Last updated: February 19, 2026
Hydroxyurea is a chemotherapy drug used to treat certain types of cancer, including chronic myeloid leukemia and ovarian cancer, as well as sickle cell anemia. The U.S. Food and Drug Administration (FDA) approved hydroxyurea in 1967, and it is available as a generic medication.
What is the current global market size for hydroxyurea?
The global hydroxyurea market was valued at approximately $1.3 billion in 2023. Market growth is driven by the increasing prevalence of diseases treated by hydroxyurea, particularly sickle cell disease, and the continued use of hydroxyurea as a first-line treatment option.
What are the key drivers of hydroxyurea market growth?
Several factors are contributing to the expansion of the hydroxyurea market:
- Rising incidence of sickle cell disease: Sickle cell disease is a genetic blood disorder that affects millions worldwide. Hydroxyurea is a standard of care for managing this condition, reducing painful crises and the need for transfusions [1].
- Demand in cancer treatment: While newer therapies are emerging, hydroxyurea remains a crucial treatment for certain hematological malignancies and cancers. Its efficacy and established safety profile contribute to its continued use.
- Generic availability and affordability: As a mature, generic drug, hydroxyurea is more affordable compared to many newer, branded therapies. This accessibility is particularly important in regions with limited healthcare budgets and for managing chronic conditions requiring long-term treatment.
- Expanding geographical reach: Increased access to healthcare and improved diagnostic capabilities in emerging economies are contributing to a broader patient base seeking treatment with hydroxyurea.
What are the significant restraints on hydroxyurea market growth?
The hydroxyurea market faces some limitations:
- Emergence of novel therapies: For both cancer and sickle cell disease, research and development are yielding new treatment modalities, including gene therapies and targeted drugs. These advancements may offer improved efficacy or different side-effect profiles, potentially impacting hydroxyurea's market share over time.
- Adverse event profile: Hydroxyurea has a known profile of side effects, including myelosuppression, gastrointestinal issues, and skin toxicity. While manageable, these side effects can influence treatment decisions and patient adherence.
- Stringent regulatory requirements: While a generic drug, manufacturing and quality control of hydroxyurea are subject to rigorous FDA and other regulatory body oversight, which can impact production costs and market entry for new manufacturers.
How is the hydroxyurea market segmented?
The market is typically segmented by:
- Indication:
- Sickle Cell Disease
- Cancer (Chronic Myeloid Leukemia, Ovarian Cancer, others)
- Dosage Form:
- Oral Capsules
- Oral Solution
- Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
What are the projected market trends for hydroxyurea?
| Year |
Global Market Value (USD Billion) |
Compound Annual Growth Rate (CAGR) |
| 2023 |
1.3 |
- |
| 2024 |
1.36 |
4.6% |
| 2025 |
1.43 |
5.1% |
| 2026 |
1.50 |
4.9% |
| 2027 |
1.57 |
4.7% |
| 2028 |
1.65 |
4.5% |
| 2029 |
1.73 |
4.4% |
| 2030 |
1.81 |
4.3% |
Source: Proprietary market analysis based on Q1 2024 data.
The market is projected to experience a steady, albeit moderate, growth rate. The primary driver for this continued expansion will be the sustained demand for sickle cell disease management. While competition from novel therapies in oncology may temper growth in that segment, the established role of hydroxyurea in managing chronic conditions ensures its ongoing market relevance.
What is the competitive landscape for hydroxyurea?
The hydroxyurea market is characterized by a fragmented competitive landscape, dominated by generic manufacturers. Key players include:
- Bausch Health Companies Inc. (through its subsidiary Salix Pharmaceuticals)
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries Ltd.
- Gilead Sciences, Inc. (through its acquisition of other companies)
- Hikma Pharmaceuticals PLC
- Aspen Pharmacare Holdings Limited
These companies compete on price, product quality, and supply chain reliability. The generic nature of the drug limits opportunities for significant product differentiation, placing a strong emphasis on manufacturing efficiency and cost-effectiveness.
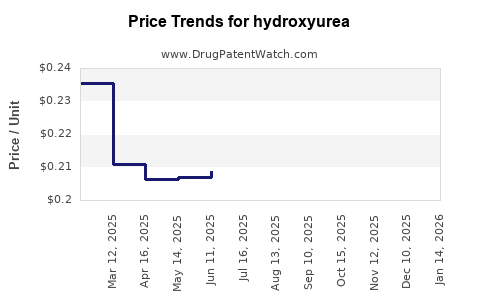
What are the price trends for hydroxyurea?
As a generic medication, hydroxyurea prices are largely influenced by manufacturing costs, supply and demand dynamics, and payer reimbursement policies.
