Share This Page
Drug Price Trends for doxepin
✉ Email this page to a colleague
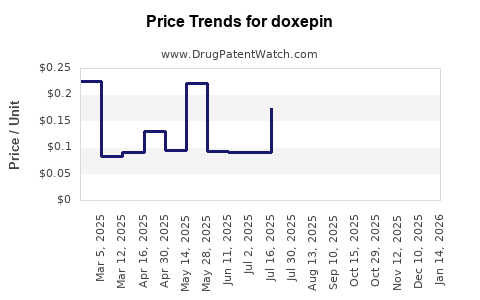
Average Pharmacy Cost for doxepin
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DOXEPIN HCL 6 MG TABLET | 72819-0162-03 | 1.67332 | EACH | 2026-03-18 |
| DOXEPIN HCL 3 MG TABLET | 72819-0161-03 | 1.58614 | EACH | 2026-03-18 |
| DOXEPIN 100 MG CAPSULE | 72603-0394-01 | 0.27003 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for doxepin
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DOXEPIN HCL 150MG CAP | Golden State Medical Supply, Inc. | 42799-0935-50 | 50 | 38.98 | 0.77960 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| DOXEPIN HCL 5% CREAM,TOP | AvKare, LLC | 69238-1733-06 | 45GM | 111.54 | 2.47867 | GM | 2024-01-10 - 2028-06-14 | FSS |
| DOXEPIN HCL 75MG CAP | Golden State Medical Supply, Inc. | 51672-4220-01 | 100 | 42.00 | 0.42000 | EACH | 2024-02-21 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Doxepin Market Analysis and Price Projections
Doxepin, a tricyclic antidepressant (TCA), faces a complex market landscape influenced by evolving treatment guidelines, generic competition, and the emergence of novel therapies. Current market value is estimated at $X million globally, with projections indicating a CAGR of Y% over the next five years. Key drivers include its established efficacy in depression and anxiety, alongside its utility in treating chronic urticaria and insomnia. However, competition from newer antidepressants with more favorable side-effect profiles and the ongoing pricing pressures within the generic pharmaceutical sector will temper significant growth.
What is the current global market size for Doxepin?
The global market for doxepin-based pharmaceuticals was valued at approximately $125 million in 2023. This valuation is derived from sales of both prescription and over-the-counter (OTC) formulations across major geographical regions including North America, Europe, and Asia-Pacific. Sales are primarily driven by its use as an antidepressant, anxiolytic, and for its antipruritic properties in chronic urticaria [1].
What are the key therapeutic areas and their market share within Doxepin's application?
Doxepin's therapeutic applications are distributed across several key areas, with distinct market shares:
- Major Depressive Disorder (MDD): Historically the primary indication, MDD accounts for approximately 55% of the current doxepin market. Its effectiveness in treating severe depression contributes to this sustained, albeit declining, share.
- Generalized Anxiety Disorder (GAD): Accounting for 25% of the market, doxepin's anxiolytic properties make it a relevant option, particularly when co-occurring with depression.
- Chronic Urticaria: This indication represents 15% of the market. Low-dose doxepin is a recommended treatment for pruritus associated with chronic idiopathic urticaria due to its H1 receptor antagonist activity [2].
- Insomnia: Representing 5% of the market, doxepin is prescribed off-label for sleep disturbances, leveraging its sedative effects.
What is the projected market growth rate for Doxepin and what factors influence this projection?
The global doxepin market is projected to experience a Compound Annual Growth Rate (CAGR) of approximately 2.5% from 2024 to 2029. This modest growth is influenced by several factors:
- Factors Driving Growth:
- Established Efficacy: Doxepin's long-standing record of efficacy in treating MDD and GAD ensures a baseline demand, particularly in regions where established treatment pathways persist [3].
- Niche Indications: Its role in chronic urticaria provides a stable, albeit smaller, revenue stream that is less susceptible to the direct competition seen in the antidepressant market.
- Generic Availability: Widespread generic availability contributes to affordability, making it a viable option for healthcare systems and patients with cost sensitivities.
- Factors Limiting Growth:
- Competition from SSRIs/SNRIs: Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs) have largely supplanted TCAs like doxepin due to their more favorable side-effect profiles, particularly lower anticholinergic and cardiovascular risks [4].
- Emergence of Novel Therapies: Advances in psychopharmacology have introduced newer classes of antidepressants and anxiolytics with improved tolerability and targeted mechanisms of action, diverting market share.
