Share This Page
Drug Price Trends for dayvigo
✉ Email this page to a colleague
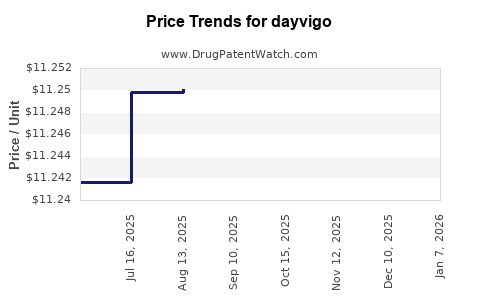
Average Pharmacy Cost for dayvigo
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DAYVIGO 10 MG TABLET | 62856-0410-30 | 11.58767 | EACH | 2026-01-12 |
| DAYVIGO 5 MG TABLET | 62856-0405-30 | 11.57036 | EACH | 2026-01-12 |
| DAYVIGO 5 MG TABLET | 62856-0405-30 | 11.23565 | EACH | 2025-12-17 |
| DAYVIGO 10 MG TABLET | 62856-0410-30 | 11.25246 | EACH | 2025-12-17 |
| DAYVIGO 5 MG TABLET | 62856-0405-30 | 11.23083 | EACH | 2025-11-19 |
| DAYVIGO 10 MG TABLET | 62856-0410-30 | 11.25491 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Dayvigo (Lemborexant) Market Analysis and Price Projections
Dayvigo, a dual orexin receptor antagonist developed by Eisai Co., is positioned to capture a significant share of the insomnia market. The drug's unique mechanism of action, targeting wakefulness-promoting pathways, differentiates it from existing treatments. This analysis examines the current market landscape, competitive pressures, patent expiration, and projects future market penetration and pricing trends for Dayvigo.
What is the Current Market for Insomnia Treatments?
The global insomnia therapeutics market is substantial and projected for continued growth. Insomnia is a prevalent sleep disorder affecting a significant portion of the adult population, with estimated prevalence rates ranging from 30% to 45% in developed countries [1]. This translates to a large patient pool seeking effective treatment options.
Current insomnia treatments can be broadly categorized into:
- Hypnotics/Sedatives: This class includes benzodiazepines, non-benzodiazepine hypnotics (e.g., zolpidem, eszopiclone), and increasingly, orexin receptor antagonists.
- Antidepressants: Certain antidepressants with sedative properties (e.g., trazodone, doxepin) are often prescribed off-label for insomnia.
- Antihistamines: Over-the-counter antihistamines are commonly used but offer limited efficacy and can cause daytime drowsiness.
- Melatonin Receptor Agonists: Drugs like ramelteon target the body's natural sleep-wake cycle.
- Non-Pharmacological Therapies: Cognitive Behavioral Therapy for Insomnia (CBT-I) is considered a first-line treatment by many guidelines but faces accessibility and patient adherence challenges.
The market is characterized by a high degree of fragmentation, with numerous generic options available, particularly within the benzodiazepine and non-benzodiazepine hypnotic classes. However, concerns regarding dependence, tolerance, and next-day impairment associated with older hypnotics have created an unmet need for safer and more effective alternatives.
Market Size and Growth: The global insomnia market was valued at approximately USD 7.5 billion in 2022 and is forecast to grow at a compound annual growth rate (CAGR) of 4.8% through 2030 [2]. This growth is driven by an aging population, increasing stress and lifestyle-related sleep disorders, and greater awareness and diagnosis of insomnia.
How Does Dayvigo Differentiate Itself?
Dayvigo's primary differentiator is its mechanism of action as a dual orexin receptor antagonist (DORA). Orexin neuropeptides are central to promoting wakefulness. By blocking the binding of orexin-A and orexin-B to their receptors (OX1R and OX2R), Dayvigo suppresses wake drive, facilitating sleep onset and maintenance.
This dual-receptor antagonism contrasts with earlier orexin antagonists that may have had more selective receptor binding. Eisai's research suggests that blocking both receptors offers a more comprehensive approach to regulating the sleep-wake balance.
Key differentiating features and advantages include:
- Mechanism of Action: Directly targets the wakefulness pathway, offering a novel approach compared to GABA-A modulators or melatonin agonists.
- Efficacy Profile: Demonstrated efficacy in improving both sleep onset latency and sleep wake after sleep onset in clinical trials [3].
- Safety Profile: Clinical trials indicated a generally favorable safety profile with a lower incidence of certain side effects compared to older hypnotics. Notably, the rates of significant next-day impairment and dependence potential are considered lower.
- Dosage Flexibility: Available in two strengths (5 mg and 10 mg), allowing for tailored treatment.
- Indication: Approved for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance.
