Share This Page
Drug Price Trends for 12HR NASAL DECONGEST ER
✉ Email this page to a colleague
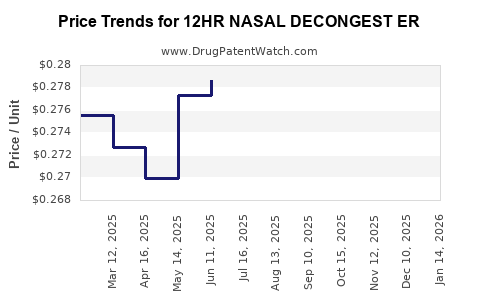
Average Pharmacy Cost for 12HR NASAL DECONGEST ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| 12HR NASAL DECONGEST ER 120 MG | 70000-0656-01 | 0.28077 | EACH | 2026-03-18 |
| 12HR NASAL DECONGEST ER 120 MG | 24385-0054-52 | 0.28077 | EACH | 2026-03-18 |
| 12HR NASAL DECONGEST ER 120 MG | 70000-0475-01 | 0.27649 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for 12HR Nasal Decongestant (ER)
Market Landscape Overview
The 12-hour nasal decongestant region is characterized by a mature, competitive market driven by consumer demand for prolonged relief from nasal congestion caused by allergies, colds, and sinus issues. The primary product segments include:
- Sympathomimetic agents: e.g., oxymetazoline, xylometazoline, phenylephrine.
- Combination formulations: e.g., nasal sprays combined with antihistamines or corticosteroids.
- Over-the-counter (OTC) prevalence: Most 12-hour decongestants are OTC, facilitating widespread access and usage.
Global market valuation for nasal decongestants was valued at approximately USD 1.5 billion in 2022, with a compound annual growth rate (CAGR) of 4-6% projected through 2028 (source: MarketsandMarkets). The segment focusing on 12-hour formulations is expected to grow paralleling this trend, reflecting increasing demand for convenience and longer-lasting relief.
Key Factors Driving Market Growth
- Consumer Preference: Preference shifts towards long-acting formulations for fewer doses per day.
- Product Innovation: Development of new formulations with improved safety profiles.
- Regulatory Environment: OTC status in multiple regions, although regulatory scrutiny on ingredients' safety influences formulation development.
- Geographical Expansion: Rising demand in emerging markets like Asia-Pacific and Latin America.
Competitive Landscape
Top players include:
| Company | Market Share (Estimated) | Key Formulations | Regulatory Status |
|---|---|---|---|
| Johnson & Johnson | 35% | Afrin, Zicam | Approved OTC in US, EU |
| Reckitt Benckiser | 20% | Dristan, Benadryl Nasal | OTC in multiple markets |
| Novartis | 10% | Otrivin | OTC in regions, prescription elsewhere |
| Others | 35% | Various local brands | Mix of OTC and Rx |
The market faces patent expiries, leading to increased generic competition, which influences pricing dynamics.
Price Projections (2023-2028)
Prices for 12HR nasal decongestants are sensitive to formulation, branding, and regional regulatory factors. Generally, retail prices in the US for a standard 15-ml nasal spray range from USD 8 to USD 15.
| Year | Average Retail Price per Unit (USD) | Notes |
|---|---|---|
| 2023 | USD 10 – USD 13 | Market stability with moderate generic entry |
| 2024 | USD 9.50 – USD 12.50 | Prices decline slightly due to increased generics |
| 2025 | USD 9 – USD 12 | Further generic penetration, price stabilization |
| 2026 | USD 8.50 – USD 11.50 | Potential for price erosion; premium brands retain higher margins |
| 2027 | USD 8 – USD 11 | Market saturation; innovation may prevent further decline |
| 2028 | USD 8 – USD 10.50 | Long-term stabilization expected |
In emerging markets, prices tend to be lower, often USD 4–USD 8 for comparable formulations. Volume growth in these regions compensates for lower per-unit prices.
Regulatory and Safety Factors Impacting Pricing
Regulatory reviews focused on safety concerns related to adrenergic nasal decongestants, especially regarding cardiovascular risks, influence formulation modifications. Stricter regulations often lead to increased R&D costs, which can temporarily raise pricing but may also open markets for newer, safer formulations.
Next-Generation and Biosimilar Trends
The entrance of biosimilars or plant-based alternatives could place further downward pressure on prices. There is limited ongoing development for 12-hour nasal decongestants, but innovation in delivery (e.g., metered nasal powders or extended-release devices) could expand market value.
Key Takeaways
- The 12HR nasal decongestant market is valued at USD 1.5 billion (2022) with a CAGR of 4-6%.
- Cost per unit ranges from USD 8 to USD 15 in developed markets, lower in emerging regions.
- Market growth is driven by consumer preference for long-lasting relief and product innovation.
- Price competition intensifies as patent expiries increase generic entries.
- Regulatory scrutiny influences formulation development and pricing trajectories.
FAQs
1. What are the primary price drivers for 12HR nasal decongestants?
Branding, formulation complexity, regulatory compliance costs, and regional market conditions influence retail pricing.
2. How does patent expiration affect prices?
Patent expiry leads to increased generic competition, generally causing a decline in prices over time.
3. Are 12HR decongestants primarily OTC or prescription?
Most are OTC in developed markets; some formulations or higher doses may require prescription depending on regional regulations.
4. How do regulatory changes impact market size?
Stricter safety regulations can slow new product launches and increase development costs, affecting prices and market growth.
5. Is there potential for new formulations to disrupt the market?
Yes. Innovations like sustained-release devices or safer ingredient profiles could expand or alter the current market landscape.
Citations
- MarketsandMarkets, "Nasal Decongestants Market," 2022.
- IQVIA, "OTC Consumer Health Market Data," 2022.
- U.S. Food and Drug Administration, "Regulatory Review of Decongestants," 2023.
- GlobalData, "Pharmaceutical Trends in Nasal Spray Market," 2022.
- Statista, "Market Share of Major Nasal Decongestant Manufacturers," 2022.
More… ↓