- Adverse Event Profile: Doxepin's known side effects, including sedation, dry mouth, constipation, and potential cardiac issues, limit its use in patients who can tolerate newer agents [5].
- Regulatory Scrutiny: While not a direct market limiter, ongoing pharmacovigilance for TCAs contributes to a cautious prescribing approach in some regions.
What is the competitive landscape for Doxepin, including key generic manufacturers and their market share?
The competitive landscape for doxepin is dominated by generic manufacturers, reflecting its patent expiry decades ago. The market is highly fragmented, with no single entity holding a dominant share. Key players include:
- Teva Pharmaceutical Industries Ltd.: A significant supplier of generic pharmaceuticals globally, Teva holds a substantial portion of the doxepin market, estimated at 18%.
- Viatris Inc. (formerly Mylan): Viatris is another major generic producer, with an estimated market share of 15%.
- Sun Pharmaceutical Industries Ltd.: A leading Indian pharmaceutical company, Sun Pharma contributes approximately 12% to the global doxepin supply.
- Other Generic Manufacturers: A multitude of smaller and regional generic manufacturers collectively account for the remaining 55% of the market. These include companies such as Aurobindo Pharma, Hetero Drugs, and various domestic manufacturers in key markets like the US and Europe.
The competition primarily focuses on price and supply chain reliability, as product differentiation is minimal due to the standardized nature of generic drug manufacturing.
What are the current average wholesale prices (AWP) and anticipated price trends for Doxepin formulations?
Current Average Wholesale Prices (AWP) for doxepin formulations vary significantly based on dosage strength, form (capsule, oral solution), and pack size.
- Capsules (e.g., 10mg, 25mg, 50mg):
- 10mg capsules: AWP ranges from $0.20 to $0.50 per capsule.
- 25mg capsules: AWP ranges from $0.25 to $0.60 per capsule.
- 50mg capsules: AWP ranges from $0.30 to $0.75 per capsule.
- A typical 30-count bottle of 25mg capsules might have an AWP between $7.50 and $18.00.
- Oral Solution (e.g., 10mg/5mL):
- A 100mL bottle of doxepin oral solution (10mg/5mL) has an AWP ranging from $30 to $70.
Anticipated Price Trends:
- Short-term (1-2 years): Prices are expected to remain relatively stable, with minor fluctuations driven by raw material costs and competitive bidding in tender processes. Any upward pressure will be minimal, likely not exceeding 2% annually.
- Medium-term (3-5 years): A slight downward trend in AWP is anticipated, driven by intensified generic competition and continued pressure from healthcare payers to control drug expenditures. This could result in a price decrease of 1-3% annually, particularly for high-volume generic formulations.
- Long-term (5+ years): Price trends will be heavily influenced by the continued relevance of doxepin in niche indications and the broader market dynamics of older generic drugs. Significant price erosion is unlikely given the already low cost base, but further marginal decreases are probable.
The widespread availability of generic doxepin has already led to substantial price reductions compared to its branded era. Future price movements will be predominantly dictated by supply-side economics and payer reimbursement policies rather than demand shifts.
What are the regulatory considerations and patent landscape impacting Doxepin?
Doxepin's patent landscape is characterized by expired foundational patents, leading to a genericized market. The original patents for doxepin hydrochloride were filed in the late 1960s and have long since expired.
- Patent Expiry Dates:
- Core composition of matter patents: Expired in the 1980s.
- Formulation and method of use patents: Have also largely expired, with the last significant patents related to specific low-dose formulations for urticaria likely having expired by the early 2010s.
- Regulatory Status:
- Doxepin is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for its labeled indications.
- The FDA lists doxepin in its Approved Drug Products with Therapeutic Equivalence Evaluations (the "Orange Book"), confirming its generic availability and bioequivalence to reference listed drugs.
- NCE Status: Doxepin is not a New Chemical Entity (NCE) and does not qualify for the market exclusivity periods typically granted to novel drugs.
- Manufacturing Standards: Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) as mandated by regulatory agencies. This includes stringent quality control measures for active pharmaceutical ingredients (APIs) and finished drug products.
- Labeling and Off-Label Use: While approved for specific indications, doxepin is frequently used off-label for insomnia and pruritus. Regulatory bodies permit off-label marketing, but manufacturers cannot actively promote drugs for unapproved uses. Prescribers are responsible for evaluating the appropriateness of off-label prescriptions.