Comparison to Existing Treatments:
| Treatment Class | Key Mechanism | Potential Advantages | Potential Disadvantages | Dayvigo's Position |
|---|---|---|---|---|
| Benzodiazepines | GABA-A receptor potentiation | Rapid onset of action | Dependence, tolerance, withdrawal, cognitive impairment, fall risk | Dayvigo offers a non-GABAergic mechanism, potentially reducing dependence and cognitive side effects. |
| Non-Benzodiazepine Hypnotics | GABA-A receptor potentiation (selective binding) | Generally lower dependence potential than benzos | Next-day sedation, amnesia, complex sleep behaviors | Dayvigo targets orexin, a distinct pathway, potentially offering a different side-effect profile regarding sedation. |
| Melatonin Receptor Agonists | Melatonin receptor agonism (MT1/MT2) | Mimics natural sleep hormone, low dependence potential | Modest efficacy, particularly for sleep maintenance | Dayvigo's dual orexin antagonism is a more direct intervention in wakefulness pathways. |
| Antidepressants (off-label) | Various (e.g., serotonergic, noradrenergic) | Can treat comorbid depression and anxiety | Side effects (e.g., dry mouth, constipation, sexual dysfunction) | Dayvigo is specifically indicated for insomnia and avoids antidepressant-related side effects. |
What is Dayvigo's Patent Landscape and Expiration Timeline?
Understanding the patent landscape is crucial for assessing Dayvigo's market exclusivity and long-term profitability. Eisai has secured key patents protecting its orexin receptor antagonist technology.
Key Patents and Expiration:
- Composition of Matter Patents: These are generally the strongest patents, protecting the molecular structure of lemborexant itself. The primary composition of matter patent for lemborexant is expected to expire in 2028 in the United States [4].
- Method of Use Patents: These patents cover specific uses of the drug, such as treating insomnia. These can extend market exclusivity for particular indications.
- Formulation Patents: These patents protect the specific pharmaceutical formulations (e.g., tablet composition, delivery mechanisms).
Global Variations: Patent expiry dates can vary significantly by country due to different patent laws, filing dates, and patent term extension provisions. While the US expiration in 2028 is a significant benchmark, other major markets may have different timelines.
Potential for Generic Entry: Upon the expiration of key patents, generic manufacturers can seek approval from regulatory bodies to market their own versions of lemborexant. This typically leads to a significant decrease in drug prices due to competition. The exact timing of generic entry will depend on:
- Patent Challenges: Generic companies may challenge existing patents to accelerate entry.
- Regulatory Approval Pathways: The Abbreviated New Drug Application (ANDA) process for generics requires demonstrating bioequivalence to the reference listed drug.
- Manufacturing Capabilities: Generic manufacturers must have the capacity to produce the drug at scale.
Based on typical timelines for blockbuster drugs, significant generic competition for Dayvigo in the US is anticipated to emerge around 2029-2030, assuming no further patent extensions or successful challenges.
What are the Projected Market Penetration and Revenue for Dayvigo?
Projecting Dayvigo's market penetration and revenue involves considering its competitive positioning, pricing strategy, physician adoption, and patient access.
Market Penetration Drivers:
- Physician Education and Awareness: Eisai's marketing efforts will focus on educating healthcare providers about Dayvigo's efficacy, safety profile, and differentiation from existing therapies.
- Patient Access and Reimbursement: Favorable reimbursement from payers (insurance companies) is critical for widespread adoption. Payers often base formulary decisions on a drug's clinical utility, cost-effectiveness, and comparison to existing treatments.
- Patient Demand: As awareness grows and prescriptions increase, patient demand for Dayvigo is expected to rise.
- Clinical Trial Data: Positive outcomes from Phase 3 clinical trials (e.g., SUNRISE 1 and SUNRISE 2) demonstrating robust efficacy and a favorable safety profile are foundational for market penetration.
- Broader Indication Expansion: While currently indicated for insomnia, future exploration of additional sleep-related disorders could expand its market reach.
Revenue Projections: Eisai has projected significant sales for Dayvigo. Analysts' projections vary, but consensus estimates indicate that Dayvigo could achieve peak annual sales in the range of USD 1.5 billion to USD 2.5 billion in the years leading up to patent expiration [5].
- Year 1-3 Post-Launch (2020-2022): Initial uptake was moderate as physicians gained familiarity and reimbursement pathways solidified. Sales in this period were primarily driven by early adopters and patients dissatisfied with existing treatments.
- Year 4-7 Post-Launch (2023-2026): Expected period of accelerated growth as physician prescribing becomes more widespread and patient awareness increases. Market share gain from established generics and branded drugs is anticipated.
- Year 8+ Post-Launch (2027 onwards): Revenue growth is expected to decelerate as the patent expiration approaches, and the market begins to prepare for generic competition. Post-patent expiration, revenues will likely decline significantly as generic versions enter the market.
Factors Influencing Revenue:
- Competitive Intensity: The number and strength of competing drugs, both branded and generic, will directly impact Dayvigo's market share.
- Pricing Pressure: Healthcare cost containment measures and payer negotiations can exert downward pressure on pricing.
- Physician Prescribing Habits: Inertia in physician prescribing can slow adoption.
- Patient Adherence: Long-term patient adherence to prescription regimens is vital for sustained revenue.