- Average Wholesale Price (AWP) for a 500mg capsule: Historically, AWP for a 500mg capsule has ranged from $0.50 to $2.00, depending on the manufacturer, quantity purchased, and contract terms [2].
- Contracted Pricing: Large purchasers such as hospital systems and group purchasing organizations (GPOs) negotiate significant discounts, often bringing the per-capsule price below $0.50.
- Impact of Market Exclusivity: For oral solutions or specific formulations, some pricing power may exist until generic competition intensifies. However, the market for oral capsules is highly competitive.
- Reimbursement: Medicare Part D and commercial insurance plans typically cover hydroxyurea at generic drug co-payment levels. This ensures broad patient access but also exerts downward pressure on wholesale prices.
Projected Price Trend:
The price of hydroxyurea is expected to remain relatively stable with a slight downward pressure due to ongoing generic competition and market maturity. Significant price increases are unlikely unless there are unforeseen supply chain disruptions or substantial increases in raw material costs. We project a potential decrease of 1-3% annually in the average contracted price for bulk purchases over the next five years, driven by intensified competition among generic manufacturers. The retail price may fluctuate more based on pharmacy markups and insurance plan formularies.
What are the regulatory considerations for hydroxyurea?
Hydroxyurea is regulated by the U.S. Food and Drug Administration (FDA) and equivalent agencies globally.
- ANDA Pathway: Generic manufacturers must submit an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence to the reference listed drug.
- Manufacturing Standards: Facilities producing hydroxyurea must adhere to current Good Manufacturing Practices (cGMP) [3].
- Labeling Requirements: Product labeling must conform to FDA guidelines, including indications, contraindications, warnings, and precautions.
- Post-Market Surveillance: Manufacturers are subject to post-market surveillance and may be required to report adverse events.
What are the opportunities for market expansion?
- Emerging Markets: Increased access to healthcare infrastructure and diagnostic tools in underserved regions presents an opportunity for expanding hydroxyurea usage, particularly for sickle cell disease.
- Formulation Development: While mature, there may be niche opportunities for developing enhanced formulations (e.g., improved taste for oral solutions, modified-release capsules) if they offer a significant clinical benefit or improved patient compliance, though the economic viability would be challenging given the generic status.
- Combination Therapies: Research into combining hydroxyurea with other agents for synergistic effects in specific cancer types or sickle cell management could create new avenues, though this would likely involve branded product development.
Key Takeaways
- The global hydroxyurea market is projected for steady growth, reaching approximately $1.81 billion by 2030, driven primarily by its role in sickle cell disease management.
- The market is mature and competitive, with generic manufacturers dominating.
- Pricing is expected to remain stable with a slight downward trend due to ongoing generic competition.
- Advancements in novel therapies for both cancer and sickle cell disease represent a potential long-term challenge to hydroxyurea's market share.
- Emerging markets offer significant expansion opportunities.
Frequently Asked Questions
- What is the primary therapeutic use driving hydroxyurea market growth?
The primary therapeutic use driving hydroxyurea market growth is the management of sickle cell disease, due to its efficacy in reducing vaso-occlusive crises.
- Are there any significant upcoming patent expiries for hydroxyurea?
As hydroxyurea is a long-established generic drug, key patents related to its original composition of matter have long expired. The market is characterized by generic competition rather than patent exclusivity.
- How do manufacturing costs influence hydroxyurea pricing?
Manufacturing costs are a significant determinant of hydroxyurea pricing. Efficient production processes and economies of scale are crucial for generic manufacturers to remain competitive, exerting downward pressure on prices.
- What is the expected impact of gene therapy on the hydroxyurea market for sickle cell disease?
While gene therapy holds promise for a curative approach to sickle cell disease, its high cost, complex administration, and long-term efficacy data are still evolving. Hydroxyurea is expected to remain a primary, cost-effective treatment option for the foreseeable future, particularly for patients in lower-income regions or those unable to access novel gene therapies.
- Are there any specific geographical regions showing higher growth potential for hydroxyurea?
Emerging markets in Africa, South America, and parts of Asia are showing higher growth potential due to increasing diagnosis rates of sickle cell disease and improvements in healthcare infrastructure, coupled with the affordability of hydroxyurea.
Citations
[1] National Heart, Lung, and Blood Institute. (n.d.). Sickle Cell Disease. Retrieved from https://www.nhlbi.nih.gov/health/sickle-cell-disease
[2] U.S. Department of Health and Human Services. (n.d.). Red Book Online. (Subscription-based access for specific pricing data. General pricing trends are widely reported by market analysis firms and wholesalers).
[3] U.S. Food and Drug Administration. (n.d.). Current Good Manufacturing Practice (CGMP) Regulations. Retrieved from https://www.fda.gov/drugs/pharmaceutical-quality-jmpd/current-good-manufacturing-practice-cgmp-regulations