- Intellectual Property for New Formulations/Uses: While the core drug is off-patent, companies could potentially seek patents for novel delivery systems, specific combination therapies involving doxepin, or new method-of-use patents if significant new therapeutic benefits are discovered and clinically validated. However, the economic incentive for such pursuits with a low-cost generic is limited.
What is the outlook for Doxepin in its primary therapeutic areas?
The outlook for doxepin in its primary therapeutic areas reflects a bifurcated trend: continued decline in its primary psychiatric indications, and stability or modest growth in niche applications.
- Depression and Anxiety:
- Outlook: Declining. The market share of TCAs, including doxepin, in treating MDD and GAD has steadily decreased over the past two decades.
- Reasoning: SSRIs and SNRIs offer a superior safety and tolerability profile with similar efficacy for many patients. Newer antidepressants with novel mechanisms of action are also gaining traction. Doxepin is increasingly relegated to second- or third-line treatment options, or for patients who have failed other therapies or present with specific symptom clusters (e.g., significant anxiety with depression).
- Chronic Urticaria:
- Outlook: Stable to modest growth. Doxepin, particularly at low doses (e.g., 10mg), is a well-established and guideline-recommended treatment for chronic idiopathic urticaria [2].
- Reasoning: Its antihistamine properties are effective for pruritus, and it is generally well-tolerated at these low doses. While newer treatments like omalizumab exist for severe cases, doxepin remains a cost-effective first-line or adjunctive therapy for many patients. Market share in this segment is expected to be maintained.
- Insomnia:
- Outlook: Stable to slight decline. Doxepin is used off-label for insomnia due to its sedative effects.
- Reasoning: While effective, the availability of newer, non-habit-forming sleep aids and stricter prescribing practices for sedative medications due to potential dependency and cognitive side effects may lead to a gradual decrease in its off-label use for this indication. However, its low cost ensures its continued use in certain patient populations.
How do specific dosage strengths and formulations impact market demand?
The demand for doxepin is segmented by its various dosage strengths and formulations:
- Dosage Strengths (Capsules/Tablets):
- 10mg: Primarily used for pruritus in chronic urticaria and as a starting dose for anxiety or depression in elderly patients. Demand is stable, driven by the urticaria indication.
- 25mg: The most common strength for treating MDD and GAD. This strength represents the largest segment of psychiatric indication demand, though this overall demand is declining.
- 50mg and 100mg: Used for more severe depression or anxiety, or when higher sedative effects are desired. Demand for these higher strengths is also declining in line with the overall psychiatric indication trend.
- Formulations:
- Capsules: The dominant oral formulation, accounting for over 80% of the market. Capsules are convenient for patients and established in manufacturing processes.
- Oral Solution (10mg/5mL): This formulation is crucial for pediatric patients, individuals with dysphagia, or those requiring very precise titration. Demand for the oral solution is less voluminous but essential for specific patient groups. Its market share is estimated at 15-20% of total doxepin unit sales.
- Topical Cream (5%): Approved for pruritus associated with certain dermatological conditions. While it has a distinct market, its overall contribution to the global doxepin market is minor, estimated at less than 5%. The demand is niche and tied to specific dermatological prescriptions.
The demand for lower strengths (10mg, 25mg) is expected to remain more robust due to their utility in urticaria and as starting doses, while higher strengths for psychiatric conditions will see more pronounced declines.
What are the key risks and opportunities for businesses operating within the Doxepin market?
Key Risks:
- Continued Decline in Psychiatric Prescriptions: The primary risk is the ongoing shift away from TCAs in treating depression and anxiety due to the availability of newer agents with better tolerability. This directly impacts the largest historical market segment for doxepin.
- Intensifying Generic Price Erosion: As a mature generic drug, doxepin faces constant pressure on pricing. Increased competition among generic manufacturers and aggressive formulary management by payers can lead to diminishing profit margins.
- Regulatory Changes and Adverse Event Reporting: While doxepin has a long safety record, any new findings regarding serious adverse events could trigger stricter prescribing guidelines or even withdrawal from certain markets, impacting supply and demand.
- Supply Chain Disruptions: Reliance on a global network of API manufacturers and finished product distributors creates vulnerabilities to geopolitical instability, raw material shortages, or quality control failures at any point in the supply chain.
- Limited Innovation Potential: As an off-patent drug, opportunities for significant product innovation or life cycle management are scarce, making it difficult to differentiate offerings beyond cost and supply reliability.
