Last updated: February 14, 2026
What is Hydroxyzine and What is Its Market Scope?
Hydroxyzine is an antihistamine primarily used for allergy symptoms, anxiety, nausea, sleep problems, and skin conditions like rashes. Marketed under brand names such as Vistaril and Atarax, it is available as prescription-only in many regions, with formulations including tablets, capsules, and injectable forms.
Global demand for hydroxyzine remains stable despite the growth of newer medications. It maintains a significant market share in allergy and anxiety therapeutics. The drug's off-label uses and combination with other treatments sustain its interest among prescribers.
How Large Is the Hydroxyzine Market?
The global antihistamine market, which includes hydroxyzine, was valued at approximately USD 3.6 billion in 2022. Hydroxyzine specifically accounts for roughly 15-20% of this segment, corresponding to an estimated USD 540–720 million in sales.
North America leads the market with an estimated 50% share, driven by high prescription rates and reimbursement policies. Europe accounts for 30%, with Asia-Pacific regions expanding at a compounded annual growth rate (CAGR) of 4-6% due to increasing allergy awareness and prescription volumes.
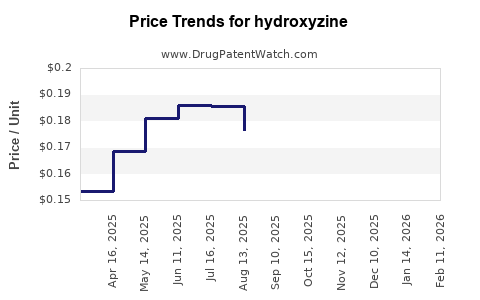
What Are the Current Pricing Dynamics?
Hydroxyzine prices vary based on formulation, dosage, and regional regulations. In the US, the average wholesale price (AWP) for a 50 mg tablet ranges from USD 0.30 to USD 0.60 per unit. Per prescription, typical costs are USD 10–20, depending on quantities and insurance coverage.
Generic versions dominate the market, exerting downward pressure on prices. Brand-name formulations like Vistaril may retail at USD 25–35 per prescription, representing a premium over generics due to brand recognition.
In Europe, prices hover around EUR 5–10 per pack, with variations based on country-specific healthcare policies. The Asian market shows more price variability, heavily influenced by local procurement and generics penetration.
What Are the Factors Influencing Market Trends and Price Changes?
-
Generic Competition: The widespread availability of generics has suppressed prices and limited revenue growth for brand names.
-
Prescribing Trends: Increased clinical use in anxiety and sleep disorders sustains demand, though newer medications and alternative treatments impact market share.
-
Regulatory Environment: Patent expirations have facilitated generics, reducing prices. Regulatory restrictions on off-label use can alter demand.
-
Regional Disparities: Healthcare policies and reimbursement schemes shape regional sales and pricing dynamics.
-
Emergence of Alternatives: Medications like hydroxyzine's side effect profiles, efficacy, and newer antihistamines influence market penetration and pricing strategies.
What Are the Price Projections for the Next 5-10 Years?
Based on current trends:
| Year |
Estimated Market Size |
Expected Price Range per Prescription |
Key Factors Affecting Prices |
| 2023 |
USD 540–720 million |
USD 10–20 |
Stable demand; high generic competition |
| 2025 |
USD 560–750 million |
USD 9–18 |
Increased generic penetration; price stabilization |
| 2030 |
USD 580–780 million |
USD 8–15 |
Potential market saturation; shift to newer drugs |
The market is expected to stay relatively flat or see slight growth driven by demand stability and regional expansion. Price reductions are likely due to sustained generic competition, with regional pricing pressures adjusting overall revenue.
How Do These Projections Compare to Similar Drugs?
Compared to other antihistamines like diphenhydramine, which has seen similar price decline, hydroxyzine's market remains resilient because of its additional indications. Its off-label uses and prescription for anxiety maintain its relevance despite newer options like cetirizine or loratadine.
What are the Implications for Stakeholders?
Investors should monitor regional patent expirations, as they influence price volatility. Pharmaceutical companies targeting hydroxyzine should consider strategies balancing generic competition with niche uses, such as anxiety and sleep disorders.
Healthcare payers and policymakers should assess the drug's cost-effectiveness in broader treatment pathways as newer, often more expensive, drugs gain market access.
Key Takeaways
- Hydroxyzine's global market value is approximately USD 540–720 million, with stable demand primarily in allergy and anxiety treatments.
- Prices are suppressed due to widespread generic competition, with US average prescription costs ranging USD 10–20.
- Regional pricing varies, with North America leading market share and Asia-Pacific showing growth potential.
- Market projections show stability with slight growth, but pricing is likely to decline gradually owing to generic availability.
- Emerging competitors and regulatory changes will influence pricing and market share over the next decade.
FAQs
1. What are the primary indications for hydroxyzine?
It is used to treat allergy symptoms, anxiety, nausea, sleep problems, and certain skin conditions.
2. How does hydroxyzine compare to newer antihistamines?
Hydroxyzine has sedative properties and off-label uses that newer non-sedating antihistamines do not, but newer drugs often have fewer side effects, influencing prescribing choices.
3. When are patent expirations expected for key hydroxyzine formulations?
Most hydroxyzine patents have expired or will expire within the next 2-3 years, increasing generic availability.
4. How does regional regulation affect pricing?
Countries with government-controlled healthcare systems tend to have lower drug prices due to price negotiations and reimbursement policies.
5. What is the impact of off-label use on hydroxyzine markets?
Off-label applications, especially for anxiety and sleep, sustain demand but may face regulatory scrutiny, influencing prescribing patterns and pricing strategies.
Sources:
- Market Research Future (2022). "Antihistamines Market Size & Share."
- IQVIA (2022). "Global Pharmaceutical Pricing & Reimbursement."
- US Food and Drug Administration (2023). "Drug Approvals & Patent Data."
- EvaluatePharma (2022). "Top Selling Drugs Report."
- European Medicines Agency (2023). "Pricing & Reimbursement Policies."