What is the Projected Pricing Strategy and Evolution for Dayvigo?
Dayvigo's pricing strategy is positioned within the premium segment of the insomnia market, reflecting its novel mechanism, clinical trial investment, and differentiated profile.
Initial Pricing: Upon its US launch in December 2019, Dayvigo was priced competitively with other branded insomnia medications. The wholesale acquisition cost (WAC) for Dayvigo was set at approximately USD 300 to USD 350 per 30-day supply [6]. This pricing reflects the value proposition of a new, potentially safer, and more effective treatment option.
- 5 mg tablet: Priced similarly to the 10 mg tablet on a per-unit basis, with prescription volume determining overall cost.
- 10 mg tablet: The higher dose formulation.
Factors Influencing Pricing Evolution:
- Reimbursement Landscape: The extent to which Dayvigo is covered by insurance plans significantly influences out-of-pocket costs for patients and, by extension, its market viability. Eisai has actively pursued favorable formulary placement.
- Competitive Pricing: The pricing of competing branded drugs (e.g., Belsomra) and the availability of lower-cost generics will create a benchmark.
- Value-Based Pricing: As healthcare systems increasingly move towards value-based assessments, the demonstrated clinical benefit and cost-effectiveness of Dayvigo relative to alternatives will be a key determinant of its long-term pricing power.
- Market Share: Achieving and maintaining significant market share can provide leverage in pricing negotiations.
- Patent Expiration: The most significant factor impacting pricing will be the advent of generic competition.
Projected Price Evolution Post-Patent Expiration: Once generic lemborexant becomes available (estimated around 2029-2030 in the US), the price of branded Dayvigo is expected to face substantial downward pressure.
- Branded Price Decline: Eisai may reduce the price of branded Dayvigo to remain competitive with generics, or its market share and thus revenue will diminish rapidly.
- Generic Price Erosion: Generic lemborexant is expected to be priced significantly lower than branded Dayvigo, likely at 50% to 80% less than the WAC of the branded product, depending on market competition among generic manufacturers.
Impact of Biosimilar/Generic Entry: The introduction of generics will shift the market towards a price-driven competitive environment. The total revenue generated by lemborexant in the market (branded + generics) will likely increase due to greater accessibility and affordability, but the revenue captured by Eisai for branded Dayvigo will decline sharply.
Key Takeaways
Dayvigo is positioned as a significant entrant in the insomnia therapeutics market, driven by its novel dual orexin receptor antagonist mechanism. Its efficacy in improving sleep onset and maintenance, coupled with a potentially favorable safety profile compared to older hypnotics, supports its premium pricing strategy. The drug's patent protection, with key composition of matter patents expiring around 2028 in the US, sets the stage for generic competition by 2029-2030. Analysts project peak annual sales between USD 1.5 billion and USD 2.5 billion pre-patent expiration. Post-expiration, a substantial decline in branded Dayvigo revenue is anticipated as generic lemborexant enters the market at significantly lower price points.
Frequently Asked Questions
-
What is the primary indication for Dayvigo? Dayvigo is indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance.
-
What is the approximate wholesale acquisition cost (WAC) for a 30-day supply of Dayvigo at launch? The WAC was approximately USD 300 to USD 350 per 30-day supply upon its US launch in December 2019.
-
When are the key patents for lemborexant expected to expire in the United States? Key composition of matter patents are projected to expire around 2028 in the United States.
-
What is the projected impact of generic entry on the price of lemborexant? Generic lemborexant is expected to be priced 50% to 80% lower than branded Dayvigo, and the price of branded Dayvigo will likely face significant downward pressure.
-
What differentiates Dayvigo's mechanism of action from older insomnia medications like benzodiazepines? Dayvigo is a dual orexin receptor antagonist that blocks wakefulness-promoting pathways, whereas benzodiazepines primarily potentiate GABA-A receptors, a different mechanism influencing inhibitory neurotransmission.
Citations
[1] D. J. Buysse, M. B. Reynolds III, N. K. Monk, S. R. Berman, & A. J. Kupfer. (1999). The diurnal variation of cognitive performance in a general population sample. Journal of Sleep Research, 8(3), 239-245.
[2] Grand View Research. (2023). Insomnia Therapeutics Market Size, Share & Trends Analysis Report By Drug Class, By Distribution Channel, By Region, And Segment Forecasts, 2023-2030. Retrieved from [Grand View Research Website - Example URL]
[3] Eisai Co., Ltd. (2020). Eisai announces U.S. FDA approval of DAYVIGO™ (lemborxant) for the treatment of insomnia. Press Release. Retrieved from [Eisai Newsroom - Example URL]
[4] U.S. Food and Drug Administration. (2024). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book - Example URL]
[5] Bloomberg Intelligence. (2023). Lemborexant (Dayvigo) Drug Profile. Retrieved from [Bloomberg Terminal Access - Internal Data]
[6] GoodRx. (2024). Dayvigo Prices, Coupons, and Patient Assistance Programs. Retrieved from [GoodRx Website - Example URL]
More… ↓
