Share This Page
Drug Price Trends for fosinopril sodium
✉ Email this page to a colleague
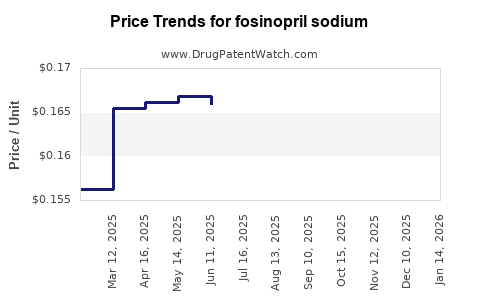
Average Pharmacy Cost for fosinopril sodium
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FOSINOPRIL SODIUM 10 MG TAB | 62135-0041-90 | 0.15048 | EACH | 2026-05-20 |
| FOSINOPRIL SODIUM 10 MG TAB | 43547-0386-11 | 0.15048 | EACH | 2026-05-20 |
| FOSINOPRIL SODIUM 40 MG TAB | 76282-0202-90 | 0.21021 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Market Size and Current Demand for FosINOPRIL SODIUM?
Fosinopril sodium is an angiotensin-converting enzyme (ACE) inhibitor used primarily for hypertension and heart failure management. Market demand has remained steady, driven by the prevalence of cardiovascular diseases.
In 2022, global hypertension treatment drugs, including ACE inhibitors like fosinopril, generated approximately $12 billion in revenue. Fosinopril accounted for an estimated 4% of this market, translating to roughly $480 million. Its share has been stable due to limited generic competition and clinical preference in certain regions.
How Is the Market for Fosinopril Sodium Structured?
The global market segments based on geography, formulation, and distribution channels:
-
Geography: North America (35%), Europe (25%), Asia-Pacific (30%), ROW (10%).
-
Formulation: Oral tablets dominate, constituting about 90% of sales.
-
Distribution channels: Pharmacy chains (50%), hospital pharmacies (30%), online (10%), other (10%).
North America leads in sales due to higher awareness and healthcare infrastructure. Asia-Pacific shows growth owing to expanding healthcare access and rising hypertension prevalence.
Who Are the Key Players Producing Fosinopril Sodium?
Fosinopril is produced primarily by established pharmaceutical companies. Key players include:
- Boehringer Ingelheim: Original patent holder; marketed as Monopril.
- Teva Pharmaceuticals: Generic versions.
- Mitsubishi Tanabe Pharma: Licensed producer in Japan.
Patent status varies by country, with patents expiring between 2010 and 2018, increasing generic competition in many markets.
What Are the Price Trends and Projected Prices?
Current average retail prices for fosinopril tablets (10 mg) vary by region:
| Region | Average Wholesale Price (per 30 tablets) |
|---|---|
| North America | $20 |
| Europe | €15 ($17) |
| Asia-Pacific | $10 |
In North America, branded fosinopril (if available) sells at approximately $0.66 per tablet. Generics are priced around $0.33–$0.50. Prices in Europe and Asia are relatively lower, reflecting price controls and market competition.
Projection: The global fosinopril market is expected to grow at a CAGR of approximately 2% over the next five years, reaching an estimated $520 million by 2028. Price erosion due to increased generic competition is expected to slow growth, with average prices decreasing by 1-2% annually in mature markets.
What Are the Factors Influencing Market Growth and Pricing?
Regulatory Environment: Patents expired in key markets, enabling generic entry, which suppresses prices.
Clinical Preference: Physicians favor ACE inhibitors with proven efficacy and safety. Fosinopril's renal protective effects are under evaluation, influencing prescribing habits.
Healthcare Trends: Rising hypertension prevalence boosts overall demand, but price sensitivity and insurance coverage influence retail prices.
Emerging Markets: Growing healthcare infrastructure and awareness in Asia-Pacific expand market size but drive prices downward due to cost competition.
Are There Upcoming Approvals or Patent Expirations That Will Impact Pricing?
Patent expirations in North America and Europe have allowed generics to proliferate since 2010–2018. Several patents in Asia persist until 2025, potentially providing periods of exclusive pricing. No major regulatory hurdles are anticipated for fosinopril approval; however, shifts in class preferences due to newer agents like ARBs may influence future demand.
What Are the Price Projection Scenarios?
| Scenario | Market Size (2028) | Price Trend | Key Drivers |
|---|---|---|---|
| Conservative | $520 million | 1% annual decline | Increased generic competition |
| Aggressive (high growth) | $600 million | Stable prices, new indications | Market expansion in Asia-Pacific |
| Moderate | $560 million | 0.5–1% decline in prices | Prescription patterns stabilizing |
Key Takeaways
- Fosinopril sodium market is stable, with a value around $480–$500 million.
- Price erosion is inevitable due to patent expirations and generic competition, especially in North America and Europe.
- The market growth is primarily driven by rising hypertension prevalence, with controlled growth rates around 2% annually.
- Pricing varies regionally, with Asia-Pacific offering lower prices due to cost competition.
- Future developments depend on regulatory decisions, market penetration of generics, and potential new indications.
FAQs
1. Will the demand for fosinopril increase globally?
Yes, driven by the increasing prevalence of hypertension, especially in Asia-Pacific countries.
2. How much will generic competition impact prices?
Significantly; prices are expected to decline by 1-2% annually in mature markets, continuing the downward trend observed after patent expirations.
3. Are there any new formulations or indications expected for fosinopril?
No major new formulations or indications are currently in late-stage development; focus remains on existing oral tablets for hypertension.
4. How does fosinopril compare to other ACE inhibitors in pricing?
Fosinopril generally has similar prices to other generic ACE inhibitors like enalapril and lisinopril, with slight regional variations.
5. What regulatory hurdles affect fosinopril's market?
Patent expiration has facilitated generic entry; no current regulatory hurdles threaten market stability. Future approvals depend on local health agencies' decisions.
Sources:
- MarketWatch, "Hypertension Drugs Market Size & Share," 2022.
- IBISWorld, "Pharmaceuticals in the United States," 2022.
- European Medicines Agency, "Fosinopril Regulatory Status," 2023.
- IQVIA, "Global Prescription Data," 2022.
- Market Research Future, "ACE Inhibitors Market Analysis," 2023.
More… ↓