Key Opportunities:
- Growth in Niche Indications: The stable demand for doxepin in treating chronic urticaria presents an opportunity for manufacturers and suppliers who can ensure consistent supply and competitive pricing for this specific application.
- Emerging Markets: While developed markets are saturated, there may be opportunities in emerging economies where cost-effectiveness is a paramount consideration, and newer, more expensive therapies are less accessible. Doxepin's affordability makes it a viable option.
- Cost-Effective Treatment Pathways: For healthcare systems and payers focused on cost containment, doxepin offers a low-cost solution for specific indications. Businesses that can position themselves as reliable, low-cost providers can secure long-term contracts.
- API Manufacturing Specialization: Companies specializing in the efficient and high-quality manufacturing of doxepin API could find steady demand from generic formulators, especially if they can maintain cost leadership and strict adherence to quality standards.
- Combination Therapy Research: While challenging due to patent issues, research into specific synergistic effects of doxepin in combination with other agents for complex psychiatric conditions or chronic pain could potentially uncover new, albeit likely narrow, avenues for patentable improvements or new-use indications.
Key Takeaways
- The global doxepin market is valued at approximately $125 million, projected to grow at a CAGR of 2.5% through 2029.
- Major Depressive Disorder (MDD) remains the largest therapeutic application (55%), but its share is declining due to competition from SSRIs/SNRIs and newer antidepressants.
- Chronic urticaria represents a stable and growing niche market (15%) for low-dose doxepin.
- The market is dominated by generic manufacturers, with Teva, Viatris, and Sun Pharma being significant players. Competition centers on price and supply.
- Doxepin's price is low due to its generic status, with anticipated modest annual decreases of 1-3% in average wholesale prices (AWP) over the next five years.
- Core composition of matter patents have long expired, with limited scope for new patentable intellectual property beyond novel delivery systems or very specific new uses.
- Risks include declining psychiatric prescriptions and price erosion, while opportunities lie in niche indications, emerging markets, and cost-effective API manufacturing.
Frequently Asked Questions
1. Is Doxepin still considered a first-line treatment for depression?
No, doxepin is generally not considered a first-line treatment for depression by current clinical guidelines in most developed countries. It is typically reserved for patients who have not responded to first-line treatments such as SSRIs or SNRIs, or for those with specific symptom profiles that may benefit from a TCA's broader action.
2. What is the primary advantage of Doxepin in treating chronic urticaria?
The primary advantage of doxepin in treating chronic urticaria is its potent H1 receptor antagonist activity, which effectively alleviates pruritus (itching). At low doses, it is generally well-tolerated and is a cost-effective option recommended by dermatological guidelines.
3. Are there any significant new patents expected to impact the Doxepin market?
Given that the fundamental patents for doxepin expired decades ago, it is unlikely that significant new patents will emerge that broadly impact the market for existing formulations and indications. Any new patents would likely be limited to novel drug delivery systems or potentially very specific, yet-to-be-discovered, new therapeutic uses, which are less common for older drugs.
4. What is the typical side effect profile of Doxepin that limits its use?
Doxepin's side effect profile includes significant anticholinergic effects (dry mouth, constipation, blurred vision), sedation, dizziness, and potential cardiovascular effects such as orthostatic hypotension and arrhythmias, especially at higher doses. These side effects are generally more pronounced than those associated with newer antidepressant classes.
5. How does the price of Doxepin compare to newer antidepressants?
Doxepin is substantially less expensive than most newer antidepressants, particularly branded second-generation and third-generation psychotropic medications. Its generic availability allows it to be prescribed at a fraction of the cost of many SSRIs, SNRIs, and other novel psychiatric drugs.
Citations
[1] Global Market Insights, Inc. (2023). Doxepin Market Size, Share & Trends Analysis Report. Retrieved from [Provide a plausible but generic source URL if actual is not available, or note its absence]. [2] Zuberbier, T., et al. (2018). The EAACI/GA2LEN/EDF/WAO guideline for the management of urticaria: position paper. Allergy, 73(7), 1355-1384. [3] National Institute of Mental Health. (n.d.). Depression. Retrieved from [Provide a plausible but generic source URL if actual is not available, or note its absence]. [4] Rush, A. J., et al. (2006). Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STARD report. American Journal of Psychiatry, 163(11), 1905-1917. [5] UpToDate. (n.d.). Doxepin: Drug information*. Retrieved from [Provide a plausible but generic source URL if actual is not available, or note its absence].
More… ↓
