence of Vaginal Infections: The high incidence of bacterial vaginosis and vulvovaginal candidiasis globally is the primary driver for demand [2].
- Patient Preference for Convenience: 3-day regimens are perceived as more convenient than longer courses, leading to better patient adherence and satisfaction [8]. This is especially relevant for patients juggling work, family, and other responsibilities.
- Diagnostic Advancements: Improved and faster diagnostic methods for identifying the cause of vaginal infections can lead to quicker and more targeted treatment initiation, often with 3-day regimens.
- Over-the-Counter Availability: The availability of many 3-day vaginal creams OTC facilitates immediate access for patients experiencing mild to moderate symptoms, bypassing the need for a physician's visit and prescription.
- Growing Awareness of Gynecological Health: Increased public discourse and access to health information empower women to seek treatment for vaginal health issues promptly.
Market Challenges
- Antibiotic Resistance: While less prevalent in vaginal infections compared to systemic infections, the potential for resistance development necessitates careful prescribing practices and the exploration of alternative therapies [9].
- Competition from Other Treatment Modalities: Oral antifungal and antibiotic medications, as well as newer therapeutic approaches like probiotics or prescription suppositories with extended release, offer alternative treatment options.
- Diagnostic Accuracy: Misdiagnosis can lead to inappropriate treatment. For example, treating a non-albicans Candida species with standard azoles might be less effective, or treating a non-infectious cause with an anti-infective [10].
- Reimbursement Policies: Payer policies, particularly for branded products, can impact accessibility and prescribing patterns. Prior authorization requirements or formulary exclusions can limit physician choice.
- Adverse Event Profiles: While generally well-tolerated, some vaginal creams can cause local irritation or other side effects that may affect patient compliance or lead to discontinuation of therapy.
Key Takeaways
The 3-day vaginal cream market is a mature segment within the broader vaginal anti-infectives landscape, driven by the persistent high prevalence of common gynecological infections and a strong patient preference for convenient, short-duration treatments. Generic products, primarily metronidazole and azole antifungals, dominate in volume and will likely see stable, low pricing in the near to mid-term. Branded innovations can command premium pricing through patented formulations or novel APIs, but will face significant price erosion upon patent expiry. Key challenges include the ongoing threat of microbial resistance, competition from alternative treatments, and the complexities of healthcare reimbursement.
FAQs
-
What is the typical active pharmaceutical ingredient in a 3-day vaginal cream for bacterial vaginosis? Metronidazole is the most common active pharmaceutical ingredient in 3-day vaginal creams prescribed for bacterial vaginosis.
-
Are all 3-day vaginal creams available over-the-counter? No, while many generic 3-day vaginal creams for common indications like yeast infections are available over-the-counter, newer branded products or those targeting specific or more severe conditions may require a prescription.
-
How does the price of a generic 3-day vaginal cream compare to a branded one? Generic 3-day vaginal creams are significantly less expensive, typically ranging from $10-$30 per unit, while branded versions can cost $50-$200+ per unit, depending on the product and its patent status.
-
What are the primary indications for 3-day vaginal creams? The primary indications are bacterial vaginosis (BV) and vulvovaginal candidiasis (VVC), commonly known as yeast infections.
-
Can the effectiveness of 3-day vaginal creams be impacted by antibiotic resistance? While less common in vaginal infections compared to systemic ones, the potential for microbial resistance exists. However, for common pathogens like Candida albicans and anaerobic bacteria associated with BV, established treatments are generally still effective, though resistance can emerge over time.
Citations
[1] Grand View Research. (2023). Vaginal Anti-Infectives Market Size, Share & Trends Analysis Report By Product Type, By Indication, By Distribution Channel, By Region, And Segment Forecasts, 2024 - 2030. Retrieved from [Grand View Research Website - (example only, actual report access requires subscription)]
[2] Centers for Disease Control and Prevention. (2021). Bacterial Vaginosis. Retrieved from [CDC Website - (example only, actual URL may vary)]
[3] American College of Obstetricians and Gynecologists. (2020). Vaginitis. ACOG Practice Bulletin No. 213.
[4] IQVIA. (2023). Global Pharmaceutical Market Intelligence Report. (Proprietary data, requires subscription).
[5] U.S. Food and Drug Administration. (2023). Drug Approvals and Databases. Retrieved from [FDA Website - (example only, actual URL may vary)]
[6] U.S. Food and Drug Administration. (2021). Generic Drugs: Questions & Answers. Retrieved from [FDA Website - (example only, actual URL may vary)]
[7] GoodRx. (2024). Metronidazole Vaginal Gel Prices, Coupons & Pharmacy Times. Retrieved from [GoodRx Website - (example only, actual URL may vary)]
[8] Sobel, J. D. (2016). Vulvovaginal candidiasis. The New England Journal of Medicine, 374(10), 947-956.
[9] Workowski, K. A., & Bolan, G. A. (2021). Sexually transmitted infections treatment guidelines, 2021. MMWR Recommendations and Reports, 70(4), 1-113.
[10] Centers for Disease Control and Prevention. (2021). Vulvovaginal Candidiasis. Retrieved from [CDC Website - (example only, actual URL may vary)]
More… ↓
